Abstract
An improved knowledge and understanding of the fundamental biological requirements is needed for many of the species of fish held in captivity and, without this knowledge it is difficult to determine the optimal conditions for laboratory culture. The aim of this paper is to review the key issues concerning environmental enrichment for laboratory-held fish species and identify where improvements are required. It provides background information on environmental enrichment, describes enrichment techniques currently used in aquatic ecotoxicology studies, identifies potential restrictions in their use and discusses why more detailed and species-specific guidance is needed.
In the UK, all establishments conducting scientific procedures with animals are controlled under the Animals (Scientific Procedures) Act 1986.
1
The Act deals with key objectives that relate to improving the care and welfare of laboratory animals, and all establishments conducting scientific procedures are required to consider the reduction, refinement and replacement of animals in experimentation. In the UK, fish are the third most commonly used experimental animals after mice and rats, and figures suggest a slow but increasing upward trend in the numbers of fish used in scientific procedures. Of the 2.9 million scientific procedures on animals started in 2005, 14% were for toxicological purposes associated with the protection of man, animals or the environment. Approximately 8% of all animals used in scientific procedures, and 10% of all animals used for toxicology, were fish, the majority of these to comply with legislation or other regulations.
2
With animal welfare in mind, and considering the advances made in the use of environmental enrichment techniques for improving the welfare of captive mammalian species, the Home Office (HO) Inspectorate who administer the Act consider it timely to consider such approaches for improving the welfare of captive fish (see
While environmental enrichment techniques have been applied extensively to mammalian systems, limited information exists concerning the application of such procedures to aquatic organisms such as fish. Handley 3 discussed the potential problems associated with the application of environmental enrichment for fish used in regulatory toxicity studies, and highlighted the limitations posed by the need for studies to meet regulatory compliance with test guidelines. In the intervening time, good progress has been made to reduce and replace fish use in certain types of regulatory toxicity studies. 4–6 However, progress with environmental enrichment has been slow, not by design but by a realization that fundamental knowledge of the biology of many captive fish species used for toxicology studies is limited and falls well behind the comparative knowledge for mammals. In the aquaculture industry, despite a wealth of knowledge on the physiological consequences of many aquaculture practices much work remains to be done to better understand the biological and environmental requirements for different fish species. 7,8 In these circumstances, it is important that the consideration and implementation of environmental enrichment procedures for aquatic species such as fish, is based on sound scientific principles and an understanding of the practical issues involved. The aim of this paper is to consider the principles and practicalities of environmental enrichment procedures for fish used for the purposes of toxicity testing associated with the environmental risk assessment (ERA) of chemical and biological products.
Animal welfare requirements/regulations
Benefiel
Use of fish in environmental safety assessment
The environmental safety assessment of chemicals is a regulatory requirement and acute and chronic toxicity tests with fish are a key component of ecotoxicity testing schemes for measuring the potential adverse effects of substances and aqueous discharges. 11,12 While initiatives have been undertaken to reduce, replace and refine the use of fish in acute toxicity tests, 4–6,13 toxicity tests using fish will remain an important component of regulatory ERA schemes for measuring the potential long-term effects of chemicals such as pharmaceuticals. 11 Prompted by concerns that specific types of chemicals (e.g. endocrine-disrupting chemicals [EDCs]) have the potential to cause long-term sublethal, rather than acute effects, regulatory approaches have focused on methods for measuring developmental and reproductive endpoints in fish. 14 Ankley and Johnson 15 provide a general overview of the use of small fish species for identifying and assessing the effects of EDCs and conclude that fish models will continue to play an important role both in research and regulation of such substances.
To conduct partial- and full-life cycle tests with fish successfully, however, it is generally necessary to have established conditions and expertise consistent with maintenance of a reproducing laboratory culture of the fish species used in the toxicity tests. Consequently, many test facilities will maintain breeding populations of certain fish species in order to provide animals for testing. In other cases, juvenile fish may be purchased from suppliers and held in the husbandry facility for relatively short periods of time until toxicity tests are performed. In either case, the welfare or wellbeing of fish used in toxicology studies is of paramount importance as poor animal welfare can potentially compromise the scientific validity and repeatability of data.
Fish also offer significant advantages over mammalian systems in certain types of toxicological studies. For example, the utility of fish as surrogates for other vertebrates has seen a significant increase in the use of small fish models in drug discovery and biomedical research. 16–24 In these circumstances, the continued use of fish in environmental safety assessment, and as replacements for mammalian testing, seems set to continue, albeit with clear justification that such procedures are necessary.
Fish as sentient animals
Much of the prevailing discussion on science, ethics and fish welfare assumes fish to be sentient animals. While it is known that fish have the capacity to perceive noxious stimuli, the question of whether they have the capacity to experience any of the adverse states usually associated with pain in mammals or birds is currently subject to considerable debate in the scientific literature.
