Abstract
Summary
Rat serum or plasma creatine kinase (CK) activity is widely used to evaluate myopathic processes, to test the myotoxicity of different drugs, or to analyse the benefits of emerging gene therapies in some neuromuscular disorders. However, great variability is found in this determination. The aim of this study has been to control some factors of variation in order to reduce variability and increase the reproducibility of analytical data. 8–10-week-old Wistar-Han rats were used. The study consisted of four sequential phases. Phase I aimed to analyse the effect of ether and isoflurane as anaesthetic drugs. The objective of Phase II was to evaluate bleeding rats via retro-orbital sinus vs. tail vein. Phases III and IV were designed as two separate, repeated measure experiments on two factors: habituation to laboratory handling procedures in Phase III and gender in Phase IV. The repeated factor was the storage temperature of blood sample prior to centrifugation. Ether did not significantly increased the CK value. Using isoflurane, getting rats accustomed to laboratory handling procedures and whole blood refrigeration prior to centrifugation and serum separation resulted in statistically significant reduction in CK value and variability. Male rats showed significantly higher values than female rats. In the light of our findings, CK value and variability in rats may be minimized by choosing tail vein as site of bleeding, getting rats accustomed to laboratory handling procedures and maintaining whole blood refrigerated until centrifugation and serum separation.
Creatine kinase (CK; EC 2.7.3.2) is an intracellular enzyme present in a variety of striated and smooth muscles and in the brain; it is an important enzyme regulator of high-energy phosphate production and utilization in contractile tissues. CK is physiologically present in the plasma, indicating a permanent release from muscle cells, and it is one of the most widely used enzymes for the quantitative evaluation of myocardial infarction (Klein et al. 1973, Maroko & Vatner 1977, Visser et al. 1981), myopathic processes (Cardinet 1989), muscle damage following intramuscular injections (Lewis & Rhodes 1978) and in various other diseases. Although not all myopathies produce a rise in CK activity, it is increased as a result of muscle fibre destruction after mechanical trauma, toxic injury or alteration of enzymatic or structural proteins. CK elevation varies within disorders, with increases that may range from 2 to 100-fold the reference value.
Blood can be sampled from animals using different techniques with differing impacts on animal discomfort due to differences in handling, restraining, anaesthesia, invasiveness and the volume taken. The method of blood sampling can also affect the outcome of blood analysis. Orbital puncture is frequently used to obtain blood samples from rats. However, this technique is controversial because it possibly causes pain and distress to the animals (van Herck et al. 1992, 1999). The BVA/FRAME/RSPCA/UFAW joint working group has stated that orbital puncture is acceptable only as a terminal procedure while the animal is under anaesthesia (Morton et al. 1993). For non-terminal blood sampling in rats, the working group advises collecting a maximum of 0.5 mL of blood by puncturing the tail vein while applying a short-acting anaesthetic to ease the rats. In spite of this, retro-orbital puncture is still widely used. This may be due to the lack of either scientific proof of negative consequences when performed by trained staff or the lack of equally good alternatives.
CK activity has been reported to vary within animal species (Matsuzawa et al. 1993) and gender (Kaspar & Norris 1977, Loeb & Quimby 1999), and with age (Shibata & Kobayashi 1978, Cardinet et al. 1989, Matsuzawa et al. 1993), stress (Lefebvre et al. 1992), site of bleeding (Matsuzawa et al. 1993), puncture technique (Fayolle et al. 1992), handling procedures (Bacou & Bressot 1976, Sneddon et al. 1989, Lefebvre et al. 1992, Lefebvre et al. 1996, Yerroum et al. 1999), serum or plasma measurement (Shibata & Kobayashi 1978, Matsuzawa et al. 1993, Aktas et al. 1994), storage of the specimen and the analytical method used (Lev et al. 1994, Clark et al. 2003). Thus, for CK activity, values ranging from 100 U/L to 900 U/L have been observed in control rats (Hsu et al. 1995, Nosaka 1996, Reijneveld et al. 1996, Matsuda et al. 1998, Yerroum et al. 1999). However, in the majority of toxicological studies, preanalytical specifications and recommendations are described either poorly or not at all, making it difficult to standardize them on a repetitive basis. This matter may be of particular interest if it is recognized that the effect of mild myotoxins could possibly be masked as a result of omitting such factors of variation. Nowadays, with the performance of myotoxicity studies on new drugs and the animal testing of emerging cellular and gene therapies for the treatment of some muscular dystrophies, the analysis of CK variation could be one of the principal markers for evaluating potential new drug candidates or the course of a disease and its potential improvement with therapy.
