Abstract
Objectives
To summarize participation and coverage rates in population mammographic screening programmes for breast cancer in Europe.
Methods
We used the European Network for Information on Cancer (EUNICE), a web-based data warehouse (EUNICE Breast Cancer Screening Monitoring, EBCSM) for breast cancer screening, to obtain information on programme characteristics, coverage and participation from its initial application in 10 national and 16 regional programmes in 18 European countries.
Results
The total population targeted by the screening programme services covered in the report comprised 26.9 million women predominantly aged 50–69. Most of the collected data relates to 2005, 2006 and/or 2007. The average participation rate across all programmes was 53.4% (range 19.4–88.9% of personally invited); or 66.4% excluding Poland, a large programme that initiated personal invitations in 2007. Thirteen of the 26 programmes achieved the European Union benchmark of acceptable participation (>70%), nine achieved the desirable level (>75%). Despite considerable invitation coverage across all programmes (79.3%, range 50.9–115.2%) only 48.2% (range 28.4–92.1%) of the target population were actually screened. The overall invitation and examination coverage excluding Poland was 70.9% and 50.3%, respectively.
Conclusions
The results demonstrate the feasibility of European-wide screening monitoring using the EBCSM data warehouse, although further efforts to refine the system and to harmonize standards and data collection practices will be required, to fully integrate all European countries. The more than three-fold difference in the examination coverage should be taken into account in the evaluation of service screening programmes.
Introduction
In Europe, most programmes for breast cancer screening have developed their own screening information systems for running day-to-day operations, managing quality, monitoring and evaluating services, and for preparing information for organizations such as local government or ministries, with no explicit priority on promoting an exchange of information between programmes in different countries.
The European Network for Information on Cancer (EUNICE) was a project co-funded by the European Union. A key aim of the project was to produce a monitoring tool capable of calculating a selection of key performance parameters and early impact indicators from the European Guidelines, which could be used to compare screening programmes across Europe on a regular basis. The user-friendly tool facilitates monitoring of screening activity in a standardized format. It enables the uniform, automatic calculation of pre-defined indicators for benchmarking and for comparison between programmes.
This paper describes the design of the tool, with selected programme characteristics, coverage and participation obtained from its initial application in 10 national and 16 regional breast cancer screening programmes in 18 European countries.
Methods
In 2007 a web-based data warehouse (EUNICE Breast cancer screening monitoring, EBCSM) was developed for collection of aggregated data on implementation and performance of breast cancer screening programmes in Europe (www.qtweb.it/eunice).
The database is accessible to authorized users only. It allows data uploading and verification, calculation of screening indicators with standardized algorithms and formats, and comparison between programmes and with benchmarks. The parameters and indicators are shown for the entire age-range and for 5-year age groups. They are generated in the following eight modules:
Coverage and participation rates Number of mammograms performed Further assessment, including needle biopsies performed (fine needle aspirations and core biopsies) Outcome of further assessment (e.g. number of referrals to surgery) Outcome of surgical referral (e.g. number of cancers, benign lesions, ductal carcinoma in situ) Number of invasive cancers detected, total and by (TNM) stage Size of invasive cancers (1–10 11–20, >20mm) Type of surgery for invasive cancer (number of breast-conserving surgeries, mastectomies)
Each module has two sections, one for routine indicators generated from a minimum set of parameters (standard), and one for optional, more differentiated indicators based on additional parameters (extended). For example, the standard section of the ‘participation and coverage’ module generates the participation rate by age, whereas the extended version shows the participation rate by age separately for women who were invited to attend screening for the first time.
The online data collection instrument also has a general section, with a questionnaire format, that includes items on programme characteristics, such as the policy on the number of mammographic views or double reading of screening mammograms. The questionnaire and the operational definition of the indicators, as well as a documentation manual (www.qtweb.it/eunice), were prepared by the Eunice Working Group, based on the fourth edition of the European Guidelines. 1 , 2 Representatives of breast cancer screening programmes from all 27 European Member States plus Norway and Switzerland were invited to join the Group. Two pan-European meetings (Brno, Czech Republic, 2006 and Budapest, Hungary, 2008) were organized to agree the design of the data warehouse, study procedures and data collection.