8,25–28
Evidence suggests that some fish have the sense organs and the sensory processes required to perceive harmful stimuli and, probably, a central nervous system capable of experiencing at least some of the adverse states that are associated with pain in mammals.
25,26,29–32
Huntingford
These principles can readily be applied to terrestrial species, however, it is more difficult to apply some of these criteria (e.g. freedom from fear and distress, freedom from discomfort) to aquatic species such as fish. To accept this means that we must accept a degree of sentience in fish that to date is at best controversial.
25–28
Therefore, applying the anthropocentric concepts of fear, distress and discomfort directly to fish has, as yet, no firm grounding. Keeping an open mind to the possibility that fish are proven sentient in the future, we can loosely use the anthropocentric states such as fear, pain, distress and sentience to describe the more primitive ‘motivational affective states’ that are common to vertebrates when we consider fish ethology. We should exercise care in differentiating between the meanings of these terms when applied to man and when applied to fish. As experiments designed to specifically test whether fish behaviour is determined by procedural or declarative representations have not been published (suggesting that they are yet to be conducted), re-interpretation of the existing literature in a
Environmental enrichment as a component of fish welfare
Although there is general agreement that animal welfare concerns the wellbeing of the animal, there has been considerable debate among scientists and philosophers about how this concept should be understood in practice. The welfare of an animal is seen as its physical and mental state, as well as the protection from unnecessary suffering, and can be defined using the ‘Five Freedoms’, as defined by the Farm Animal Welfare Council's (FAWC) 34 revised version of the freedoms initially proposed by the Brambell Committee in 1965, and included in the World Organisation for Animal Health (OIE) guiding principles for animal welfare. 35 Freedom to express normal behaviour is one of the five freedoms defined by the FAWC, however, some have the opinion that the ‘naturalness’ of an animal's behaviour does not necessarily have a direct positive or negative correlation with welfare. 8 Other approaches for defining what constitutes fish welfare have involved reworking of the five welfare domains specified by Mellor and Stafford 36 for farm animals into a form that is more appropriate for fish. 37 Applying the welfare principles described for terrestrial farm animals (Table 1) to aquatic animals is a complex and challenging task as this involves several factors unique to aquatic animals. In contrast to terrestrial animals, aquatic animals encompass extremely diverse, divergent and distantly related taxonomic groups of greatly varied phylogenetic ages and linkages. 38 While there have been considerable improvements in environmental enrichment procedures for terrestrial animals, comparatively less attention has been applied to non-mammalian aquatic species and, as described above, the challenge has been to apply and/or adapt welfare principles adopted primarily for terrestrial mammalian species to aquatic species. Although it is difficult to define and measure scientifically there appears to be general agreement that an enriched captive environment enhances the welfare of captive animals.
Welfare principles described for terrestrial farm animals
Fish welfare is an area of increasing public and regulatory concern and recent publications provide a current understanding of the key issues – what it means, why it matters, what humans do that may compromise it and how, in practical terms, it might be measured. 8,33,37–40 In the UK, there is a legal requirement to consider the welfare of animals used for scientific procedures, and workers share a legal and moral obligation to respond to concern on this issue. Although the issue of sentience in fish remains open to debate, the evidence supports a pragmatic approach that adopts a simple working position that fish welfare matters, both for moral and practical reasons.
Background to environmental enrichment
The environment and holding facilities of laboratory animals have often been designed on the basis of economic and human ergonomic requirements, with little or no consideration for animal welfare. Laboratory housing conditions can, therefore, deprive animals from performing a full repertoire of normal behaviour and, as a response to this lack of stimulation, animals may show abnormal behaviours. 41 Stereotypical behaviour is believed to be an indicator of poor welfare and has been well documented in higher vertebrates. 42 This deviation from natural behaviour creates unnatural stress levels (e.g. altered blood chemistry and metabolism), and other behavioural problems (e.g. aggression). In addition to the consideration of humane treatment, these problems can adversely influence the results of experimental studies and reduce the scientific validity of any resultant data. Unfortunately, for scientific reasons, it is often necessary for animals to be placed in conditions that, while providing all the basic elements required for survival, are clinically sterile and devoid of any enrichment.
With the advent of modern ethological studies, biologists better understood the behavioural, dietary, veterinary and environmental needs of captive wild animals, so that their physical and psychological health could be taken into consideration. 43 As a result, there has been a trend towards constructing larger and more naturalistic habitats intended to replicate many aspects of an animal's natural habitat. 44 This has led to the development of environmental enrichment programmes using various techniques to create more interesting and interactive captive environments for zoo and laboratory animals. 45
Environmental enrichment is a challenging concept to define precisely and although definitions may vary, the common emphasis is on the provision of a more stimulating environment.
46
For example, environmental enrichment has been defined as altering the living environment of captive animals in order to provide opportunities to express more of their natural behavioural repertoire.
47
Most animal care professionals would agree that an enriched captive environment enhances the psychological and physiological wellbeing of animals under our care; therefore we are assuming that enrichment enhances animal welfare.