The aim of this study has been to analyse a number of factors affecting CK activity in Wistar-Han rats, including anaesthesia, site of bleeding, handling, getting the rats accustomed to laboratory procedure techniques, whole blood sample storage prior to centrifugation and gender, in order to minimize variability and improve the reliability of the measurement of CK activity.
Materials and Methods
Design and phases of trial
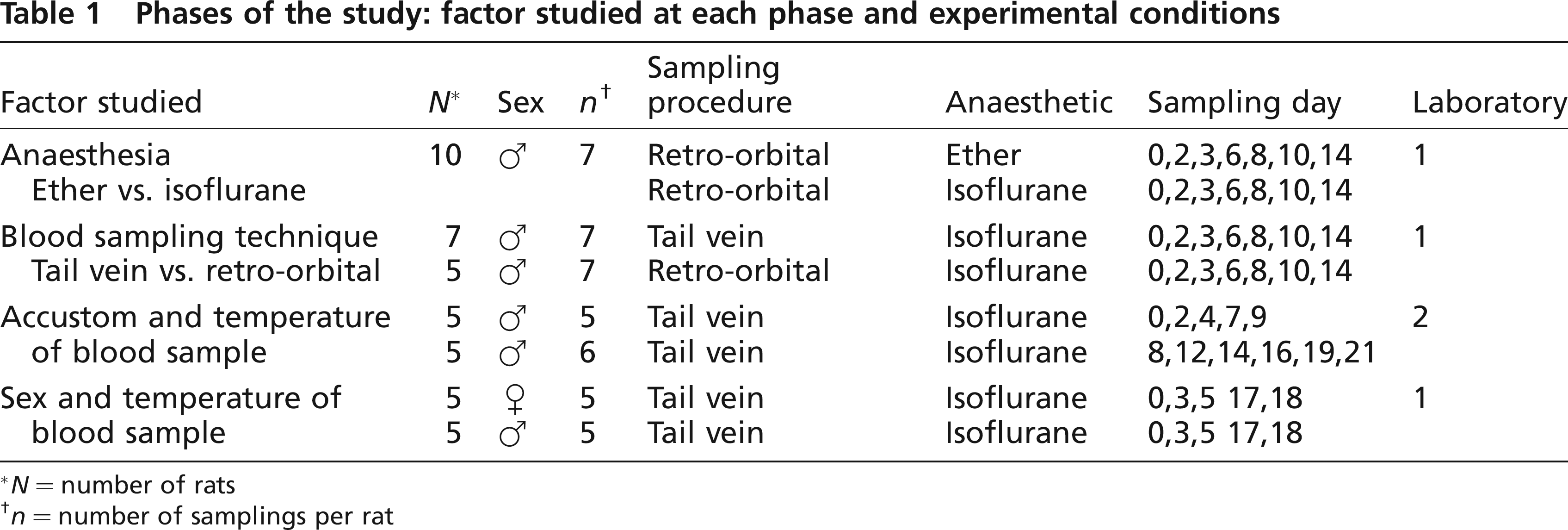
The study consisted of four sequential phases in which the results of each stage conditioned the design of the following phase. Phase I aimed to analyse the effects on CK activity of the use of ether and isoflurane as anaesthetic drugs. The objective of Phase II was to compare two different blood sampling techniques: retrobulbar plexus vs. tail vein. Phases III and IV were designed as two separate, repeated measure experiments on two factors: habituation to laboratory handling procedures in Phase III and gender in Phase IV. The repeated factor was the storage temperature of blood sample prior to centrifugation. Phases I, II and IV were performed in one laboratory (Laboratory 1), whereas Phase III was carried out in another one (Laboratory 2). The study was undertaken in accordance with the ‘Real Decreto 1201/2005’ about animal welfare and its associated Codes of Practice.
The experimental design is summarized in Table 1.
Phases of the study: factor studied at each phase and experimental conditions
N = number of rats
n = number of samplings per rat
Animals
Eight-to-ten-week-old male and female HsdRccHan™:WIST rats (Harlan, Barcelona, Spain) were housed in individual Makrolon™ cages, allowing a one-week acclimatization period. All the animals were kept at a constant temperature (22°C) in a 12:12 h light–dark cycle, and were provided with food and water ad libitum. Carbon dioxide (CO2) was used for euthanasia.
