Abstract
Schizophrenia is associated with a variety of physical manifestations (i.e. metabolic, neurological) and despite psychotropic medication being blamed for some of these (in particular obesity and diabetes), there is evidence that schizophrenia itself confers an increased risk of physical disease and early death. The observation that schizophrenia and progeroid syndromes share common clinical features and molecular profiles gives rise to the hypothesis that schizophrenia could be conceptualized as a whole body disorder, namely a segmental progeria. Mammalian cells employ the mechanisms of cellular senescence and apoptosis (programmed cell death) as a means to control inevitable DNA damage and cancer. Exacerbation of those processes is associated with accelerated ageing and schizophrenia and this warrants further investigation into possible underlying biological mechanisms, such as epigenetic control of the genome.
Introduction
Of the many different mental disorders, schizophrenia is still one of the most challenging, enigmatic and controversial. Schizophrenia is a relatively new concept, only being recognized as a distinct condition in the 19th century. The concept of schizophrenia since then has been repeatedly revised, expanded, narrowed and re-expanded, reflecting changes in disease models, diagnosis and therapeutics.
Many efforts have been made to describe schizophrenia in behavioural and psychological terms, even though Kraepelin and Bleuler thought it to be ‘endogenous’ and arising from brain pathology. Diagnostic criteria for schizophrenia have been constantly changing, often reflecting efforts to define a condition thought to be biological in the absence of robust physical evidence. More concrete biological criteria to define a diagnosis of schizophrenia would definitely help, but the hunt for a biological marker has proved elusive. Many different biological findings have been statistically associated with schizophrenia, but still there is no pathognomonic feature, and most putative markers to date also appear in the ‘normal’ population, or in other mental disorders.
Why is this the case? Perhaps because most efforts to physically investigate schizophrenia have to date focused only on the CNS. This limited focus however, means that little attention is paid to characteristic non-CNS findings in schizophrenia. We propose that schizophrenia should be regarded as a whole body syndrome rather than as a demarcated brain disease. Furthermore we propose that this syndrome has escaped formal definition thus far because the findings are to a large extent, those which occur in the human body as it ages, although in schizophrenia, they tend to occur earlier. Table 1 formulates our hypothesis.
Formulation of our hypothesis
We have to acknowledge that schizophrenia is a term referring to a variety of clinical presentations and some of those will lack a physical component. Furthermore, schizophrenia might well not have one underlying biological mechanism. Consequently, the concept of schizophrenia being a whole body disorder might apply only to some forms of schizophrenia or even represent an endophenotype of the disorder.
Formulating the hypothesis
An old dilemma in schizophrenia: neurodegeneration vs neurodevelopment
While clinicians and researchers are aware of them, these physical manifestations of schizophrenia in the rest of the body tend to be regarded as either indicative of parallel neurodevelopmental interruption or as complications of the illness and its treatment. However, while there is evidence for neurodevelopmental abnormality in schizophrenia 1 researchers have also recently returned to the idea that the disorder also exhibits elements of neurodegenation. 2 Table 2 summarizes the main arguments of both sides.
Neurodegeneration vs neurodevelopment in schizophrenia
Progeroid syndromes and metabolic syndrome share common features with schizophrenia
Progeroid syndromes are heritable human disorders that display features of premature ageing. They exhibit ‘segmental ageing’ in that only some features of the ageing process are present in each syndrome, leading to the hypothesis that only a few cell lines show an accelerated ageing. Among the most prominent progeroid syndromes, sharing clinical features of premature ageing and a heritable pattern are:
Hutchinson-Guilford Progeria syndrome, caused by mutation in the LMNA gene and presents with lack of hair, large head, stiffness of joints and a senile appearance; Werner syndrome, an adult progeria, caused by mutation in the RECQL2 gene with clinical features of scleroderma-like skin changes, cataracts, subcutaneous calcification, arteriosclerosis and diabetes.
Schizophrenia is likewise a highly heritable condition, leading to a dramatic reduction in life span, largely due to cardiovascular disease.
3
An increased risk of diabetes has been linked to schizophrenia since the 19th century. Henry Maudsley was among the first psychiatrists who noticed an association between diabetes and schizophrenia, clearly in the pre-antipsychotic era. Even today, a significant number of studies have demonstrated that antipsychotic naïve patients have impaired glucose tolerance, increased insulin resistance and increased visceral fat distribution compared to normal controls.
