Abstract
An experimental model of membranous nephropathy (MN) has not been established fully in mice. We characterized the time course of MN in a murine MN model induced by cationic bovine serum albumin (cBSA). Preimmunized mice received cBSA intravenously for six weeks to induce MN and were then sacrificed at different times. Metabolic profiles, renal histopathology, lymphocyte subsets, serum anti-cBSA immunoglobulins (Igs), antibody subclasses and circulating immune complexes (CIC) were evaluated to study the severity and mechanisms of disease initiation and progression. Clinical symptoms of overt proteinuria, hypoalbuminaemia and hypercholesterolaemia were observed from week 4, and typical histological findings of diffuse thickening of the glomerular basement membrane and subepithelial deposition were identified after week 6. Granular fluorescent staining for IgG and complement C3 were observed as early as week 4. Total splenocyte number increased, but the percentages of CD4+ and CD8+ cells did not change as the disease progressed. The predominant isotype of anti-cBSA Igs was IgG1, suggesting a T-helper 2 cell-prone immune response in the development of MN. The strong positive immunofluorescent staining of the immune complex concomitant with higher concentrations of Igs in serum but no significant change in CIC levels before week 4 suggest the involvement of
Membranous nephropathy (MN) is one of the most common causes of nephrotic syndrome in adults. About 30–40% of patients develop progressive renal impairment that results in end-stage renal failure after 10–15 years (Glassock 2003, Remuzzi et al. 2006). MN is thought to involve the
This study was designed to characterize the clinical and pathological time course of cBSA-induced MN. The advantages of this murine model are its low cost and potential applications of genetic and monoclonal antibody techniques to study the pathogenesis of MN.
Materials and methods
Animals
The study was performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health and was approved by the Animal Care and Ethics Committee of the National Defense Medical Center (Taipei, Taiwan). BALB/c mice (4–6 weeks old, about 20 g body weight) were purchased from the National Laboratory Animal Center (Taipei, Taiwan). Mice were kept in the Laboratory Animal Center of the National Defense Medical Center under specific pathogen-free conditions and strictly controlled room temperature conditions of 20 ± 2°C, relative humidity of 50 ± 10% and a 12:12 h light–dark cycle. The mice were fed a commercial pelleted diet (SDS, Essex, UK) comprising 14.7% protein, 2.6% fat, 5.3% fibre and 5.9% ash; tap water was available from plastic bottles
Experimental design
The mice were divided randomly into two groups: an experimental group and a control group. Animals in the experimental group were immunized with 0.2 mg of cBSA emulsified in an equal volume of complete Freund's adjuvant (CFA), and those in the control group were immunized with CFA alone. Two weeks later, these mice received cBSA (13 mg/kg) intravenously three times per week, every other day, for six weeks to induce MN, and the control group received saline alone at these times. Six mice were sacrificed at weeks 0, 2, 4, 6 and 8 after immunization. We collected blood, urine and kidney samples for analysis. cBSA was prepared as described previously (Chen et al. 2004).
Laboratory tests for blood and urine biochemistry
Blood and urine samples were collected, microcentrifuged and stored at –70°C until assayed. Urinary protein was quantified with a bicinchuninic acid (BCA) protein assay kit (Pierce, Rockford, IL, USA) and proteinuria was calculated as the ratio of urinary protein (mg/mL) to urinary creatinine (mg/dL) (Up/Ucr). Blood urea nitrogen (Sigma 640-5) and serum concentrations of creatinine (Sigma 555-1), albumin (Sigma 631-2P) and total cholesterol (Sigma 352-20) were measured with kits from Sigma-Aldrich Inc (St Louis, MO, USA). All assays were performed in duplicate according to the manufacturer's instructions.
Histological studies of renal tissues
Formalin-fixed and paraffin-embedded sections of mouse kidney tissue were cut and stained with haematoxylin and eosin. Frozen tissues were sectioned, dried in air, fixed in acetone for 10 min at room temperature and washed with phosphate-buffered saline (PBS) before incubation with fluorescein isothiocyanate (FITC)-conjugated goat anti-mouse IgG, IgA, IgM or C3 (Cappel, Durham, NC, USA). Fluorescence was observed and evaluated semi-quantitatively as described previously (Chen et al. 1998). Glomerular polyanions were quantified with a procedure using colloidal iron staining for light microscopy. For electron microscopic examination, ultrathin sections were double-stained with uranyl acetate and lead citrate, and then they were examined using a transmission electron microscope (Hitachi 7000, Hitachi Co, Japan).
