Abstract
Summary
Performing stressful procedures in view of cage mates may cause stress in observer animals. However, it is not known if stressful procedures performed in close proximity to, but not in view of cage mates are stressful for the (observer) cage mates. Radiotelemetry and postmortem in vitro studies of the vas deferens were used to determine the effects of stress on observers. Heart rate (HR) and core body temperature (cBT) were recorded for 1 h following weighing of a cage mate or 1 h during restraint of a cage mate and the hour following return of the restrained mouse to the cage. This procedure was repeated daily for 15 days. HR and cBT were increased in observers during both restraint and weighing of cage mates. Analysis of the area under the curve showed that HR and cBT in observers were significantly higher during restraint of a cage mate than after weighing of a cage mate. When mice were returned to the cage after weighing or restraint, HR and cBT were significantly higher in the cage mates of restrained animals. Comparison between days 1, 3, 7 and 14 found that, as the experiment progressed, HR and cBT were significantly reduced in the observer mice during the hour following return of the cage mates after restraint. Results from previous studies have shown that chronic stress causes the vas deferens to become hypersensitive to exogenous application of noradrenaline (NAd). In this study, vas deferens from observers of restraint had a significantly increased response to NAd. These results indicate that stressful procedures should be conducted in isolation from other mice.
The imposition of stress on laboratory animals is unavoidable in animal research. This is due to a number of potentially stress-inducing factors which are present in a typical laboratory environment. Caging, routine cleaning, restraint, noise and lighting have all been shown to have a significant effect on the stress levels and behaviour of laboratory animals (Poole 1997). Even the entry of personnel into a room in which mice are housed has been shown to cause stress in the mice (Kramer et al. 2004). The sympathetic nervous system (SNS) and the hypothalamic-pituitary-adrenocortical axis are the core neuroendocrine components of the stress response (Negrao et al. 2000). Activity in these systems is characterized by the release of adrenaline, noradrenaline (NAd) and corticosterone in rodents (Manser 1992) and is responsible for the well characterized fight or flight response (Henry 1992). Prolonged sympathetic activity due to chronic stress can cause lasting changes in the physiology of the animal. One change that has been observed following chronic stress is hypersensitivity of the vas deferens to NAd in both mice and rats (Singh et al. 2001, Einstein et al. 2004).
Although stress levels can be significantly reduced through the use of appropriate handling techniques and environmental enrichment (Klein et al. 1994, Marashi et al. 2003), handling, injections, restraint and other techniques included in experimental protocols will cause a stress response in the animal being tested. It has also been shown that testing in an experimental animal can lead to a stress response in its cage mates. Sharp et al. (2003) showed that animals witnessing euthanasia of cage mates had increases in heart rate (HR) when compared with those that occurred when the animal itself was weighed and/or handled. Similarly, studies by Zethof et al. (1995) found that when group-housed mice were removed from the cage, one by one, for rectal temperature measurement, those mice removed last from each cage had a higher temperature than those removed first. This was considered to be due to the animals becoming stressed in anticipation of an aversive event, and has been found to occur in many species including rats, rabbits and humans (Snow & Horita 1982, Briese & Cabanac 1991, Marazziti et al. 1992).
Information regarding the effects of a cage mate's distress on mice is currently unavailable. It is possible that non-visual signs of another animal's distress, such as vocalization and pheromone release, may impact on cage mates in close proximity, but not necessarily in visual contact with the distressed animal. This situation may arise in typical situations where animals are removed from the cage for a stressful procedure, such as injection or restraint. The present study was designed to determine whether mice become significantly stressed when in aural and olfactory contact with a cage mate in distress. HR and core body temperature (cBT) were monitored by telemetry to evaluate the acute response to a cage mate's restraint and response of vas deferens to NAd was measured to evaluate long-lasting changes after repeated stress of cage mates.