4–6
More importantly, some other studies have shown increased glucose intolerance in the siblings of people with schizophrenia and an increased prevalence of type 2 diabetes in the parents of non-affective psychosis subjects.
7,8
These findings suggest that diabetes and schizophrenia may share familial risk factors or common genetic predisposition.
These observations led a group of American researchers to hypothesize that schizophrenia is a syndrome of accelerated ageing and that physiological abnormalities related to this ageing process can explain its increased mortality rate. 9 Kirkpatrick et al. highlighted the shared risk factors between schizophrenia and progeroid syndromes, such as the advanced paternal age and common prenatal risk factors of prenatal stress, prenatal famine and low birth weight. Kirkpatrick et al.'s own empirical findings are consistent with further cognitive decline in schizophrenia, however they also admitted that psychological tests used in the past have not been sensitive enough to measure this further deterioration in cognitive functions.
Normal ageing appears to have similar but not identical clinical features with schizophrenia
Table 3 summarizes some of the similar but not identical clinical and biochemical features shared between schizophrenia and normal ageing. Viewing schizophrenia as a form of ‘segmental progeria’ leads consequently to a search her physical characteristics in various body areas that are similar between patients with schizophrenia and the normal aged population, not only in the CNS but also in peripheral organs and systems. Ideally those features should be clearly manifested in young and drug naïve patients with schizophrenia as well as in elderly people, suggesting that various cell lines of these patients age faster, and their physiological status, despite their young age, resembles ones of the aged individuals. To keep with our hypothesis, clinical features similar to ageing features, which present in older and chronic patients with schizophrenia, should be more pronounced than expected for their age. Interpretation of the significance of these features however is made more complicated by antipsychotic medication. Undoubtedly, the strongest evidence arises from the study of young and unmedicated, if possible, schizophrenics, however the larger bulk of data come from studies focusing on chronic patients with schizophrenia.
Similarities between schizophrenia and normal ageing
Molecular profiles of schizophrenia vary at different ages, but can still be shared with normal ageing
Another line of evidence comes from molecular biology. Genetic analysis demonstrates a molecular association with the changes in schizophrenia from early to chronic stages reflecting alteration in the nature of the disease as it progresses. 10 Narayan et al.'s analysis revealed variable gene expression profiles and differences in pathway dysfunction at different stages of the illness. There is also a certain degree of overlap between various molecular dysfunctions across patients with short, long and intermediate illness durations. There is also a significant overlap between gene expression associated with normal ageing and that seen in people with early-stage schizophrenia. 11
Some investigators have started to look these links further; newly diagnosed, antipsychotic-naïve patients with non-affective psychosis have both decreased telomere content and an increased pulse pressure. 12 Ageing cells have shortened telomeres, which act as ‘disposable buffers’ and are consumed during cell division, and so telomere erosion reflects cellular ageing to an extent. Would this observation suggest that the cells of psychotic patients age faster? It may not be possible to answer this question yet, but it seems relevant that those with early onset schizophrenia (who are generally of a young age) have molecular profiles which are more similar to those of middle-aged/elderly people ageing normally, than to young people without schizophrenia of a similar age group to themselves.
The above observations are indicative that at a molecular level, there might be similarities between schizophrenia and ageing. However pointing at specific genes only would not be enough to establish a clear link between those conditions. Ageing of the genome is a much complicated process involving a great deal of epigenetic control which goes beyond the alteration of DNA sequence.
Evaluating the hypothesis
Objections to our hypothesis
Despite all the evidence provided above in support of our hypothesis, we should also consider arguments against it. It is known that the great majority of schizophrenics are exposed to antipsychotic medications, which have been linked to diabetes, hyperlipidaemia and a variety of cardiovascular conditions as a result of this. Antipsychotic agents also raise serum prolactin levels, which may lead to reduced bone density and osteoporosis. They also can be sedative and stimulate appetite and this, together with sedentary lifestyles can potentiate obesity and metabolic syndrome. Additionally, lifestyle habits in schizophrenia cannot be ignored. Smoking is much more common among patients with schizophrenia than in the general population and is a well-known risk factor for cardiovascular disease and ageing of the skin.
Some of the features of early ageing in schizophrenia appear to have a clear link to medication. However, the presence of other ageing characteristics, apparently independent of antipsychotic medication, suggests that we need to look more carefully even at findings such as insulin resistance or lens opacities. Besides, a careful observant would point out that there are no identical clinical features shared between schizophrenia and normal ageing, but a rather clinical resemblance of these two conditions.