Lymphocyte subsets determination
Spleen cells (splenocytes) were collected aseptically from mice at different times and analysed for lymphocyte subpopulations. Isolated splenocytes (106 cells in 0.1 mL of PBS) were incubated on ice and stained with marker-specific antibody (0.5 µg antibody per 106 cells): FITC-conjugated anti-mouse CD19 (B-cell marker) clone 1D3, phycoerythrin-conjugated anti-mouse CD8α (cytotoxic T-cell marker) clone 53-6.7 or FITC-conjugated anti-mouse CD4 (helper T-cell marker) clone RM4-5 (Tipping & Kitching 2005). Flow cytometry was performed with a FACSCalibur cell sorter (BD Biosciences, San Diego, CA, USA) and analysed with CellQuest software (BD Biosciences). All antibodies used for flow cytometry were purchased from BD PharMingen, San Diego, CA, USA.
Production of antibody and circulating immune complexes
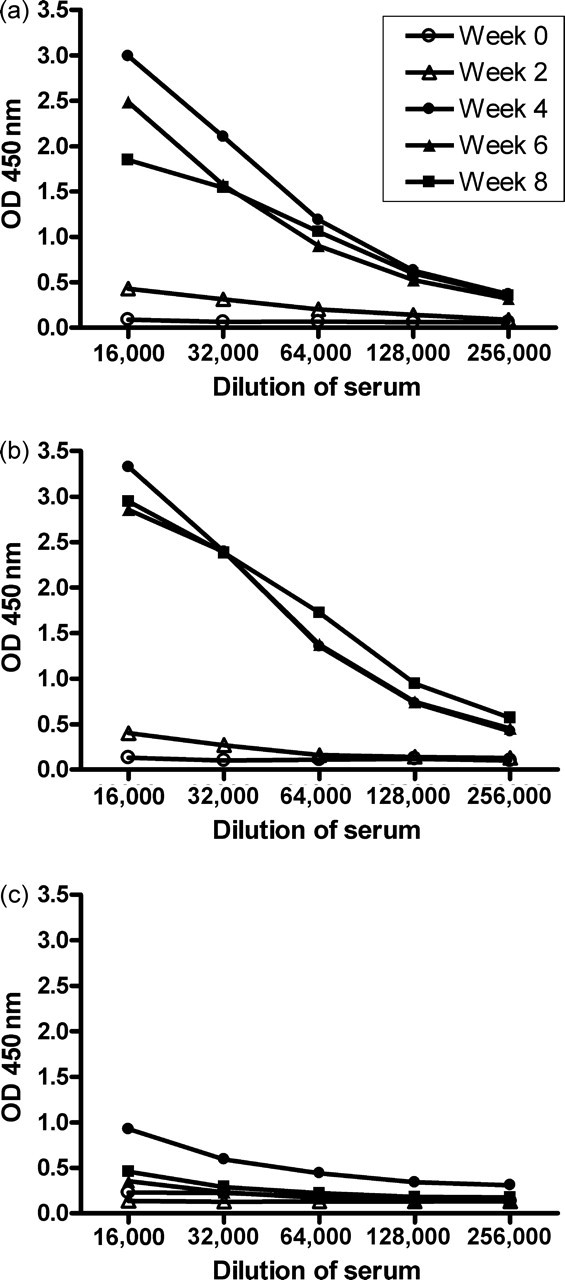
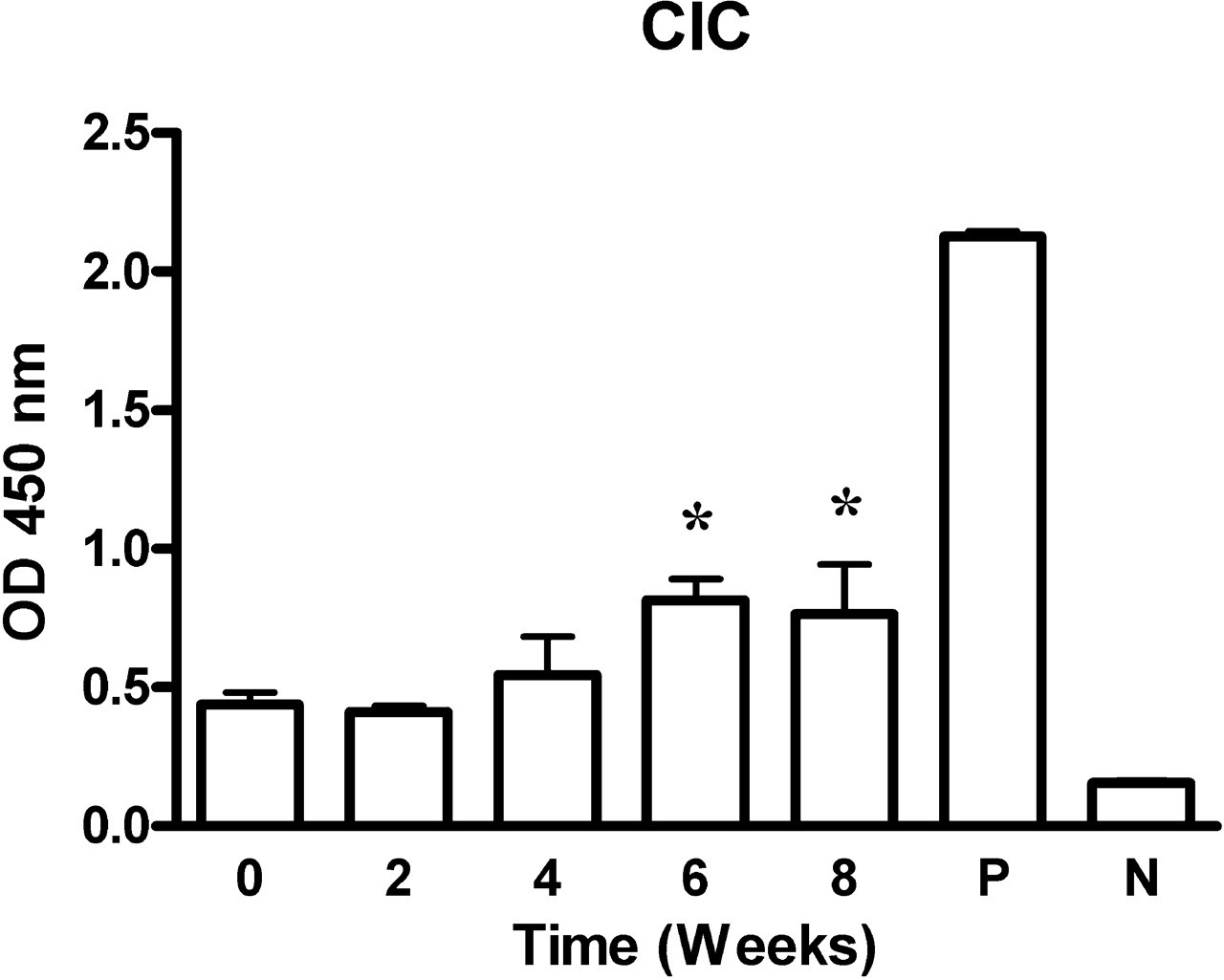
Serum concentrations of the anti-cBSA immunoglobulins (Igs), IgG1 and IgG2a were measured by enzyme-linked immunosorbent assay as described previously (Chen et al. 2004). Horseradish peroxidase-conjugated goat anti-mouse Igs (DAKO, Carpinteria, CA, USA), IgG1 and IgG2a (Bethyl Laboratories Inc, Montgomery, TX, USA) were used. The serum was serially diluted (1:16,000–1:256,000) and its optical density was checked at 450 nm. To detect circulating immune complexes (CIC), plates coated with C1q to detect CIC (Alpha Diagnostic International Inc, San Antonio, TX, USA) were used with serum samples diluted 10,000 times in sample buffer. The procedures were performed according to the manufacturer's instructions. Data were read and were expressed as optical density (OD) at 450 nm.