A survey was then conducted using the EBCSM. The previously identified reference persons from these 29 European countries were asked to provide aggregated data describing service screening activity in the reference year 2005, and supplemental information on programme characteristics in the reference year 2007. Completion of all standard sections was requested, plus the extended sections, where feasible. Checks for internal consistency and completeness were performed on the data received, and detected errors in classification or data entry were corrected. Missing data were reported to participants and completed where possible.
The main outcome measures we report here are coverage and participation, and key organizational and policy characteristics of the programmes. Coverage is defined as the extent to which the screening programme covers the eligible population within the appropriate interval in a given period by invitation (invitation coverage) and the extent to which the screening programme covers the eligible population with screening tests (examination coverage). In practice, coverage has been calculated as the annual number of invitations (or tests) divided by the annual target population, which in turn is represented by the total target population divided by the screening interval in years. Participation is defined as the proportion of women attending screening of those personally invited.
To provide a more detailed picture of the organization of screening services, the extent of invitations and tests performed per screening unit and mammography machine in 2007 were estimated, using data from programme organization in 2007 and the programme performance in earlier years (in most cases 2005 and/or 2006).
Results
Eighteen of the 29 European countries provided aggregated data and information on programme characteristics (Figure 1). National data was provided by 10 countries: Czech Republic, Estonia, Finland, Hungary, Italy, Luxembourg, Norway, Poland, the Netherlands and the United Kingdom (Figure 1). The eight other countries provided data limited to 16 regional programmes: Belgium (Flanders), Denmark (Copenhagen), Germany (pilot projects), Portugal (Centre and North), Republic of Ireland (East), Spain (Asturias, Baleares, Galicia, Navarra, Pais Vasco, Valencia), Sweden (Södermanland, Stockholm, Västmanland) and Switzerland (Fribourg). The results are presented for 26 national or regional programmes. Although the UK also provided national data, we include here only those related to England, as these data are more complete. Performance data from the reference year 2005 were provided by 24 programmes, 10 of which included data from one or two additional reference years. The data from one programme (Germany, pilot projects) referred to the years 2001–2004. The data from Poland referred only to the year 2007.
Countries represented in EUNICE Breast Cancer Screening Monitoring survey by type of data provided. National data (black): Czech Republic, Estonia, Finland, Hungary, Italy, Luxemburg, Norway, Poland, The Netherlands, United Kingdom; Regional data (grey): Belgium (Flanders), Denmark (Copenhagen), Germany (pilot projects), Portugal (North, Centre) Republic of Ireland (East), Spain (Asturias, Baleares, Galicia, Navarra, Pais Vasco, Valencia), Sweden (Södermanland, Stockholm, Västmanland), Switzerland (Fribourg), Regional and national data: Hungary, Italy, United Kingdom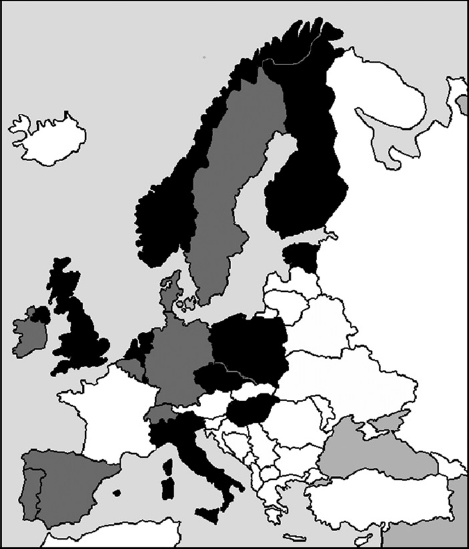
Policies and organization
Basic information on the programmes is shown in Table 1. Most programmes began in the late 1980s or early 1990s. Exceptions were Belgium (Flanders), Czech Republic, Estonia, Germany (pilot projects in Bremen, Weser-Ems and Wiesbaden), Hungary, Poland, Republic of Ireland and Switzerland (Fribourg), which started more recently.