Handling and anaesthesia
To induce sedation with ether, this was first applied to an absorbent material and then placed in the anaesthetic chamber, being removed as soon as it lost consciousness. For the maintenance of sedation, the absorbent material soaked in ether was placed in an empty syringe cartridge and moved closer or further away from the animal's face to adjust the concentration.
When using isoflurane (Forene®, Deutsche Abbott, Delkenheim, Germany), rats were lifted by the base of the tail and rapidly introduced into the anaesthetic chamber and sedated with 5% isoflurane for 1 min. The anaesthesia was maintained using a polycarbonate mask with 2.6% isoflurane. As complementary material, a heated blanket was used to raise rat body temperature, lowered by the effect of the isoflurane. Blood sampling commenced approximately 5 min after the rat was placed on the electric blanket in order to permit vasodilatation of the tail veins. In Phases III and IV, rats were handled by the manipulator and placed in the anaesthetic chamber with oxygen for 1 min and then returned to the cage once a day for 1–2 weeks prior to samplings in order to get the rats accustomed to laboratory handling procedures.
Blood sampling procedure
Blood samples (0.5–1 mL) were taken from the retrobulbar venous plexus or the tail vein after anaesthesia on consecutive days (Table 1) using microhaematocrit tubes Na-hep BRAND® and 24G (0.7 × 19 mm) BD Insyte™ IV catheter respectively and placed in gel serum separator tubes (BD Microtainer™).
Processing of the blood samples
Blood samples from Phases I and II remained at room temperature (RT) for a minimum of 30 min until centrifugation. In Phases III and IV, 1 mL of whole blood was extracted from each rat and divided into two tubes to be stored at RT or refrigerated for a minimum of 30 min until centrifugation, after all samplings had been completed. In Laboratory 1, blood samples were centrifuged at 4°C for 15 min at 2778
Analysis of the results
In Phases I and II, the statistical analysis of results was based on the Student's t-test to assess the influence of anaesthesia and site of sampling on the CK level. In Phases III and IV, the results were obtained by repeated measure ANOVA. Statistical significance was established at α = 0.05. The analysis was performed using the SYSTAT version 9.0 statistical program.
Results
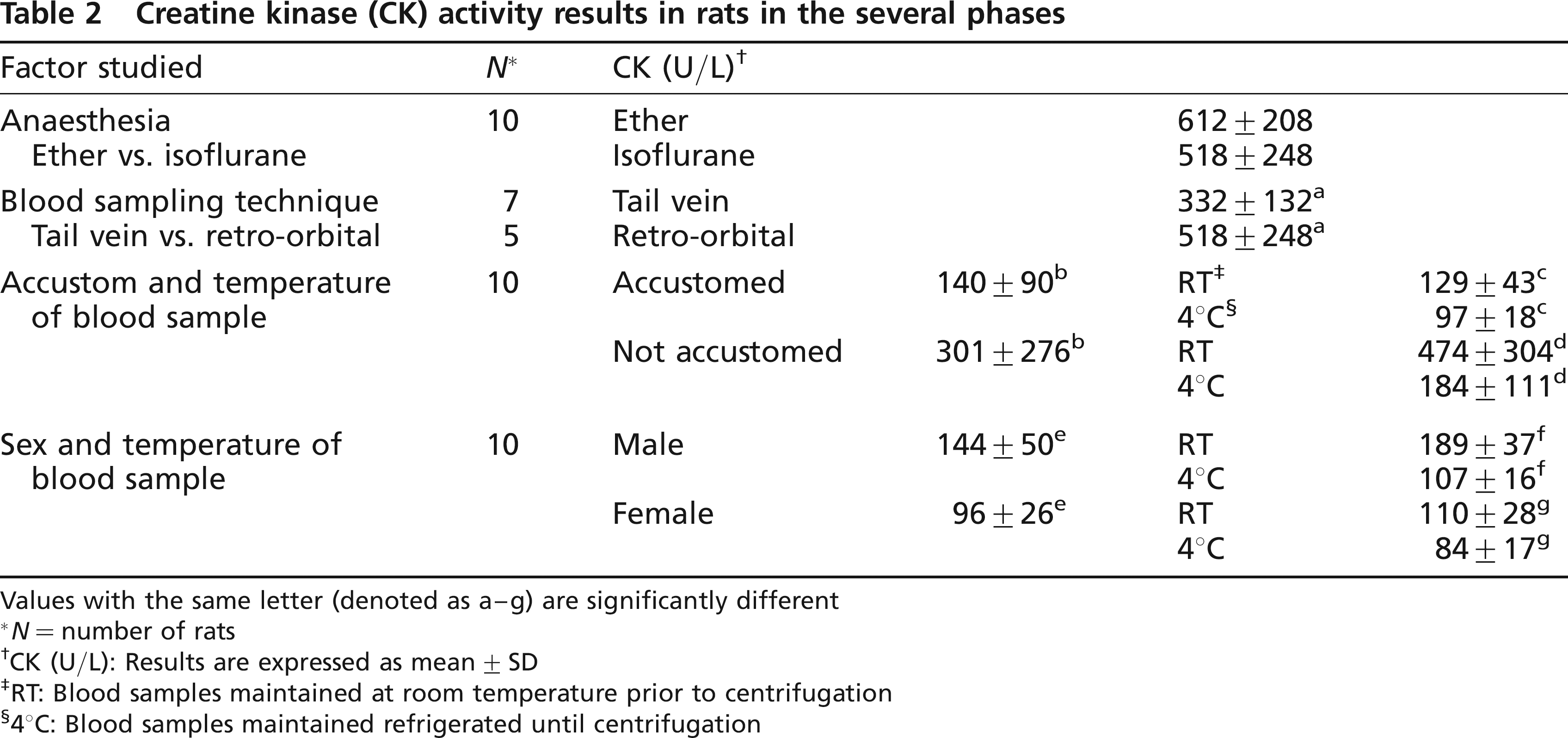
Results are summarized in Table 2.
Creatine kinase (CK) activity results in rats in the several phases
Values with the same letter (denoted as a-g) are significantly different
N = number of rats
CK (U/L): Results are expressed as mean ± SD
RT: Blood samples maintained at room temperature prior to centrifugation
4°C: Blood samples maintained refrigerated until centrifugation