Statistical analyses
All data were expressed as means ± standard deviation (SD). The data were analysed by one-way analysis of variance for multiple comparisons. Two-group differences were analysed by
Results
General characteristics
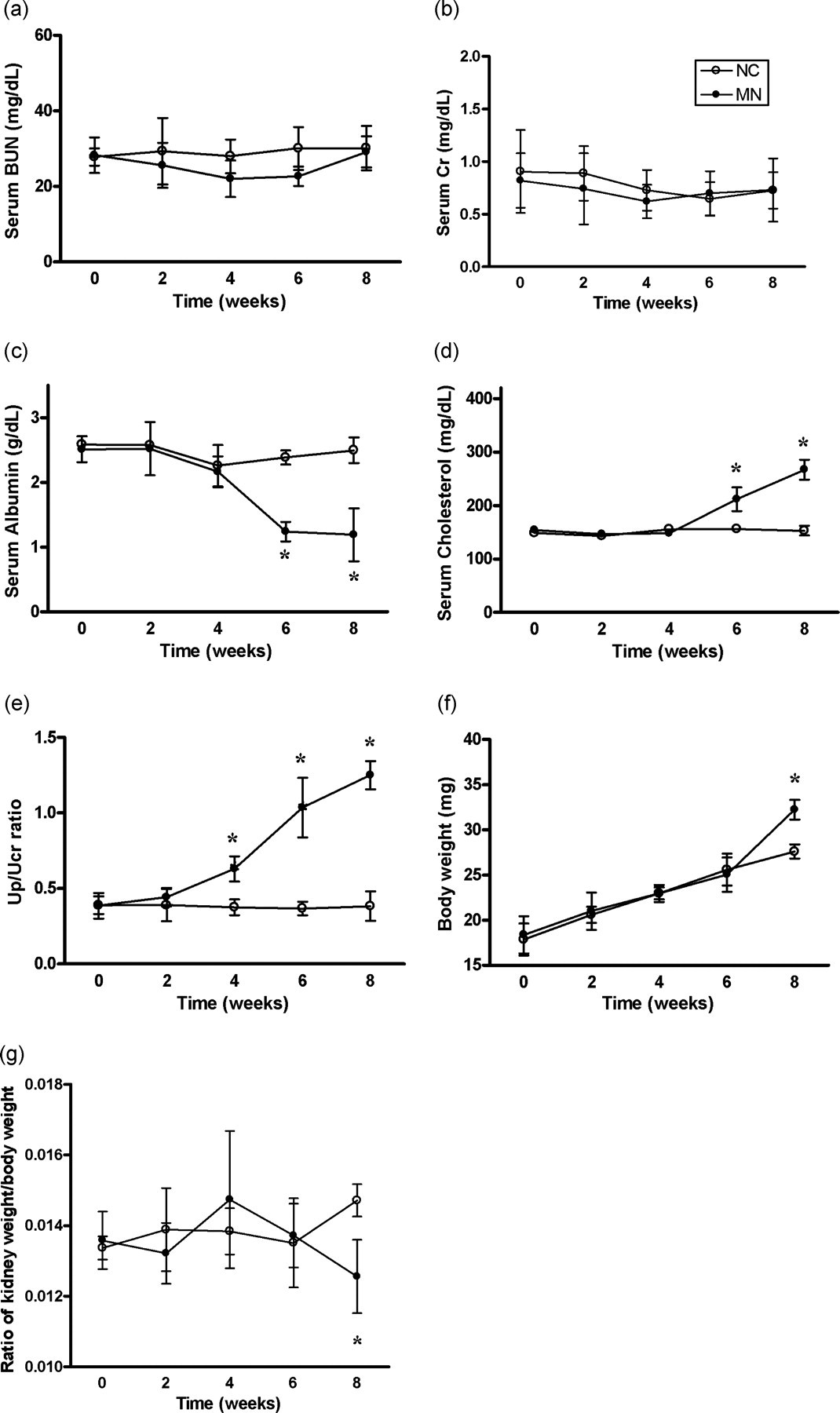
In a series of preliminary experiments, we induced MN with cBSA in both male and female mice. All BALB/c mice developed the characteristic clinical symptoms of proteinuria, hypoalbuminaemia and hypercholesterolaemia. These did not differ between sexes and only the results for the females are given in this report. Figures 1a and b show that blood urea nitrogen and serum creatinine concentrations did not change during the experiment. Overt proteinuria, described in terms of the Up/Ucr ratio, appeared in week 4 and reached a plateau at week 8 (Figure 1e). Urinalysis revealed no haematuria or leukocyturia during the study (data not shown). Serum albumin concentration declined markedly after week 6 in the MN group and reached its nadir at week 8. However, serum cholesterol concentration showed a compensatory increase at week 6 and was maximal at week 8 (Figures 1c and d). The mean body weight of the MN group and control group increased in parallel before week 6. Thereafter, some MN mice developed ascites and their body weights increased rapidly (Figure 1e) resulting in a decrease in the ratio of kidney weight to total body weight at week 8 (Figure 1f).