It appears that following the path of peripheral measures is not sufficient to reveal a clear association between ageing and schizophrenia. To shed more light into this topic, we need to use evidence from the biological sciences, namely molecular biology and genetics.
Evidence from molecular biology
We do not as yet have an underlying biological mechanism to relate schizophrenia to ageing nor is there a uniform biological theory for ageing. An ageing phenome is the sum total of multiple proliferative and degenerative processes which can manifest as disease (i.e. atherogenesis, diabetes, cancer), relatively benign non-disease symptoms (i.e. loss of hair) or symptoms generally recognized as biomarkers of ageing (i.e. changes in T-cell subset, cataracts). 13 Besides, we know that schizophrenia is a highly heritable, probably polygenetic condition while longevity is moderately heritable, with heritability accounting for around one-quarter of variability in life expectancy. 14
At a cellular level ageing is associated with loss of division potential suggesting that normal cells are programmed to undergo senescence, perhaps as a mechanism to compensate for accumulating DNA lesions. The ageing of the genome is far from a complicated process, however DNA damage, in the form of a variety sequential alterations due to endogenous and exogenous factors, is a major factor. Mammalian cells have developed a sophisticated system of detecting and repairing errors during DNA replication and translation, including DNA excision repair, repair of DNA-double strand lesions and others, which all contribute to DNA maintenance. 15 When these attempts to safeguard DNA stability fail, cells recruit two further mechanisms: cellular senescence (mostly in the form of replicative senescence) and apoptosis. Replicative senescence, refers to an arrest in cell division and is caused by telomere shortening, as described above. Additionally, if cells are no longer needed, they commit ‘suicide’ by activating an intracellular death program, known as apoptosis, or programmed cell death. Compared to senescence and to necrosis (acute cellular death due to an external factor), apoptosis is a very clean process, by which cells are eliminated quietly by phagocytosis, without the occurrence of an inflammatory cascade. At a molecular level, apoptosis is mediated by an intracellular proteolytic cascade, delivered by proteases called caspases, and a number of genes are involved, including tumour suppressor genes Mdm2, p53 and p21 (GenBank accession nos. 164785, 191170, 116899).
Excessive apoptosis is thought to play a role in the development of schizophrenia, with excess CNS neuronal loss resulting in reduced level of function and psychotic symptoms. Apoptosis in schizophrenia may be mediated by excessive glutamergic activity. The NMDA receptor hypo-activity hypothesis of schizophrenia postulates that structures other than the cortex provide glutamergic input to the striato-thalamic filter and to dopaminergic neurons. Reduced activity at NMDA receptors is caused by various factors, including genetic, developmental and environmental (mimicked by PCP). Their hypo-activity causes a reduction in GABAergic inhibitory tone onto glutamergic neurons in the thalamus and cortex. Hyper-activity of the later is implicated in the induction of psychotic states. 16 At a cellular level, glutamergic hyper-activity results in an increased influx of Ca++ ions into the neuron through the NMDA Ca channel. Increased intracellular levels of Ca cause excitotoxicity, resulting in the release of free radicals and eventually in an indiscriminate destruction of various neuronal parts and neuronal death. 17 What is remarkable is that even in dermal fibroblast cell lines, schizophrenia patients show increased apoptosis. 18 The p53 gene, known to suppress tumours and with a major role in promoting apoptosis, has been investigated as a candidate susceptibility gene in schizophrenia 19 with some early positive results. 20
So if apoptosis and senescence are associated with both ageing and schizophrenia, could this be the link that brings these two syndromes together? Apoptosis could also account for another observation in schizophrenia: the decreased incidence of various cancers, not only in patients themselves but also in their parents and siblings. 21 Remarkably this applies even to lung cancer, even after adjusting for smoking prevalence. Of course this observation does not apply to every kind of cancer and we know for instance that breast cancer presents with an increased incidence in female patients with schizophrenia, which again could be aetiologically associated with reduced parity and hyper-prolactinaemia. 22 But even this kind of discrepancies could be explained by considering that the proposed ageing process has a segmental presence. Cancer is primarily a disease of genomic instability, most likely due to accumulating mutations. Cells that become cancerous have lost their susceptibility to apoptosis and multiply incessantly. This was already described as a case of antagonistic pleiotropy: both apoptosis and senescence help to suppress cancer at an early stage, possibly at the cost of promoting ageing at later ages, by exhausting progenitor or stem-cell reservoirs. 23 As body tissues age there is a shift from mitotic cell activity to a senescent cellular state, and this might be the cost of a defence mechanism which initially developed to target tumour formation (Figure 1). Perhaps in schizophrenia, the reduced rates of some cancers are explained by an increased apoptotic process in the affected cellular lines. This could provide an explanation for the epidemiological observation that schizophrenia appears to be antagonistic towards some kind of cancers.