Materials and methods
In vivo methods
The experiments were approved by the Animal Ethics Committee of the University of Sydney. Eight-week-old BALB/c male mice (>23 g, Animal Resources Centre, Canning Vale, WA, Australia) were used for this study. The mice were housed in opaque mouse cages (width: 14 cm, height: 11 cm, length: 30 cm) with paper bedding and environmental enrichment consisting of plastic shelters and tissues. The cages were changed twice weekly following experimental procedures for those days. Food (standard laboratory chow) and water were available ad libitum and artificial light was provided between 07:00 and 19:00 h. Room temperature was maintained at 21 ± 2°C with humidity approximately 50%. All mice had a seven-day acclimatization period prior to surgery. The mice were randomly divided into two groups: the handling-associated mice (n = 6) and the restraint-associated mice (n = 6). The handling-associated mice were housed with a cage mate that was taken from the cage each day, weighed and returned immediately to the cage. The restraint-associated mice were housed with a cage mate that was taken from the cage each day and subjected to restraint for 1 h, and an untouched cage mate whose purpose was to avoid isolation of the restraint-associated mice during the hour that the other cage mate was restrained.
The mice were anaesthetized using Hypnorm (fentanyl citrate 0.315 mg/mL and fluanisone 10 mg/mL) (Janssen Pharmaceutica, Beerse, Belgium): midazolam (Pharmacia & Upjohn, Bently, WA, Australia): water in the ratio of 1:1:2 (0.07 mL/10 g intraperitoneally). Surgical implantation of radiotelemetry transmitters (Model no. TA10ETA-F20, Data Sciences International, MN, USA) was performed using aseptic techniques as described previously by Kramer et al. (1993), with the mouse placed on a sterile surgical drape over a heat mat (∼30°C, Passwell Pty Ltd, Mt Baker, SA, Australia). Postoperative analgesia (buprenorphine 2 mg/kg intraperitoneally) was administered 6 h after surgery. Following surgery, the mice were allowed 14 days to recover before commencement of handling and restraint procedures on cage mates. Recovery was assessed by return to circadian rhythm of HR and cBT, which was established in all cases by 14 days.
Restraint and handling association procedures
The implanted radiotelemetry transmitters were used to measure HR and cBT of the handling- and restraint-associated mice. Ten-second samples of HR and cBT values were recorded at 5 min intervals for the entire experiment. The restraint and handling association procedures were started between 08:00 h and 09:00 h throughout the experiment.
The cages containing the handling- or restraint-associated mice and their cage mates were moved into a procedure room adjacent to the home room. The room consisted of a bench for restraining and a computer for recording, with the experimenter only being present during the time required for weighing and restraining the animals. Light intensity and background noise were approximately equal to those in the home room, with all conditions being equal for both groups. The mice were allowed a settling time of 30 min after which a 1 h baseline telemetry recording was taken. Following baseline recording, the cage mate of the handling- or restraint-associated mouse was subjected to either the handling or restraint procedure. The handling- and restraint-associated mice were untouched throughout the entire procedure, except for being weighed on cage change days.
The handling procedure involved removal of the cage mate from its home cage and weighing it before returning it to the home cage. This took approximately 1 min to complete. The restraint procedure involved removal of the cage mate from its home cage, weighing and then restraining in a hollow metal tube (approximately 27 mm diameter, 67 mm long) directly beside the home cage for 1 h. After restraint, the mouse was returned to its home cage. Following the restraint or handling procedure and return of the animals to their home cage, telemetry recordings were taken for a further 1 h (the recovery period), after which the cage was returned to the home room. These procedures were repeated for 15 days.
In vitro methods
Immediately following the procedure on the 15th day, the mice were humanely killed by cervical dislocation and the left vas deferens was isolated and cleared of connective tissue and blood vessels. The vasa deferentia were then cut to approximately 15 mm length from the prostatic end, mounted using silk thread (2–0 braided silk, Johnson and Johnson Medical Products, Ontario, Canada) on a tissue hook with platinum electrodes for electrostimulation and placed in a standard 10 mL organ bath. A modified Krebs solution adapted from Westfall and Westfall (2001) (NaCl, 118 mmol/L; KCl, 4.6 mmol/L; MgCl4, 1.2 mmol/L; MgCl2·6H2O, 1.2 mmol/L; KH2PO4, 1.2 mmol/L; NaHCO3, 24.8 mmol/L; CaCl2·2H2O, 147.02 mmol/L; glucose, 5.6 mmol/L) gassed with 95% O2: 5% CO2 was used for the tissue preparations. The responses were measured using a force-displacement transducer (Grass Force-Displacement Transducer FT03C, Grass Instruments Co, Quincy, MA, USA) connected to a bridge amplifier. Recordings were made via a PowerLab data acquisition system (ADInstruments, Australia) connected to a Windows (Microsoft Corporation, USA)-based computer and recorded in Chart 5 (ADInstruments).