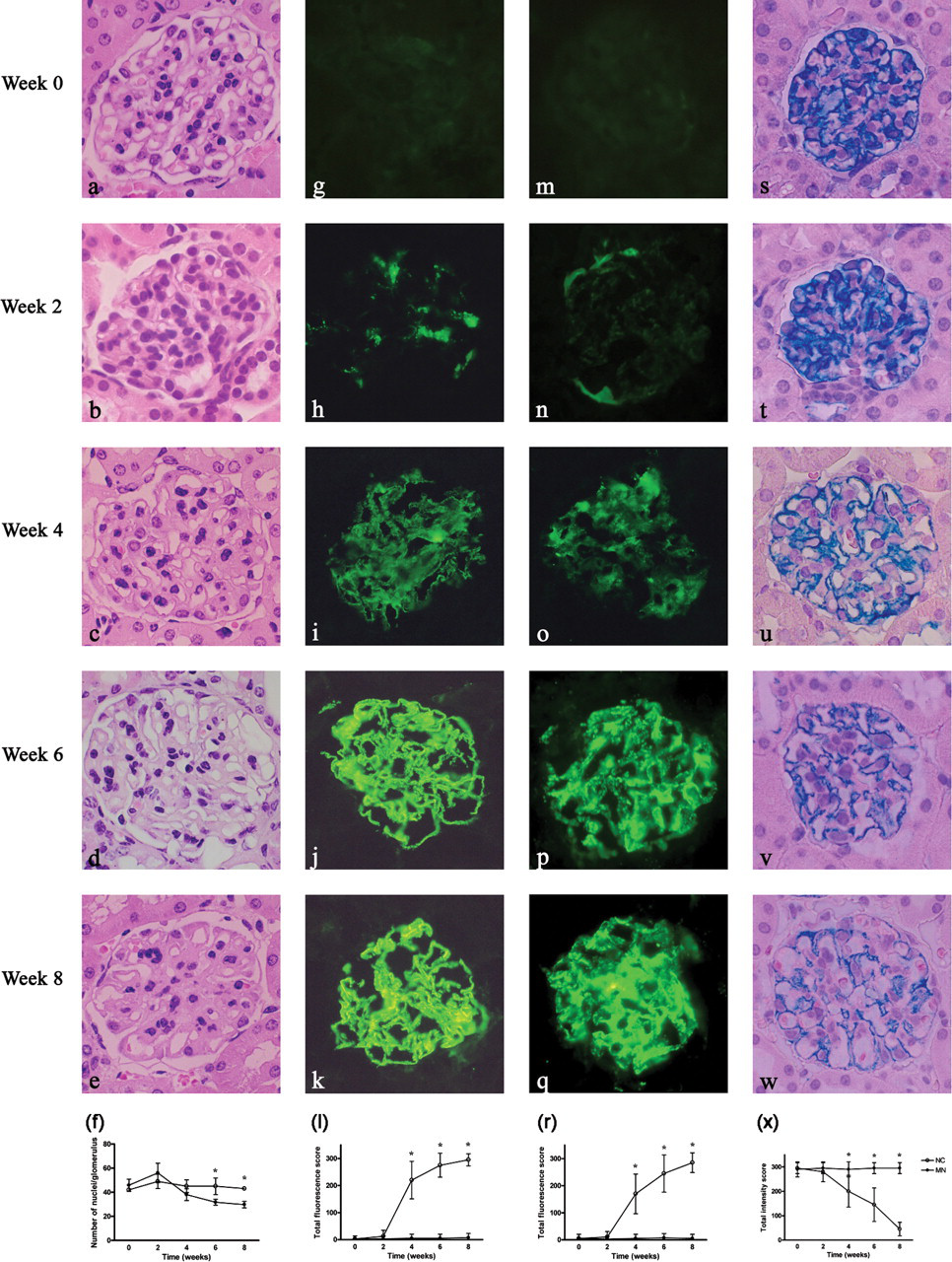
Morphometric and histopathological studies
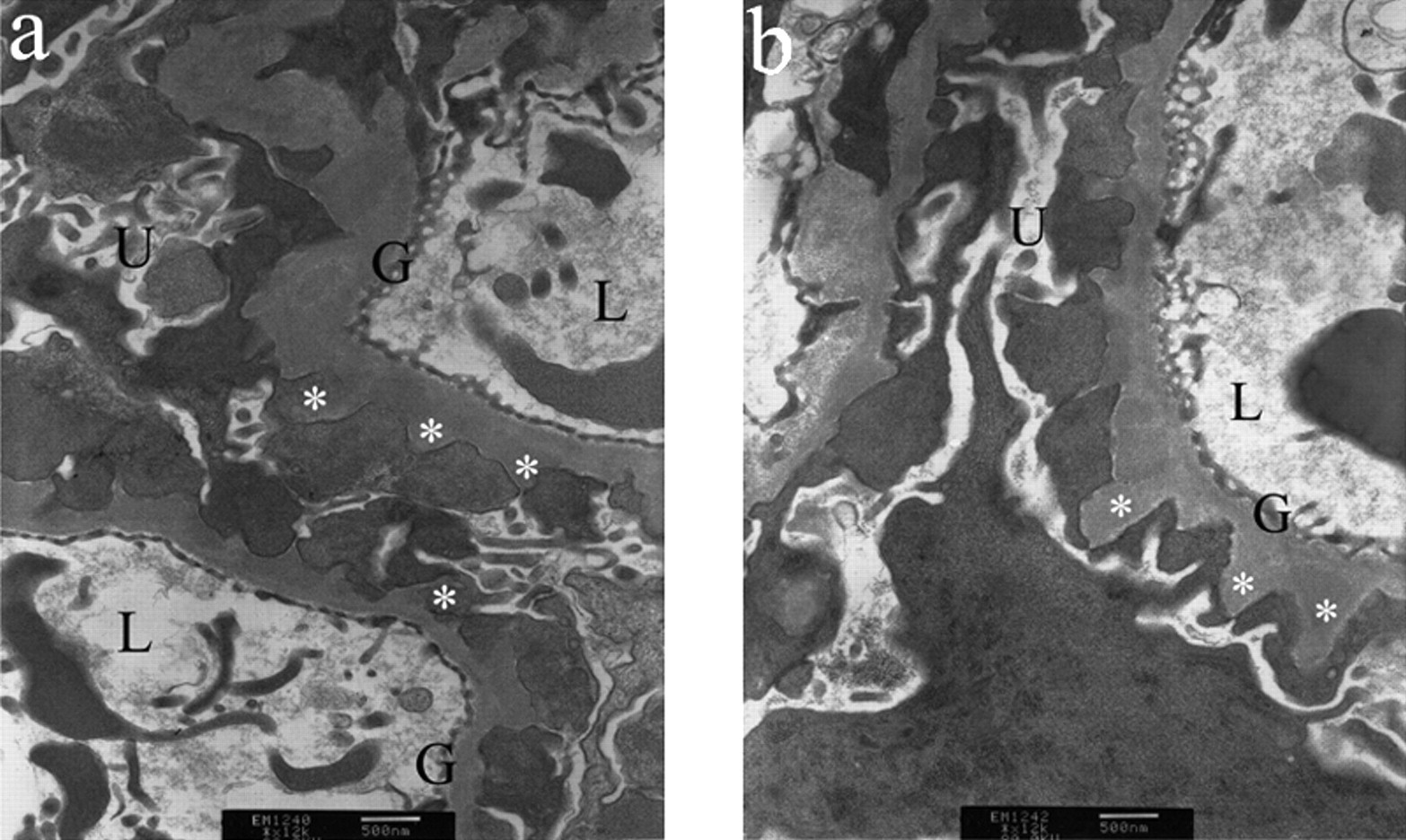
Pathological findings were observed and revealed similar pictures in both male and female BALB/c mice. Light microscopy demonstrated serial changes in the experimental kidneys (Figures 2a–e). The mean number of cells decreased and no significant mesangial proliferation was noted in the MN group (Figure 2f), coincident with a diffuse thickening of the glomerular basement membrane (GBM). Positive immunofluorescent staining for IgG was noted after week 4, and this was followed by progressively stronger granular fluorescence intensity along the glomerular capillary wall (GCW) with a discrete beaded appearance (Figures 2g–k). Immunofluorescence staining for C3 also revealed granular fluorescence intensity along the GCW, with a similar time course and pattern to that of IgG (Figures 2m–q). IgA and IgM were both negative on immunofluorescent staining (data not shown). The intensity of colloid iron staining decreased progressively during the course of MN induction, indicating the loss of glomerular anions and the impairment of charge selectivity (Figures 2s–w). Ultrastructural analysis identified severe irregular thickening of the basement membrane and subepithelial deposits in mice with cBSA-induced MN (Figure 3). GBM spikes were observed extending between the deposits, and there was a loss of podocyte foot processes. Normal control mice showed no epithelial cell abnormalities (data not shown).