48
However, evidence that environmental enrichment benefits an animal's wellbeing is not conclusive for all species. For example, based on studies of laboratory rodents, Benefiel
The definition of environmental enrichment is important because it determines how enrichment should be measured and what indicators should be used. For example, definitions based on expression of normal behaviour, included in the ‘Five Freedoms’ 34 and five welfare domains 36 imply an understanding of what constitutes normal behaviour in captive species (Table 1). The difficulty in specifying what is ‘normal’ behaviour for a captive animal has been identified previously. 49 Also, interpretation of behavioural measures in isolation is subject to doubt. 50 Even for well-studied species such as rodents, a recent review concluded that the mechanisms by which rodents respond physically to environmental challenges are not understood sufficiently to implement them in a knowledgeable manner. 9 With many species of aquatic animals, for which basic knowledge is limited, the problems of defining what are normal behaviours are further compounded by the lack of knowledge and awareness of the specific requirements of those species in captivity. Despite these reservations, however, the published literature espouses the view that environmental enrichment is by definition a good thing, without consideration of the possible effects on experimental outcomes or evidence that it is essential for animal wellbeing. 9
Compared with mammals, limited information exists concerning the application of environmental enrichment procedures to aquatic organisms other than cephalopods.
51,52
This lack of information is surprising, given that fish are widely used in research in the UK and currently are the third most widely used taxonomic group in terms of numbers.
2
The
Sheperdson
Techniques for measuring effects of enrichment
When we assess wellbeing, we commonly look for signs that animals are physiologically and behaviourally well adapted to the environment, are thriving and demonstrate a normal appetite, growth rate and breeding performance.
46
In order to determine which environmental enrichment techniques provide actual benefit, and not just a perceived increase in welfare, visual or otherwise, it is necessary to identify suitable methods to assess both the positive and negative effects of any proposed enrichment technique. This is essential, because without the ability to quantify the results of any study, the data cannot be correctly interpreted. Both the National Research Council (NRC)
All animals have a wide range of species-specific behaviour that can be displayed if they are provided with an enriched environment that can facilitate the behaviour and potentially improve the animal's wellbeing or welfare.
46
As the scientific debate on the ability of fish to suffer develops, behavioural aspects will become increasingly important for balanced welfare assessment.
8
Unfortunately, for many species of fish, adequate knowledge of species-specific behaviours is lacking, therefore, it may be difficult to provide a baseline of information that can then be applied to fish held in a captive environment. Newberry
49
points out the difficulty in specifying what is ‘normal’ behaviour for an animal that has been selectively bred for captivity. Although retaining many behaviours of its wild ancestors, a laboratory animal should be expected to have a behavioural repertoire that is different and better adapted to confinement.
9
Observation of species-specific behaviour can provide a valuable indication of the animal's wellbeing, however, without a good understanding of what is normal species-specific behaviour for fish, and how it can be measured, it may prove difficult to evaluate how such changes contribute to wellbeing. The most suitable forms of test to quantify the positive and negative benefits of any enrichment would be non-invasive, as these are unlikely to bias results due to the induction of stress or stereotypical behaviour. If the focus of enrichment is to provide environments that enhance animal wellbeing by maximizing species-typical behaviour and minimizing stress-induced behaviour, then some reliable and valid measures of wellbeing that assess these criteria are needed.
58
Non-invasive methods include measurement of physiological stress responses such as cortisol concentrations in water
59
or in their faeces.
60
However, these techniques may not be specific to poor welfare and lack the precision of direct measurements on individual fish. Simple, non-intrusive indices of welfare are required for practical management of welfare, and a number of possible welfare indicators based on direct observations of fish are described by Huntingford
Application of enrichment techniques
General principles
Using environmental enrichment for improving the welfare of fish is an important objective and has relevance in both husbandry and toxicity testing. There are many different approaches to environmental enrichment commonly used by aquarists (Table 2). It may be possible to employ some of these techniques to broodstock fish reared in the laboratory to supply testing requirements. However, due to the prescriptive nature of regulatory test guidelines, and the scientific requirements associated with these studies, it may be difficult to implement many of these procedures in laboratories performing regulatory toxicity tests. With this in mind, it is important to balance the enrichment refinements with the need to have a scientifically valid, high-quality study so that refinements do not compromise results and require repeat testing, thus elevating animal usage. 3 It is important, therefore, to achieve a balance between effective enrichment programmes that could be transferred from the husbandry aspect of animal production directly into regulatory studies, without compromising study robustness.
Commonly used techniques of environmental enrichment within the home aquaria (modified after Handley 3 )
While it would be advantageous to have enrichment techniques that could be applied to similar species of fish (e.g. cyprinids), the wide diversity in natural history and wide range of physiological and behavioural traits, suggests that a species-specific approach may be required. For example, some of the most common fish species used in the laboratory for the purpose of toxicity testing are shown in Figure 1. There is no single ‘model’ fish and the choice of surrogate species is based on both biological and practical considerations. However, a fundamental understanding of the biology and ecology of the different species used for culture and toxicity testing will provide a good foundation for considering the options for environmental enrichment. For example, Figure 1 shows that although most of these highlighted species are cyprinid species, differences between the species and among the ontological stage within the species exist.