Optimal tissue tension was determined prior to the resting period through electrostimulation at tensions between 250 and 400 mg. After a 30 min equilibration period, concentration response curves to exogenous NAd were determined. Serial dilutions between 10−7 and 10−3 mmol/L were prepared from a 10−2 mmol/L stock solution of NAd bitartate (Sigma Chemical Co, St Louis, MO, USA). NAd was added to the bath and washed out after 30 s, or after the maximum response to that dose had been observed. A 10 min resting period was allowed before the next dose of NAd was applied.
Following the NAd dose–response curve, the tissue was subjected to high potassium physiological salt solution (KPSS: 128 mmol/L KCl, 1 mmol/L MgSO4·6H2O, 1.3 mmol/L KH2PO4, 6.77 mmol/L NaHCO3, 3 mmol/L CaCl2 and 4.42 mmol/L glucose) to determine the maximal contractile response.
Data analysis
All statistical analyses were carried out using StatView 5.0 (SAS Institute Inc, Cary, NC, USA) with P ≤ 0.05 being considered significant.
The average body weights for days 1–3, 4–6, 7–9, 10–12 and 13–15 were analysed between days within a group to determine if there was a significant weight gain. Repeated measures ANOVA was used to determine if there was a significant difference between groups.
HR and cBT data recorded from the telemetry transmitters were compiled in Excel (Microsoft Office 2003, Microsoft Corporation). Baseline values were determined by taking the average of the recordings for the hour before the procedure was conducted. During the recovery period following the handling- and restraint-association procedures, and during the restraint period of the restraint-association procedure, the values plotted were the means ± SEM for 10 min intervals throughout the hour.
The baseline and 10 min mean values were calculated for each day of the procedures. The area under the curve (AUC) was calculated from the mean values for all days during the recovery period for the handling-associated group and during the restraint period for the restraint-associated group. These were then tested for significance using ANOVA. Data-sets from days 1, 3, 7 and 14 for each parameter were analysed for significance between days for the recovery period of the restraint-associated group using repeated measures ANOVA.
NAd responses were determined from the second contraction of the biphasic contractile response of the vas deferens. NAd responses were plotted as a percentage of the maximal KPSS response of the tissue. Concentration responses for NAd were compiled in Excel (Microsoft Office 2003, Microsoft Corporation) and concentration response curves were plotted and EC50 values calculated using GraphPad Prism 4.2 (GraphPad Software Inc, San Diego, CA, USA). The linear portion of the dose–response curve was analysed with repeated measures ANOVA.
Results
Body weight
At the start of the procedure, the mice in the handling- and restraint-associated groups had an average body weight of 23.5 g ± 0.4 g and 23.0 g ± 0.4 g, respectively and these weights were not significantly different. All mice were found to gain a significant (P < 0.0001, ANOVA, 4 d.f.) amount of weight throughout the course of the experiment, with mice in both groups gaining a mean total of 1.6 g.
Heart rate and core body temperature
A significant increase in HR (P < 0.0001, repeated measures ANOVA, 6 d.f., Figure 1a) and cBT (P < 0.0001, repeated measures ANOVA, 6 d.f., Figure 1b) was observed during the hour of recovery in the handling-associated mice following the return of the cage mate to the cage after weighing. Similarly, a significant increase in HR (P < 0.0001 repeated measures ANOVA, 6 d.f., Figure 1a) and cBT (P < 0.0001 repeated measures ANOVA, 6 d.f., Figure 1b) was observed during the hour of restraint of the cage mate in the restraint-associated mice.