Ether did not significantly increased CK values. A high variability in CK activity (coefficient of variation [CV] = 34% with ether and 48% when using isoflurane) was observed. For ethical and safety reasons isoflurane was used in the rest of the experiments.
Average CK activity was significantly higher when sampling from the retro-orbital sinus than when using the tail vein as the site of puncture, although high variability was still observed (CV = 48 and 40%, respectively).
We assessed the effect of refrigeration of the blood sample prior to centrifugation and the effect of habituating the rats to laboratory procedures over a period of one week. The mean CK value and variability were lower in rats accustomed to laboratory handing procedures. Moreover, refrigeration diminished CK mean value and variability in sera from both groups, although it was more pronounced in rats habituated to laboratory handling procedures than in those not habituated (CV = 19 and 33%, respectively).
Finally, the CK in samples from 10-week-old male and female Wistar-Han rats after being accustomed to handling procedures was determined. Mean CK activity was found to be lower in the male rats. There was also a significant difference between refrigerated and non-refrigerated blood samples for both sexes, with a greater reduction after refrigeration being observed in the male rats.
Thus, mean CK value of 10-week-old male and female Wistar-Han rats after being accustomed to handling procedures for two weeks, anaesthetized with isoflurane, sampled from tail vein and blood refrigerated prior to centrifugation and serum separation was shown to be 107 ± 16 and 84 ± 17 with a CV of 15 and 21%, respectively.
Discussion
The aim of this study has been to analyse a number of factors affecting serum CK activity in order to minimize variability and improve the reliability of the measurement of CK activity in neuromuscular research using rats. Some aspects affecting this determination might be:
Blood sampling site
Numerous blood collecting methods have been described for the rats that have been shown to have an impact on haematological and biochemical parameters (Dameron et al. 1992, Matsuzawa et al. 1994, Gummert et al. 1999, Mahl et al. 2000). Periorbital puncture may cause adverse effects including retro-orbital haemorrhage, corneal ulceration, damage to the optic nerve, fracture of the osseous orbit puncture of the vitreous body, necrotic dacryoadenitis of the Harderian gland and retinal degeneration (McGee & Maronpot 1979, Krinke et al. 1988, van Herk et al. 1992, Diehl et al. 2001). In spite of this, retro-orbital puncture is still widely used. This may be due to the lack of either scientific proof of negative consequences when performed by trained staff or the lack of equally good alternatives.