Figure highlighting the differences in stocking density and water flow at different ontological stages for species of fish commonly used in toxicity testing. The outer three rings (large grey circles) represent three potential stocking densities (fish held individually, in pairs [or spawning groups], and in group housing). The inner three rings (small grey circles) represent three potential water flow rates within typical husbandry systems: (slow 0–3, medium 3–10, fast >10 cm/s)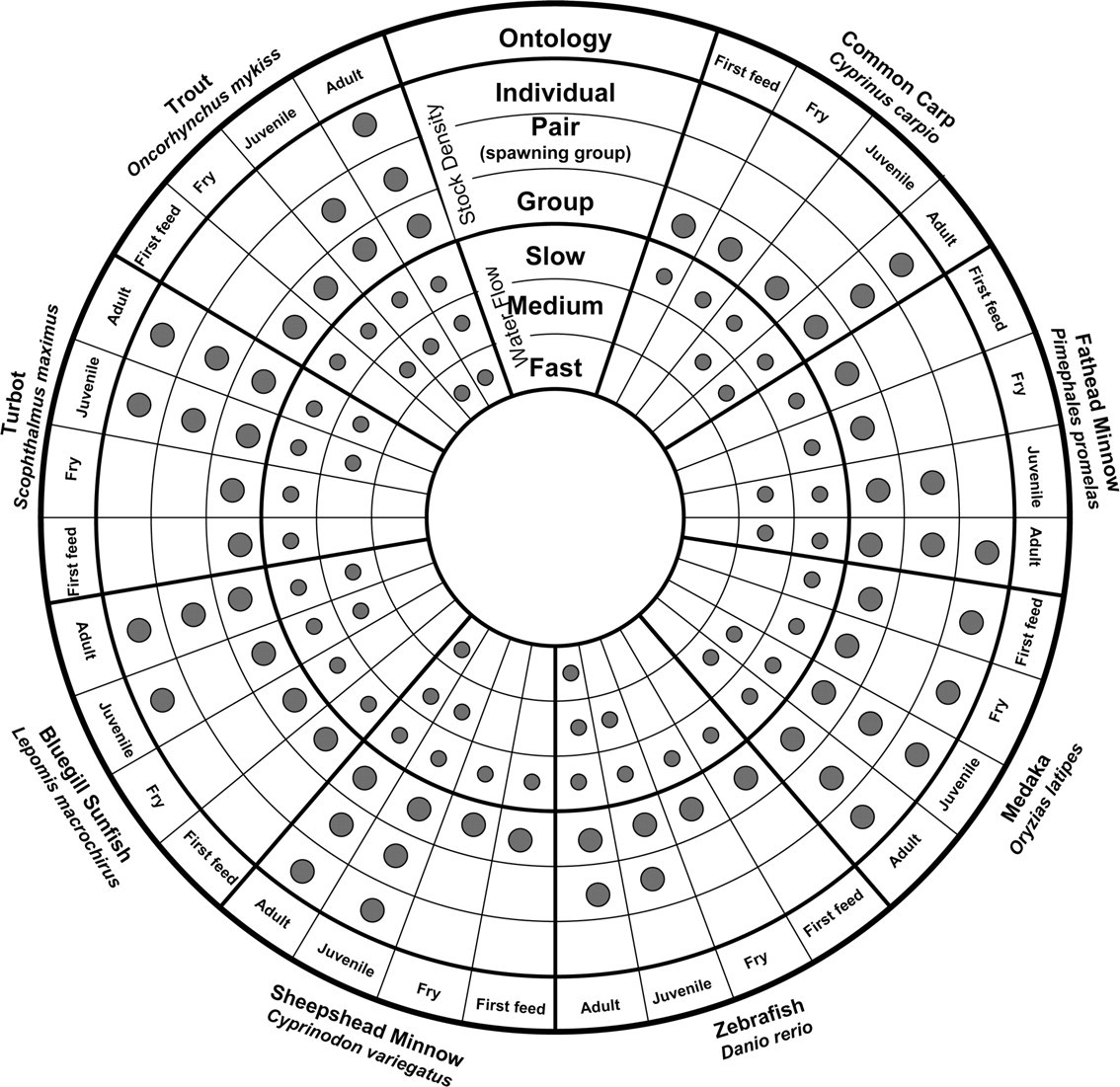
As described earlier, the natural history of an animal can be used as an initial guide for defining an appropriate captive environment for the species in question.