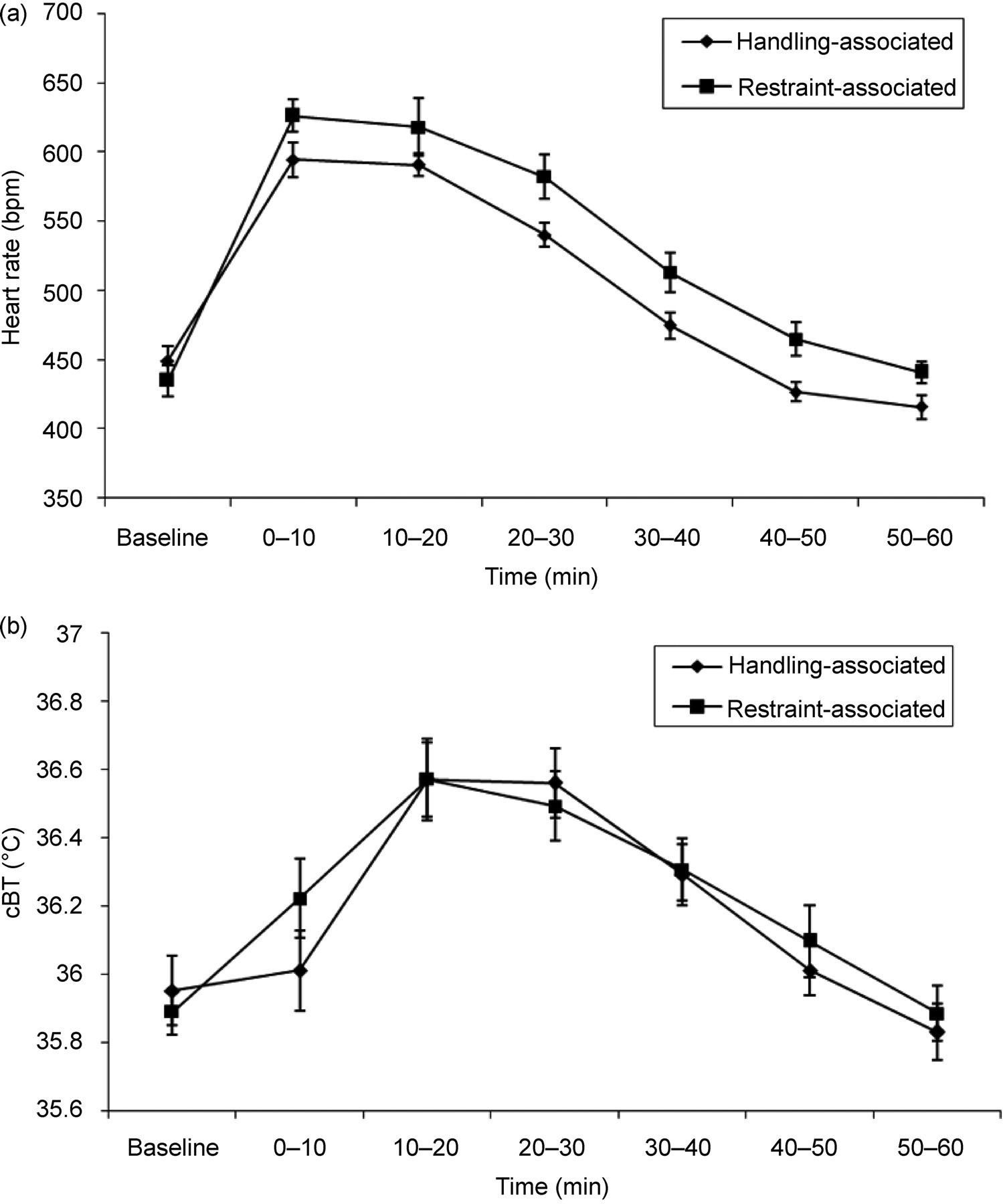
The HR during both the handling-associated recovery period and the restraint-associated restraint period (Figure 1a) peaked during the 0–10 min time period, returning to baseline by 50–60 min. The cBT (Figure 1b) peaked during the 10–20 min time period for the handling-associated mice, and during the 20–30 min period for the restraint-associated mice. The cBT returned to baseline within 50–60 min for both groups.
Comparison of the AUC for both HR (Figure 1a) and cBT (Figure 1b) showed that it was significantly higher during the restraint-associated restraint period than during the handling-associated recovery period (HR: P = 0.0008 ANOVA, 1 d.f.; cBT: P < 0.0001 ANOVA, 1 d.f.).