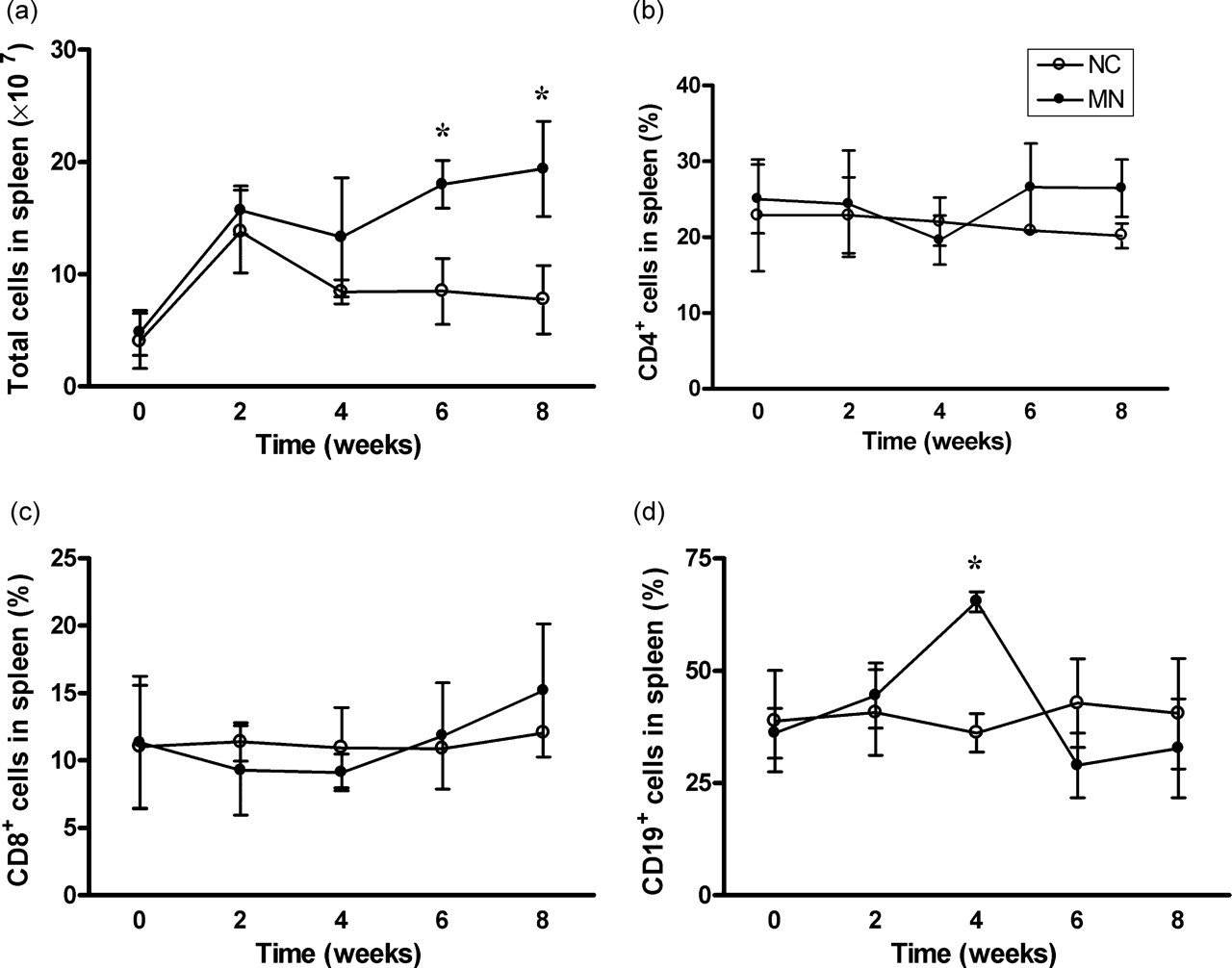
Changes in major lymphocyte subsets in the spleens of MN mice
Immune cells are involved in the development and progression of MN (Ruggenenti et al. 2003, Tipping & Kitching 2005). We used flow cytometry to analyse the peripheral lymphocytes in spleens and to determine whether the expression of lymphocyte populations correlated with MN induction. Total splenocyte number increased as the disease progressed in MN mice (Figure 4a). The percentages of CD4+ and CD8+ splenocytes did not change significantly at any time in the experimental animals (Figures 4b and c). A significant increase in the B-cell percentage was noted at week 4, but this returned to the normal range (Figure 4d). These results suggest that the initial immune response of MN involves primarily a humoral-mediated mechanism (Tipping & Kitching 2005).