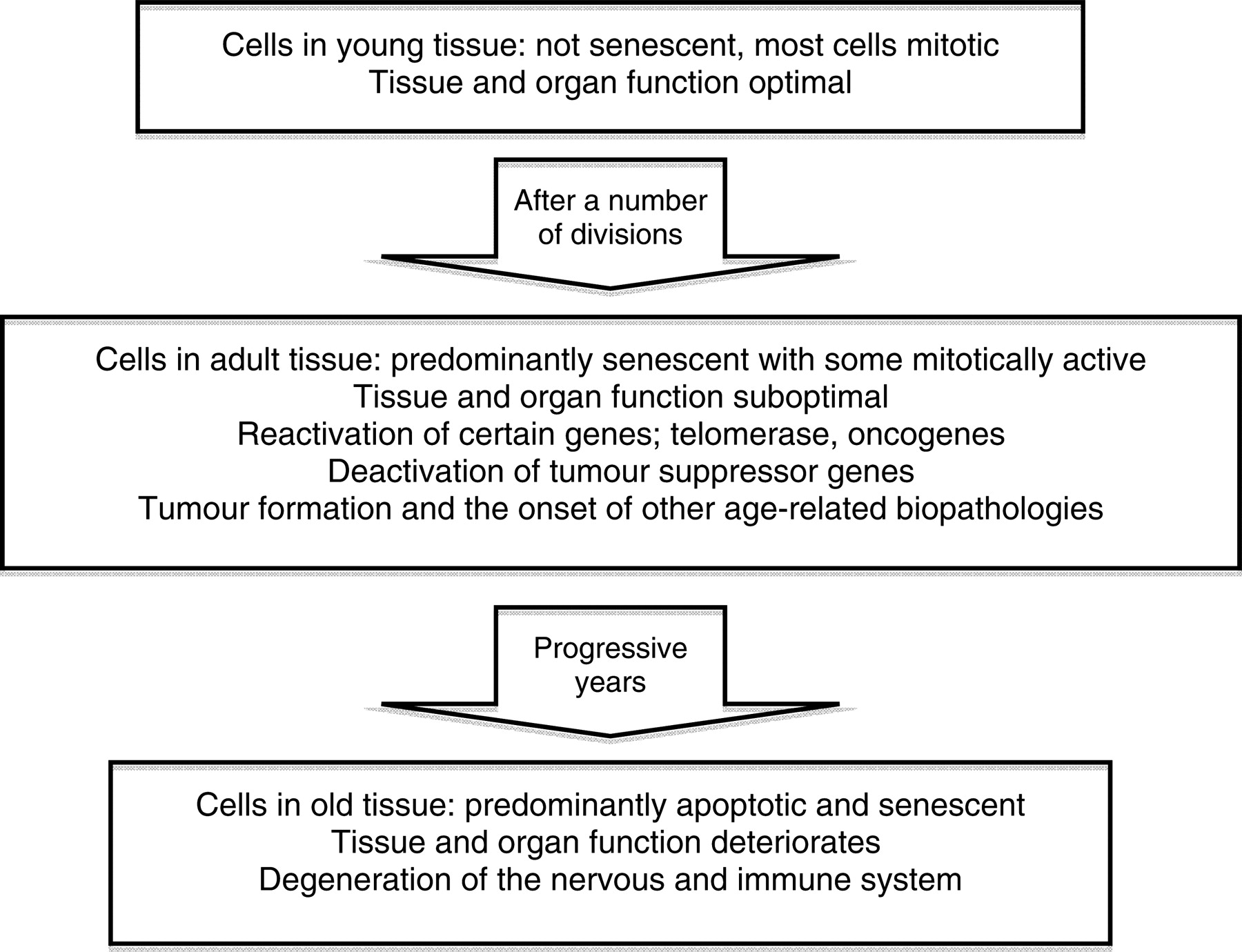
Cellular and tissue changes occurring with the onset of ageing. An internal genetic clock causes the cells to switch from mitotically active to slow proliferating cells as seen in adult tissue, with predominant silent but metabolically active senescent cells (adapted from: Saldanha SN, McCollum A, Tollefsbol TO. Environmental Effects on Age-Associated Epigenetics. In: Tollesfsbol TO, ed. Epigenetics of Aging. New York, NY: Springer, 2010:417–432)
From genetics to epigenetics
The study of epigenetics, the collective heritable and reversible changes in phenotype due to processes that arise independent of primary DNA sequence, has evolved to the new science of genetics, and has attracted a special interest with regards to cancer and ageing. DNA replication is subject to epigenetic control via various mechanisms, such as the control of chromatin condensation through histone modifcation, 24 or telomere formation through telomerase activity. 25 If DNA replication ceases, cells enter the state of senescence, which leads to ageing, on the other hand if DNA replication becomes uncontrollable, cancer occurs. Epigenetic factors can disrupt this fine balance and divert cells towards one or the other end. A number of epigenetic processes have been described and are summarized in Table 4.