Breast cancer screening programme features by country or region in 26 European programmes (2007)
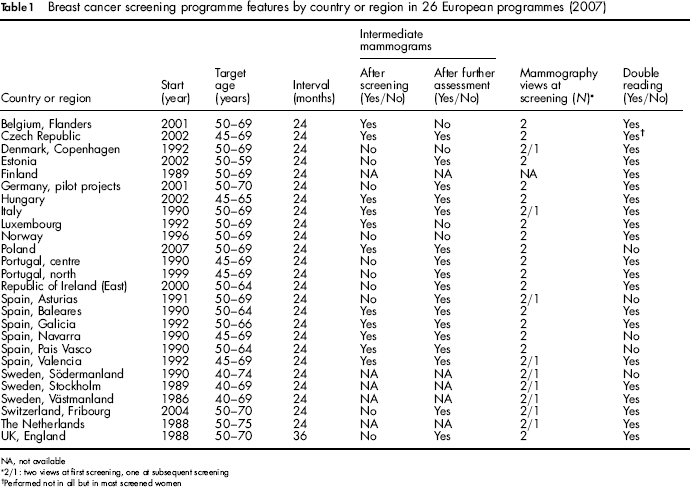
NA, not available
2/1: two views at first screening, one at subsequent screening
Performed not in all but in most screened women
Women were targeted from age 50 in 17 programmes, while a lower target age was applied in nine programmes. The target age specified in the European Union policy on cancer screening 3 (50–69 years) was adopted in eight programmes, though three others used 50–70 years. In addition to age, gender and geographical area, some programmes applied other eligibility criteria, such as exclusion of women with previous breast lesions, previous mastectomies, breast implants, pregnancy or terminal illness. All eligible women received an individual invitation letter, except in the Czech Republic, where women were referred by general practitioners or gynaecologists. Personal invitations were introduced in the Czech Republic on a pilot basis in 2007.
All programmes reported the use of two-view mammography for the initial screening examination; nine programmes used only a single-view at subsequent screening for all or selected groups of women (one missing value). Screening mammograms were read by two independent radiologists in all but five programmes. Mammography was the only screening test performed in 25 programmes. In Hungary, clinical breast examination (BCE) was also used. The screening interval was two years in all programmes except for the United Kingdom (England) where the maximum interval was three years.
In all but two programmes, further assessment was performed on recall. In Spain (Valencia) and the Czech Republic it was possible to perform assessment on the same day as the screening examination. With the exception of the Netherlands, women were recalled for further assessment in units dedicated full or part-time to the screening programme.
Table 2 presents estimates of the annual number of screening tests and the average annual numbers of tests per screening unit, and per mammography machine in 2007 based on the reported programme characteristics in 2007 and volume of tests reported by most programmes in earlier years, assuming that there was no change in volume over time. For most programmes, performance data were provided for the years 2005 and/or 2006. On average, 19 programmes performed less than 10,000 tests per screening unit per year. Ten programmes performed less than 5,000 screening mammograms per machine per year.
Breast cancer screening facilities and estimated annual volume of activity in 26 European programmes (2007)
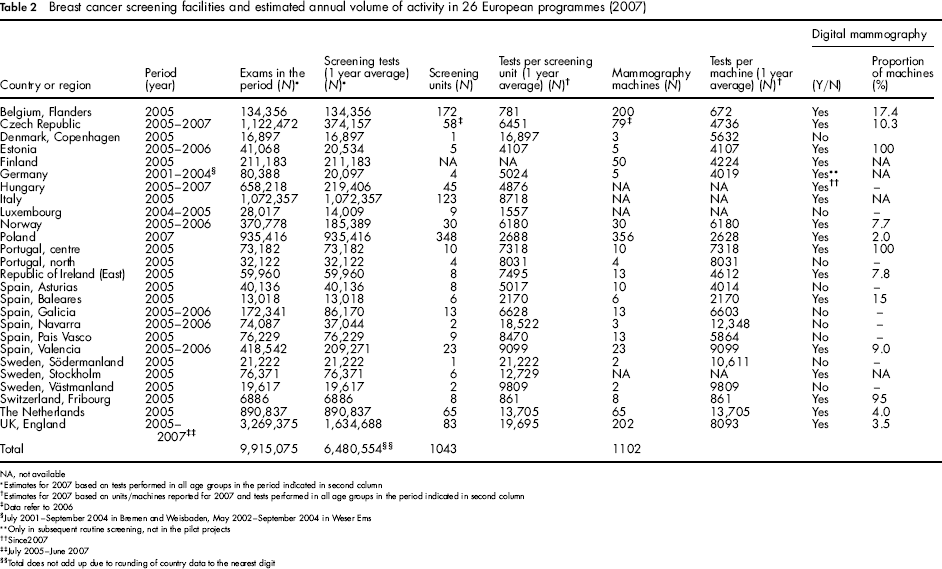
NA, not available
Estimates for 2007 based on tests performed in all age groups in the period indicated in second column
Estimates for 2007 based on units/machines reported for 2007 and tests performed in all age groups in the period indicated in second column
Data refer to 2006
July 2001 – September 2004 in Bremen and Weisbaden, May 2002-September 2004 in Weser Ems
Only in subsequent routine screening, not in the pilot projects
Since 2007
July 2005-June 2007
Total does not add up due to rounding of country data to the nearest digit
Table 2 also shows the availability of full-field digital mammography (FFDM) in 2007. In the 16 programmes using digital mammography, only two relatively small programmes (Estonia and Switzerland, Fribourg) were equipped essentially only with FFDM machines (100% and 95% respectively). In the other programmes FFDM accounted for less than 20% of mammography machines.