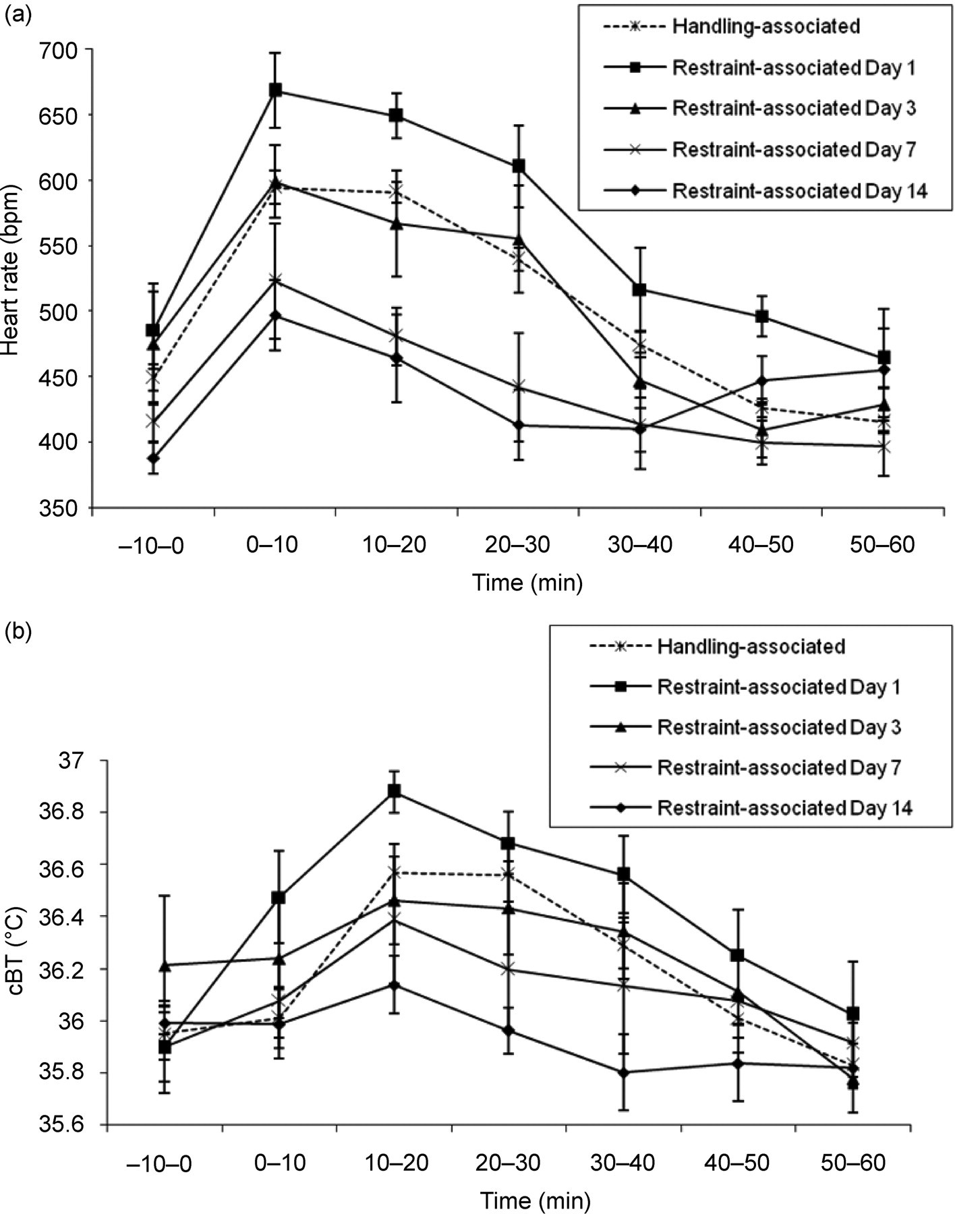
Comparison between days found no significant difference in either HR or cBT for both the recovery period of the handling-associated mice and the restraint period of the restraint-associated mice (data not shown). However, during the recovery period of the restraint-associated mice there was a significant difference in both the HR (P = 0.009 repeated measures ANOVA, 6 d.f., Figure 2a) and cBT (P = 0.050 repeated measures ANOVA, 6 d.f., Figure 2b), with less marked increases in HR and cBT as the experiment progressed.