48
This approach can help identify the key scientific and practical issues involved and show where potential improvements in environmental enrichment can be made in both husbandry and test situations. The review of natural history/ecology need not be exhaustive (e.g. information of the natural history of some species may be limited by comparison with well-studied species such as fathead minnow), but can provide a valuable starting point for initiating or improving laboratory culture procedures for captive fish species. For example, the fathead minnow (
The fathead minnow is a member of the ecologically important Cyprinidae family. It has a relatively broad distribution in both lotic and lentic environments across North America, and is tolerant of a wide range of basic water quality characteristics including pH, alkalinity/hardness, turbidity and temperature. 66 The species has been described as an opportunistic herbivore, with a diet consisting primarily of detritus, algae and other plants, insects, small crustaceans and other invertebrates. 67–70 It is a strongly shoaling fish and courtship/reproductive behaviour is elaborate and relatively well defined. 66,71 During the breeding season, fatheads are sexually dimorphic and, from late spring through late summer, males establish territories around suitable spawning sites 72–75 and prepare the nests for spawning. From these territories, the male performs distinctive courting and leading behaviour 76 and, if courtship is successful, females will deposit a batch of eggs on the underside of the defended nest structure. Females produce from 4000 to 5000 eggs per year, 77,78 and the incubation period is about 4–5 days (at 25°C). Following hatch, the larvae are active and begin feeding almost immediately. 79 Under optimal conditions, the fathead achieves reproductive maturity within 4–5 months of hatch and, depending on water temperature and photoperiod, can spawn continually for several months. 80 These details of the natural history of fathead minnows illustrate a complex and elaborate series of behaviours that should be taken into account for laboratory culture of this species. As described in the following sections, many of the key biological/behavioural requirements of this species have been recognized and can, therefore, be incorporated into current laboratory culture and test regimes where practicable. This approach can be applied to different species of fish and used to identify where potential improvements can be made and the practical issues involved.
Enrichment techniques used in toxicity tests
Restrictions
Considering toxicity testing, the maintenance of populations of fish with a proven health status is not only a regulatory requirement, but is essential for providing scientifically robust and valid toxicity data. In contrast, using fish of poor health will most likely produce inferior data, invalidate studies and will require repeat testing, which would lead to increased animal use. Many toxicity tests are performed to regulatory guidelines, which are very prescriptive in nature and impose strict scientific validity criteria, which, if not met, invalidate the study. In toxicity testing, therefore, these considerations may limit the enrichment procedures that can be applied, without compromising the scientific validity of the study. By comparison, husbandry procedures are not subject to the same constraints. However, when considering what improvements can be made in husbandry, it is important to consider the implications of such changes upon transfer of animals from husbandry to test. Generally, fish in both husbandry and testing are maintained under the same (or very similar) conditions, so as to minimize stress upon transfer to the test environment. This is an important point as potential differences in welfare conditions between husbandry and testing may exacerbate the problems of transfer and acclimatization of fish between the different locations (and holding conditions).
Maintaining exposure concentrations under test conditions can be difficult, especially for chemicals that are prone to such factors as biodegradation and photolysis. 81 The environmental conditions required for maintaining fish in the exposure system (e.g. relatively high temperature of 25°C and long daylight photoperiod of 16 h light: 8 h dark for fathead minnow) are likely to exacerbate these problems and others, such as excessive promotion of microbial growth.
With these requirements in mind, enrichment procedures that involve the addition of substrates into the test system must be avoided as they may increase the surface area for chemical binding or microbial growth to occur and, therefore, potentially interfere with the exposure chemistry of the substance under test. Certain materials such as polycarbonates may also contain chemical components (e.g. phthalates) that, if released into the test vessel, are above limit values prescribed in the regulatory guidelines. These materials may also exert adverse biological effects on the test organisms and invalidate studies. For example, oestrogenic substances associated with several materials, even those of a medical grade, are well documented. 82
The use of gravel or sand substrates in exposure tanks has been proposed as environmental enrichment for some species of fish (e.g. flatfish). However, in addition to the type of problems described above, substrates can harbour parasites and disease-causing organisms (e.g. White spot cysts [
In many toxicity experiments, carrier solvents (e.g. acetone, methanol) are employed to aid solubilization of the test chemical. Solvents tend to stimulate microbial growth, and additional surfaces (e.g. from spawning substrates added for environmental enrichment) may promote further growth. Enhanced microbial growth may also exert a biological oxygen demand in the test medium causing dissolved oxygen (DO) concentrations to fall to concentrations that adversely affect fish survival, growth and reproduction. To combat this, some regulatory guidelines may permit the use of aeration to raise DO concentrations, however, potential loss of volatile chemicals through enhanced volatilization during aeration is a concern. Practical experience has shown that microbes can interact with the test chemical to influence exposure concentrations, and can also have adverse effects on the biology of the fish (e.g. promote fungal growth on fish eggs in reproduction experiments). Enhanced cleaning regimes can be employed to combat microbial growth, however, these procedures may influence chemical analysis and are likely to increase disturbance of the fish in the exposure system.
Although the factors described above may limit or exclude some options for environmental enrichment, there are others that can be employed in test systems that maintain and improve fish welfare (see below).