We found significantly higher values of CK activity in samples from the retro-orbital plexus than those from the tail vein that might suggest a more severe degree of tissue damage and a higher degree of haemolysis in such samples. Haemolysis in test specimens affects not only haematological parameters but also blood coagulation and various biochemical parameters such as CK (Hall 1991). Increased values and variability of CK in comparison with other sampling sites were also reported by other authors (Friedel et al. 1975, Horton et al. 1986, Matsuzawa et al. 1993). Moreover, according to endocrinology data higher stress is associated to the retrobulbar sampling procedure when it is compared with other sampling sites such as the sublingual vein (Mahl et al. 2000). Thus, puncture of the tail vein may represent a reasonable alternative to retro-orbital sampling for neuromuscular research purposes.
Habituation to laboratory handling procedures
Stress has been reported to be the factor that has the greatest effect on CK activity. Plasma CK activity in native rabbits increased by up to 10-fold if sampled every 2 h to 12 h but showed no alteration when the rabbits had been accustomed to laboratory handling procedures for two weeks (Lefebvre et al. 1992). The same phenomenon has been reported in pigs (Addis et al. 1974) but not in dogs or horses (Aktas et al. 1993, Volfinger et al. 1994). The physiological origin of this variation in CK activity appears to be a consequence of the emotional stress caused by handling, possibly associated with stress due to the vein punctures (Bacou & Bressot 1976). We have observed that there is a statistically significant reduction of CK activity in rats accustomed to handling procedures for one week, as previously reported in other animal species (Lefebvre et al. 1992).
Storage conditions
Blood sample storage conditions until centrifugation was another determining issue in CK activity observed in this experiment. We found significant increases in CK activity in non-refrigerated blood samples that might be attributed to contamination by CK released from platelets. In 1975, Shibata and Kobayashi demonstrated that substantial amounts of CK leaked out of rat platelets and suggested that CK might contribute to some reactions which consume ATP during aggregation, presumably donating the energy for shape change (Shibata & Kobayashi 1978, Shibata 1978). Importance of sample preparation after blood sampling in regards to platelets interference has also been reported by others (Matsuzawa et al. 1993) for lactate dehydrogenase (LDH), RBC, glutamate oxalate transaminase (GOT) and K measurement as well. The elevation of LDH and GOT and CK activities in whole blood was attributed to the liberation of these enzymes from platelets during the clotting process (Matsuzawa et al. 1993).
We suggest that CK might be liberated from rat blood platelets, and that this increases with the time allowed for clotting before centrifugation, as also occurs with LDH, malate dehydrogenase and GOT (Holmsen et al. 1969, Friedel & Mattenheimer 1970, Aktas et al. 1994). This increase might not be observed in other species such as dogs, as previously reported (Aktas et al. 1994). It could also be postulated that refrigeration may cause a reduction in rat platelet metabolism, possibly by reducing creatine phosphate (CP), as observed in 1975 by Shibata and Kobayashi when platelet rich plasma (PRP) was stored at 4°C for 45 min and the CP content in the rat platelets fell to 90% of the initial value (Shibata & Kobayashi 1978). Although it has not been confirmed, the elevated CK from platelets might be of the brain-type (CK-BB), as Meltzer and Guschwan (1972) only found this isoenzyme in rat platelets.
Some authors recommend the use of plasma in studies using laboratory animals, particularly rodents, in view of the marked release of intracellular substances from blood corpuscles, platelets in particular, during blood coagulation (Stevens & Gallo 1982, Matsuzawa et al. 1993).
Gender
Significantly lower CK activity was found in female than in male rats, as previously reported in Sprague-Dawley rats by some authors (Kaspar & Norris 1977, Loeb & Quimby 1999) and in contrast with published results of others (Matsuzawa et al. 1993). Sex differences in rats have been previously reported for other parameters such us haemoglobin concentration (Hb), haematocrit (Ht), white blood cell count (WBC), glucose, cholesterol, triglycerides and total protein (Matsuzawa T et al. 1993).
In the light of our findings, CK value and variability in rats may be minimized by choosing the tail vein as the site of bleeding, and getting rats accustomed to laboratory handling procedures and maintaining whole blood refrigerated until centrifugation and serum separation.
Footnotes
Acknowledgements
We would particularly like to thank Marian Ostáriz, from the Experimental Unit, for her continuous dedication to this research study and her technical assistance. We are also grateful to Arantxa Begiristain, from the Biochemistry Department, for the rapid analyses of the CK. We would also like to thank Dr Otaegui and Dr Ferre from the Center for Animal Biotechnology and Gene Therapy in Barcelona, and Dr Martín from the ‘Minimally Invasive Surgery Centre’ (CMI) in Cáceres, for their initial guidance. Finally, we are also grateful to all the members of the Experimental Unit for their constant support and collaboration.