Information on breast cancer screening data management and monitoring in 2007 is presented in Table 3, including website addresses from which further information and reports can be obtained. Regional and national monitoring was implemented for 16 programmes in nine countries. Four programmes in three countries used only regional monitoring systems. Monitoring was established only at the national level in five programmes. Most of the programmes used either individual (n = 8) or mixed individual and aggregated data (n = 14) for monitoring; three programmes used only aggregated data.
Breast cancer screening data managing and monitoring in 26 European programmes (2007)
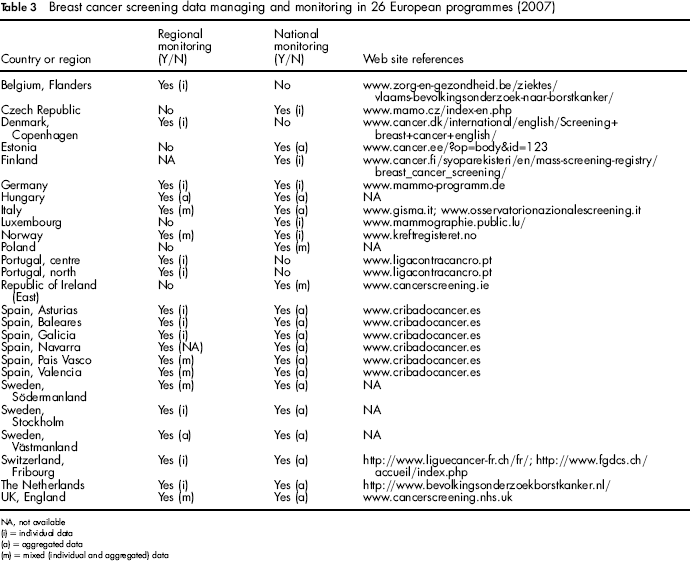
NA, not available
(i) = individual data
(a) = aggregated data
(m) = mixed (individual and aggregated) data
Coverage and participation
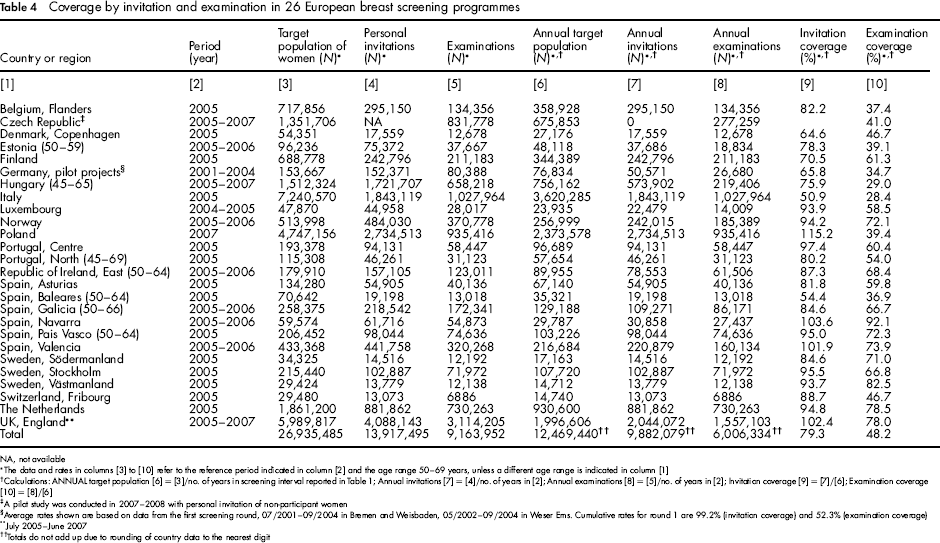
All 26 programmes completed the standard section of the coverage and participation module. The results shown for most programmes refer to the age group 50–69 years (Table 4). The total number of women in the target population of the 26 programmes used to calculate coverage rates was 26.9 million, and the total number of invitations used to calculate coverage by invitation in the 26 programmes was 13.9 million.