Noradrenaline dose–response curves
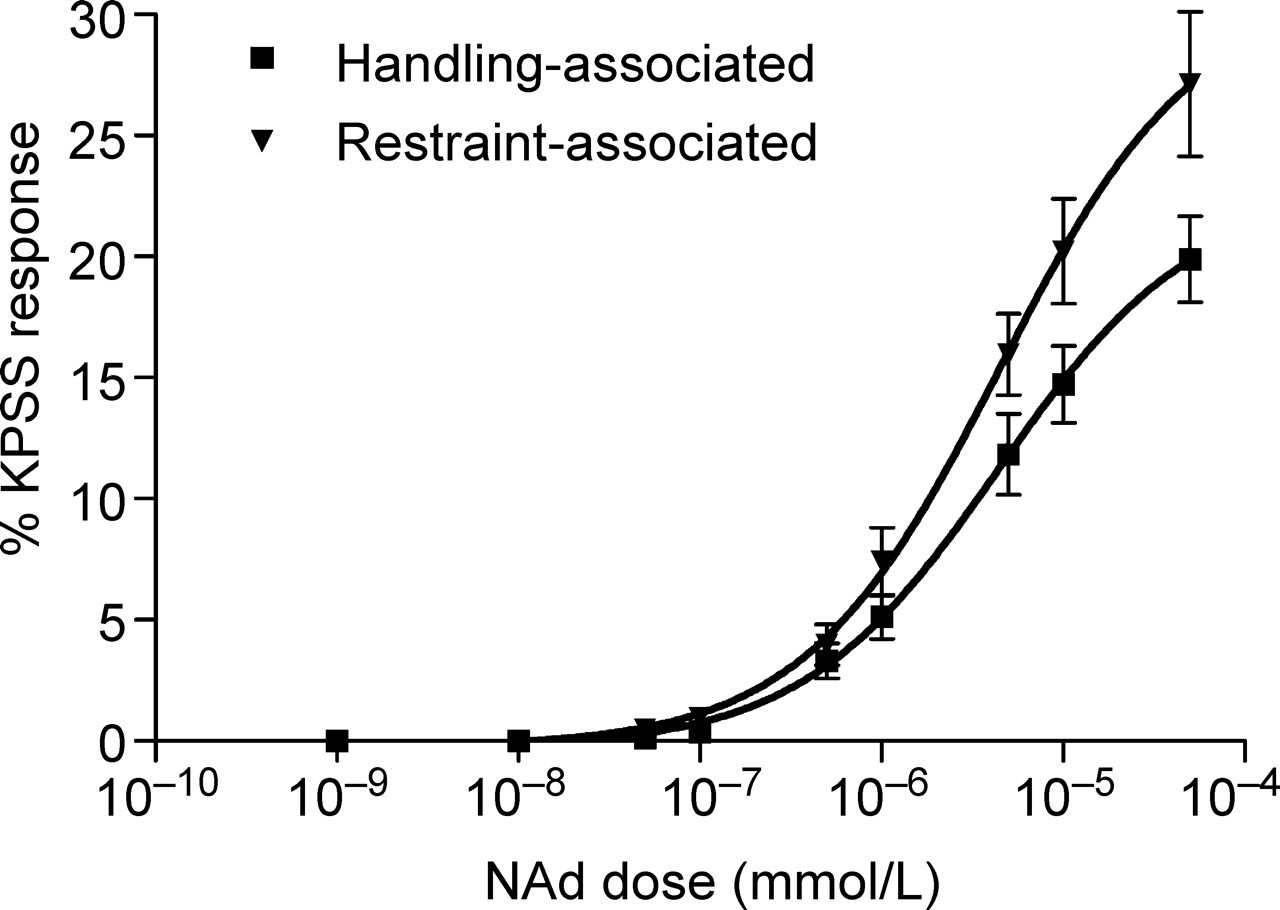
Analysis of the KPSS responses showed that there was no significant difference between the maximal responses in vas deferens from the handling- and restraint-associated mice (2095 ± 310 mg and 1776 ± 436 mg, respectively). Therefore, the responses to NAd are shown as a percentage of the KPSS response.
There was a dose–response relationship for exogenously applied NAd in tissues from both the handling- and restraint-associated groups (Figure 3). A significant increase in the maximum response in tissues obtained from restraint-associated mice when compared with handling-associated mice was found (P < 0.0001, repeated measures ANOVA, 6 d.f.); however, there was no difference in the EC50's (Figure 3; 2.81 × 10−6 mmol/L and 3.52 × 10−6 mmol/L for the handling- and restraint-associated groups, respectively).