Major epigenetic processes
Most of the above mentioned processes have been implicated in ageing, however among the most widely encountered findings are:
26
A significant number of methylation errors, mainly stochastic in nature, resulting in genome wide hypo-methylation, gene-specific hypermethylation and aberrant methylation metabolism; Changes in high order chromatin structure, associated with chromatin condensation such as:
histone modification; changes in HAT:HDAC balance (Histone acetyltransferases/Histond deacetylases); changes in sirtuins expression (class III HDACs); telomere control, shortening of telomeres; other changes, including those in telomeric repeat-containing RNA (TERRA), ncRNAs, PcG genes and Wnt pathway.
Epigenetic changes have also been reported in schizophrenia, especially in prefrontal cortex, where abnormal DNA or histone methylation at sites of specific genes and promoters is associated with changes in RNA expression. Those changes are remarkably found not only in postmortem brains of patients with schizophrenia, but also in peripheral blood cells, as for instance the finding of global hypomethylation of peripheral leukocyte DNA in male patients with schizophrenia, which was even more prominent in younger patients.
27–29
So, current evidence suggests the presence of similar epigenetic changes between ageing and schizophrenia, such as widespread DNA hypomethylation. There is also increasing evidence that schizophrenia is also associated with an aberrant epigenetic profile and abnormal GABAergic neurotransmission in cortical areas. The list of affective genes includes reelin, GAD67, SOX10 and COMT, primarily involved in neurodevelopment and neurotransmission.
30
If evidence for our hypothesis has to be sought, there is no better place to look at than the epigenetics area. If we show that schizophrenia comprises similar epigenetic changes with normal ageing or age-related physical conditions (conditions which are the outcome of cellular senescence), then we can provide initial evidence that a process of accelerated aging is part of the aetiological mechanisms of schizophrenia. Such an approach should target not only genes associated with the neurodevelopmental or neurodegenerative correlates of schizophrenia, but the total of the human genome.
Conclusion
In this article we tried to present some of the data supporting our hypothesis, which unifies both mental and physical manifestations under the diagnosis of schizophrenia. We have to acknowledge that schizophrenia is a term referring to a variety of clinical presentations and some of those will lack a physical component. Consequently, the concept of schizophrenia being a whole body disorder might apply only to some forms of schizophrenia or even represent an endophenotype of the disorder.
There may be other molecular processes, which explain the findings in schizophrenia. The first step is to consolidate the clinical picture by bringing together wide-ranging clinical studies and perform a cluster analysis of symptoms. Then this information could by used to target laboratory research. Another approach would be to combine clinical and laboratory findings and try to discover any possible correlations between those.
Identifying biomarkers as fundamental features of schizophrenia is of great significance: not only it would render the diagnosis more reliable and valid, but it would also emphasize the importance of taking care of the overall physical health of patients with schizophrenia, thus contributing to improvements in their quality of life. As an aid or component of diagnosis, the concept of whole body disorder would underline the biological character of schizophrenia, inspire more certitude and confidence about the essence of schizophrenia and possibly stimulate more somatic-focused research. As a practical orientation, it would contribute towards the abolition of the philosophical consideration of body/mind dualism and encourage practising psychiatrists to take into account the physical and mental complaints of their patients at an equal basis.
In this short review we tried to conceptualize schizophrenia as a whole body disorder, by using the paradigm of segmental progeria. We acknowledge that the evidence provided is not conclusive, and more research is needed to support this hypothesis. We suggest that the study of epigenetic processes appears the most promising towards this aim.
DECLARATIONS
Competing interests
None declared
Funding
None
Ethical approval
Not applicable
Guarantor
EP
Contributorship
All authors contributed equally
Acknowledgements
None