Techniques currently used
As described above, there are restrictions to what enrichment procedures can be applied to toxicity tests. However, several enrichment procedures have already been applied which do not appear to cause these problems. For example, in fish full life-cycle (FFLC) studies with the fathead minnow conducted in our laboratory, the size of the test vessels has been increased to provide additional space (water volume) for individual breeding pairs. External shading was also provided which, in addition to reducing disturbance of fish by test operators, visually reduced the light intensity within the breeding chambers. While reproduction by individual breeding pairs in control treatments held within this experimental design was good, further experimental work is needed to establish the effect of these factors on reproductive behaviour and breeding success. In reproduction studies, a number of factors are likely to subtly influence breeding behaviour and relatively small changes can influence the welfare of fish and performance in toxicity tests. By utilizing knowledge of species innate behavioural traits, and adapting the test environment to allow more natural behaviours, it may be possible to produce more reliable and robust toxicity data.
The introduction of external shading of tanks to minimize disturbance and optimize breeding conditions for specific species of fish deemed to benefit from such conditions is relatively simple to carry out. However, as described above, these procedures need to be validated before they are introduced into chronic toxicity tests. In the above case, while the changes made to shading of test vessels holding fathead minnows suggested an improvement in reproduction, results from one study reported a higher than usual incidence of early mortality of female fish in breeding pairs. Mortalities appeared to occur randomly across the control and exposure treatments, suggesting that these effects were not due to test substance exposure. Visual observations of the dead fish suggested that aggressive breeding behaviour by individual male fish was a causative factor (e.g. skin damage to the female leading to secondary infections). As shading was provided between individual replicate tanks, there was no opportunity for visual ‘interaction’ between male fish in adjacent replicate tanks (separated by a glass panel). It was hypothesized that, without these visual interactions, which stimulated the male fish to defend its breeding territory from the perceived threat from other male fish, aggressive behaviour was directed more towards the female of the breeding pair. Although subsequent studies have not shown a similar occurrence, this example provides a cautionary note and illustrates how changes made to test procedures, with the purpose of improving welfare and performance of fish, can sometimes produce unforeseen changes in test organism biology that can influence the outcome of experiments. Considering that changes in environment can have a considerable effect on the physiology and behaviour of fish, it should be of little surprise that changes in environment can influence test results and affect the validity of experimental data. Published data for mammalian species provide supportive evidence that alterations in environment can affect the validity of research results. Based on a review of alterations in laboratory rodent environments on research, Benefiel
Although photoperiod length is prescribed under regulatory guidelines, lighting systems can be controlled to provide a timed transition at dawn/dusk periods. The gradual, controlled change (increase or decrease) in light levels eliminates potential stress to fish in the form of a startle response caused by sudden changes in light intensity. Startle is a fast response to sudden intense stimuli and probably protects the organism from injury by a predator or by a blow. 83 The phased transition and length of photoperiod also help maintain healthy broodstock as in many species day length is a trigger to seasonal spawning, and eggs are often laid soon after dawn.
Food quality and quantity are important factors in animal welfare. Diets used in sublethal/chronic toxicity test procedures are selected carefully to provide optimal nutrition and meet the requirements of the test (e.g. maximize growth and reproduction in life-cycle studies). Considering environmental enrichment, the use of live foods can stimulate foraging behaviour and improve body condition, as fish take a more active role in seeking food. Live diets are particularly important in the early developmental stages of fish in partial and full life cycle studies with fathead minnow and, in addition to providing essential nutritional components, may possibly provide environmental enrichment. Live diets may, however, not always be practical in toxicity experiments (e.g. irregular supply, inconsistent nutritional value and poor quality control) unless cultured in-house.
In exposure systems, physico-chemical environmental conditions (e.g. water quality, temperature, photoperiod) are controlled within a relatively narrow range. These parameters are selected to provide optimal conditions for survival, growth and reproduction of control animals within the specific test, and regulatory test guidelines specify control validity criteria (e.g. percentage survival must exceed 90% throughout the test). In considering enrichment options, although the control of physico-chemical environmental conditions may be prescribed in test protocols, it is important to recognize that some of these practices provide a source of environmental enrichment and contribute to improvements in fish welfare (see below). Application of some of these procedures within the fish husbandry unit in our laboratory is described below in more detail.
Further options
The use of optical barriers within test vessels has been shown to be an effective tool for reducing aggression and test substance-independent mortality, when conducting acute or chronic fish testing using rainbow trout. 84
Due to the potential problems posed by the addition of substrates (discussed earlier), it is necessary to investigate other options such as external shading and patternation to disrupt surfaces. Therefore, for specific test requirements (e.g. reproduction studies), there is scope for further investigative work to better define the effect of key parameters on biological performance (e.g. breeding behaviour and reproduction). Improving biological performance within tests has additional benefits by removing or minimizing potential sources of variability in the biological data that confound the interpretation of results. These considerations are particularly important for long-term chronic studies, where confounding factors can compromise the interpretation of data and, at worst, can invalidate a study, requiring repeat testing and increased animal use.