Antibody production and CIC
We measured the serum concentrations of anti-cBSA Igs and CIC in MN mice to determine whether cBSA-induced MN in mice resembles the
Discussion
Despite the literature describing MN in rats, the details of the time course of MN in a murine MN model have not been established (Couser & Abrass 1988, Salant et al. 1989, Ronco & Debiec 2005). In this study, we extended our previous work (Chen et al. 2004) and report on the time course of appearance of glomerular deposits, changes in nephritic parameters and levels of anti-cBSA Igs. Extending the MN model to the mouse has the advantages of lower cost, easy manipulation and the potential benefits of using gene-knockout and transgenic mice to investigate the mechanisms of disease initiation and progression.
Mice with cBSA-induced MN had typical features of MN, including diffuse GBM thickening, granular immunofluorescent staining, subepithelial deposits and a lack of inflammatory cell infiltration and mesangial cell proliferation (Cattran 2001). Reflecting the equal incidence of MN in men and women, the MN induction rate did not differ between male and female mice. The glomeruli of MN mice appeared histologically normal under light microscopy despite strong positive immunofluorescent staining for IgG and C3 early in the course of the disease. We note that proteinuria was induced weeks after the deposition of the immune complexes. It is proposed that many factors other than Igs and complements may participate in glomerular injury (Cunningham & Quigg 2005, Cybulsky et al. 2005, Nangaku et al. 2005). Deposition of immune complexes in the glomerular epithelial cells can trigger a cascade of events that contribute to the progression of the disease and result in glomerular injury and proteinuria (Ravnskov 1998, Ronco & Debiec 2005). In contrast, the impairment of charge and size selectivity contributes to proteinuria in such models. The coincident timing of the development of proteinuria, hypoalbuminaemia and hypercholesterolaemia indicates a relationship between these parameters. The most frequently used model of human MN, Heymann nephritis in rat, is rarely applied to mice. Our murine MN model produced morphological and biochemical outcomes that are similar to those in human MN. The murine MN model may be a useful means of studying the entire course of MN, from initiation to progression, which is difficult to study in humans.
During the induction of MN, the percentages of CD4+ and CD8+ T-cell phenotypes did not change significantly, although the total counts of immune cells increased progressively throughout the entire course of MN. In contrast, prominent B-cell activation and concomitant abundant antibody production were noted at week 4, but had decreased at week 6 and week 8 despite the persistent injection of cBSA antigen. The development of tolerance may be considered as an explanation. The functionally distinct Th1 and Th2 subsets regulate Ig switching to the IgG2a and IgG1 subtypes, respectively (Snapper & Paul 1987). In our study, IgG1 was the predominant antibody produced, and its production was noted as early as week 2. This confirms that an initial Th2-type response, the predominantly humoral antibody-mediated immune response, plays an important role in the pathogenesis of MN.
Immunofluorescence analysis revealed positive staining for both IgG and C3. Although a strong positive immunofluorescent staining of immune-complex deposition was concomitant with higher serum Ig concentrations, serum CIC concentration did not increase significantly before week 4. These observations suggest the
The precise nature of the idiopathic MN-initiating antigen is unknown (Kerjaschki 2004). Megalin (gp330) has been identified as a pathogenic antigen in Heymann nephritis, but it has not been found in human or mice glomeruli (Kerjaschki & Farquhar 1982, 1983). Although the cBSA used in this study is an exogenous rather than an endogenous antigen, the increased serum concentrations of specific Igs measured by highly specific ELISA can be used to assess its immunopathological potential during MN.
We did not consider it useful, practical or ethical to extend the study to eight weeks to produce full-blown MN. Repeated intravenous injections through the tail vein are difficult to maintain over an extended time, and most MN mice presented with exacerbated proteinuria, which would have caused severe ascites and rapid death had immunization continued after week 8.
In conclusion, we established a stable and reproducible murine model of MN induced with cBSA and characterized its clinical and pathological features over time. Because of its similarity to human MN, this murine model will provide a valuable means of investigating the pathogenesis of MN and will help in the development of preventive and therapeutic strategies for MN. The model is economical and can take advantage of gene-knockout and transgenic mouse technologies.