Discussion
HR is known to increase in response to stress (Chrousos & Gold 1992, Bernatova et al. 2002, Spani et al. 2003) and increases in HR, mediated via activation of the SNS, are generally considered to be an indication of stress. An acute rise in cBT following a stressful event is also well established and has been previously reported (Briese & De Quijada 1970, Zethof et al. 1995, Van Der Heyden et al. 1997), although the mechanism underlying this phenomenon is not yet fully understood. In this study, there was a significant increase in HR and cBT both in the handling-associated mice following weighing of their cage mates and in the restraint-associated mice following the restraint of their cage mates. This suggests that even the removal of a cage mate from the cage is stressful to the mice remaining in the cage. In these experiments, the handled animals were only removed from the home cage for a short time. It would be of interest to delay the return of these animals to the home cage for an hour and to observe the responses in handling-associated mice during this period.
However, comparison of the AUCs showed that the HR and cBT in the restraint-associated group during the cage mate's restraint was significantly greater than that in the handling-associated group during the recovery period. Thus, stressing of a cage mate outside the cage, but within close proximity, significantly increases HR and cBT in the untouched mice remaining in the cage, and this is probably indicative of stress in those animals.
Adaptation to chronic stress as a coping mechanism is referred to as habituation, i.e. a phenomenon in which repeated stress leads to a reduction in the apparent impact of that stress. This has been observed by others in rats subjected to chronic intermittent stress (Pitman et al. 1988, Konarska et al. 1989) and has been shown to involve an interplay between the central and autonomic nervous systems and endocrine system (Blaszkowski et al. 1970). There was a clear habituation effect in the restraint-associated mice, so that the increases in HR and cBT during the recovery period became less apparent from day 1 to day 14 of the experiments. It is possible that either the restrained mice became habituated to the restraint procedure and therefore communicated less stress to their cage mates or the cage mates became habituated to the restrained mice being stressed, or a combination of both of these effects occurred. However, a habituation effect was not seen during either the recovery period of the handling-associated mice or during the actual restraint period of the restraint-associated mice.
It might be expected that the mice being exposed to a less stressful procedure, such as the handling-associated mice, would be more likely to display signs of habituation. However, a similar effect was observed by Pitman et al. (1988), where although partial habituation occurred in rats exposed to a more intense stressor (restraint for 2 h), no habituation occurred in the rats exposed to a less-intense stress (handling).
Stress is commonly known to affect weight gain in animals (Blaszkowski et al. 1970, Pitman et al. 1988, Shimizu et al. 1994, Rybkin et al. 1997), and weight gain is considered to be a useful indicator of the level of stress to which an animal is exposed. Weight loss as a result of chronic restraint stress has been observed in rats (Harris et al. 1998, Valles et al. 2000), and has been suggested to be caused by up-regulation of neuropeptide Y, which has been found to result in decreased food intake (Rybkin et al. 1997, Thorsell et al. 1999). In the present study, there was no significant difference in daily weight gain and total weight gain between mice in handling- and restraint-associated groups. This suggests that, although restraining a cage mate caused significant changes in the HR and cBT of the mice remaining in the cage, the level of stress was not sufficient to affect weight gain in these mice.
It has previously been shown that repeated stress causes the vas deferens to become hypersensitive to NAd (Singh et al. 2001, Einstein et al. 2004). In this study, it was found that the vas deferens from the restraint-associated mice had a significantly higher responsiveness to NAd than the vas deferens isolated from the handling-associated mice. These results suggest that the mice whose cage mates were restrained were significantly more stressed than the mice whose cage mates were subjected to the handling procedure. Although it has been shown that handling of rats was stressful for the animals (Uphouse et al. 1982, File & Fluck 1994), the effect of handling of mice on those remaining in the home cage has not previously been reported.
Thus, this study has shown that restraint of a cage mate was significantly more stressful than simple handling and weighing of a cage mate. Although the stress was not sufficient to significantly affect the weight gain of mice remaining in the cage, it was sufficient to have significant and immediate effects on the HR and cBT. Some habituation to the stress was noted but, despite this, after 14 days of repeated stress there was an increased responsiveness of the vas deferens to exogenous NAd. The findings indicate that performing stressful procedures on a mouse can induce a stress response in a cage mate in close proximity to, but not necessarily visual contact with, the mouse being stressed. In the interests of animal welfare and to prevent confounding of results, stressful procedures should be performed in total isolation from cage mates remaining in the home cage.