Provision of sufficient space (area and quality) is an important consideration for the welfare of mammalian species. While loadings of fish per volume of water are generally kept low in toxicity tests, the effects of tank size (dimensions and water volume) on factors such as breeding behaviour of even the most common laboratory species are relatively unknown and warrant further investigation.
Enrichment techniques used in husbandry
Techniques currently used
When considering environmental enrichment, husbandry of animals prior to testing and maintenance of broodstock are not subject to the same constraints that apply to toxicity testing. Within the husbandry unit, environmental parameters such as water quality, temperature and salinity are usually controlled within a relatively narrow range of values that provides optimal welfare for the species of fish being cultured. These ‘core’ parameters are required to be the same as those applied to toxicity tests for a prescribed period of time prior to the tests, thereby minimizing the potential stress involved in the transfer and acclimatization of fish from husbandry to testing. However, during acclimatization and holding periods, parameters may be held at levels more beneficial to the animal's wellbeing. Depending on the definition used for environmental enrichment, some or all of the core parameters described below, which provide the basic requirements for fish husbandry, may be considered to provide some form of environmental enrichment for fish.
Water quality
Fish inhabit a complex three-dimensional medium and are influenced by their environment to a much greater degree than terrestrial species. Consequently, control of water-quality parameters such as temperature, pH, hardness and salinity is considered important for meeting the fundamental biological requirements of fish species. 39 Whether these welfare parameters provide enrichment is open to interpretation. For example, husbandry tanks may be aerated (using compressed air) to provide additional oxygenation. The motion of air bubbles, their effects on mechanical vibration/noise and water movement could provide an additional stimulus for the fish stocks. However, this may prove beneficial for some species, but detrimental to others.
Flow rates
Species of fish may have different requirements for flow rates. For example, in the natural environment, young salmonid fish such as rainbow trout are usually located in fast-flowing water, however, their ontology means that salmonid fish must adapt to a variety of habitats at different stages of their life cycle, whether that is fast flowing shallow streams, lakes or the open ocean. In contrast, cyprinid fish such as the fathead minnow are more usually found in static or low-flowing watercourses. Location of fish in specific habitats may also depend upon their ability to take up available oxygen from the water, for example several species of fish have developed various ways of taking additional oxygen from the air or water. Within husbandry, flow rates are manipulated to meet the biological requirements of the different species of fish (e.g. water flows may be directed in a manner to promote swimming behaviours).
Stocking density
Stocking density in an aquatic system incorporates more than just the number of animals in a given space. 27 Density incorporates the number of fish per unit of three-dimensional space as influenced by the number of fish or weight of fish per volume of static or flowing water. 85 The combination of these factors is what influences water quality and fish-to-fish interaction, and affects the animal's welfare. 27 Appropriate stocking densities can therefore be used as an enrichment tool. Low stocking densities are used primarily to avoid overcrowding and to decrease biological load, thereby reducing stress caused by lower water quality (reduced DO, increased solids and pH shifts) and competition for space and food. Alternatively, higher stocking densities for some species (e.g. rainbow trout, arctic char) have been shown to alleviate stress-related behavioural responses. 27 There may however be a subsequent deterioration in water quality, in terms of DO, suspended solids and nitrite levels, even under flowthrough conditions, if fish numbers are maintained at too high a density. Manipulation of stocking densities is not an option in toxicity tests, where the numbers of fish per replicate are prescribed by regulatory test guidelines, and considerations for robust statistical analysis of data. However, stocking densities in tests are usually low, and sufficient to maintain good water quality.
Group housing
In husbandry (and toxicity testing), fish are typically kept in a group that allows interaction between individuals. In husbandry, fish are always kept in groups unless there are particular circumstances to isolate individuals for veterinary reasons (e.g. treatment of disease) or for behavioural reasons, such as aggression caused by dominance. Dominance can be a particular problem with rainbow trout, especially if there is a size difference, promoting the need for regular grading of stocks to keep fish in the same size class. 86
Food quality, quantity and presentation
Diets are a critical element in the welfare of fish. For species of fish such as fathead minnow that are bred in the laboratory, the early larval stages are usually fed a live diet of rotifers (
Further options
Addition of objects into tank
It is self-evident that objects can be added to tanks (e.g. artificial plants, substrates) to stimulate behaviour, however, there is little published evidence that doing so improves fish welfare. Within the aquaculture industry, changes in behaviour of salmonids has been observed in response to background or substrate colour, 87 and to rearing in enriched environments containing submerged structures, overhead cover and gravel substratum. 88 For some species used in reproduction studies, it is necessary to add objects to the tank (spawning substrates) during the reproduction phase in order to collect eggs (e.g. the use of Schesney rings allow the collection of eggs from broadcast spawners, while the use of half guttering pipe to create a cave territory allows for the collection of eggs from clump spawners). Therefore, there are specific applications (e.g. breeding of fish) when objects are added to tanks to meet a fundamental biological requirement for fish, but they may also provide an important source of environmental enrichment. Of the fish species generally used in toxicity testing, only juvenile turbot are routinely benthic and might be considered to benefit from addition of a base substrate (e.g. sand or gravel), however, the potential benefits of doing so may be outweighed by the potential problems alluded to earlier.
Manipulation of environmental parameters
Consideration should be given to implementing a phased dawn/dusk transition as this will alleviate any stress caused by sudden, steep transitions in light intensity, which could cause a startle response as defined by Koch. 83 For species where environmental factors (e.g. lunar cycles) act as triggers for spawning, it may be possible to implement such procedures to improve reproduction and hence provide environmental enrichment. The use of ‘daylight’ bulbs that produce a more natural light spectrum may also provide a more natural environment for the fish. Care to match the light intensity at the surface to those encountered in the wild will ensure that the species that routinely live at depth or in turbid waters are not unduly stressed by overtly intense light, but benefit from the correct spectral environment.
Tanks and flow rates
In many facilities, flow rates may not be sufficient to provide a swirling current in larger fibreglass tanks. Provision of much higher flow rates, or use of submersible pumps to drive water flow, would have several benefits. It would create a more natural environment for fish that prefer to hold in a flow of water (e.g. rainbow trout juveniles) and the motion of water carries detritus to the centre of the tank, thereby aiding maintenance of good water quality. This procedure has additional benefits. For example, salmonid fish, which maintain aggressive dominance hierarchies, show increased aggression and dominance in slow water, which results in reduced food intake for subordinates, and a greater size range of fish within the population. 89 The use of circular holding tanks would also provide a useful husbandry tool for crudely diagnosing the health of fish – those fish that are unable to maintain position in the flow (i.e. weaker or diseased fish) hold in slacker water making them easier to identify. Furthermore, maintaining position in a flow of water reduces the intensity of aggressive interaction, reduces the frequency of burst swimming, and encourages development of finage and body shape (Foultons condition index) more in line with that of wild fish. 90,91
Dither fish
The use of Dither fish is a practice that involves placing groups of well acclimatized fish around the tanks of particularly nervous individuals, such as those recently introduced into a husbandry unit. This may help to reduce stress, as the new arrivals will be calmed by the lack of ‘flashing’ which is associated with predator-prey interactions. Polyculture may also be used to reduce aggression and enhance growth. For example, Nortvedt and Holm 92 used arctic charr as Dither fish for atlantic salmon and found that the addition of charr increased the mean size and growth rate of salmon significantly.
Summary and conclusions
An improved knowledge and understanding of the fundamental biological requirements for captive fish species is needed, without which it is difficult to determine the optimal conditions for laboratory culture and decide whether improvements are needed. Environmental enrichment is generally associated with enhancing animal welfare, however, the published literature suggests a cautionary note as not all enrichment procedures have provided a positive benefit. Importantly therefore, before enrichment procedures are implemented, there is a need to evaluate whether the changes made to an animal's environment enhance animal welfare; we must also ensure that such changes do not compromise the scientific purpose for which the animal is being used. The diversity in life history patterns, physiological, behavioural and environmental requirements of the 28,000 or so different fish species are wide ranging and suitable conditions and enrichment for one species is not necessarily transferable to another. These potential differences suggest that a species-specific approach is needed to consider environmental enrichment of captive fish species. Considering how best to proceed, information concerning the natural history of the proposed species and how this compares to the planned captive setting may help to identify specific needs and to determine whether these needs can be met in the laboratory.
Regulatory toxicity test methods are very prescriptive and, in terms of environmental enrichment, limit those procedures that can be applied without compromising the scientific validity of the study. Although husbandry procedures may not be subject to the type of restrictions that apply to testing (e.g. addition of substrates into tanks), implementation of enrichment procedures in husbandry should consider the potential implications for testing in a novel environment, acclimatization and the potential need for de-acclimatization to enrichment techniques before testing. Currently, a limited range of environmental enrichment procedures are successfully employed in both husbandry and test areas at our facility. Further improvements in enrichment, and subsequently fish welfare, will require further research to identify the scientific merit and practical applicability of such procedures in both husbandry and toxicity testing regimes.
Recognizing the limited amount of information on environmental enrichment procedures for aquatic species such as fish, it is hoped that this paper will stimulate a wider debate within the scientific community – agreement or consensus on the key issues concerning enrichment for laboratory-held fish species will help to focus ideas and priorities for further research in this area.
Footnotes
ACKNOWLEDGEMENTS
We are grateful to Dr Grace Panter and Dr Matthew Winter for helpful comments on the manuscript. This article is dedicated to the memory of our friend and colleague Edward Branson, a highly respected fish veterinarian and expert on fish welfare, who died suddenly in 2007.