Coverage by invitation and examination in 26 European breast screening programmes
NA, not available
The data and rates in columns [3] to [10] refer to the reference period indicated in column [2] and the age range 50–69 years, unless a different age range is indicated in column [1]
Calculations: ANNUAL target population [6] = [3]/no. of years in screening interval reported in Table 1; Annual invitations [7] = [4]/no. of years in [2]; Annual examinations [8] = [5]/no. of years in [2]; Invitation coverage [9] = [7]/[6]; Examination coverage [10] = [8]/[6]
A pilot study was conducted in 2007–2008 with personal invitation of non-participant women
Average rates shown are based on data from the first screening round, 07/2001–09/2004 in Bremen and Weisbaden, 05/2002–09/2004 in Weser Ems. Cumulative rates for round 1 are 99.2% (invitation coverage) and 52.3% (examination coverage)
July 2005-June 2007
Totals do not add up due to rounding of country data to the nearest digit
The total number of screening tests in predominantly 50–69-year-old women reported in the 26 programmes and shown in Table 4 (9.16 million) was 7.6% less than the total number performed in women of all ages (9.92 million, Table 2). About 20% of the tests reported in the study were for initial (prevalent) screening, with substantial respective volumes of initial screening reported for the Czech Republic, Estonia and Poland (data not shown).
Coverage by invitation
The coverage by invitation ranged from 50.9% in Italy, to 115.2% in Poland, the latter exceeding 100% as more than 50% of the target population were invited in a single year (i.e. exceeding 100% coverage for a two-year programme) (Table 4). The invitation coverage in Poland was inflated in 2007 due to the initiation of personal invitation in the screening programme in that year. The overall coverage by invitation across all 26 programmes was 79.3%. Excluding Poland, the overall coverage by invitation was 70.9%.
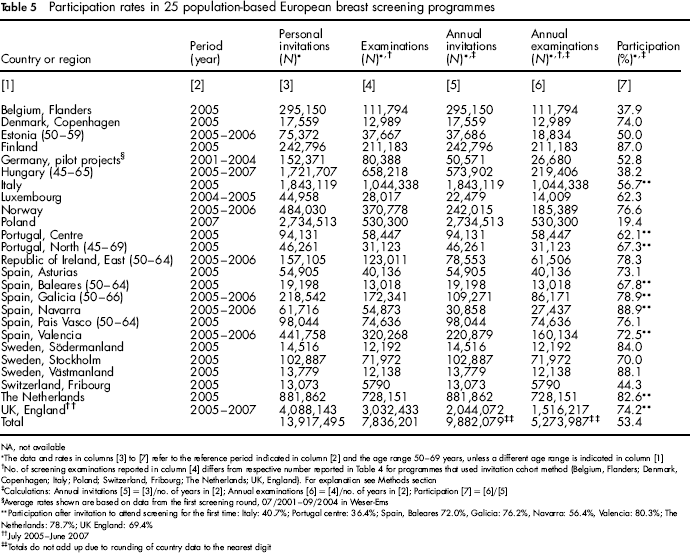
Participation rates
The participation rate varied from 19.4% in Poland to 88.9% in Navarra (Spain) (Table 5). Half of the programmes (13 out of 26) achieved the acceptable level of participation recommended in the European Union Guidelines (>70%). Nine programmes achieved the higher desirable level specified in the European Union Guidelines (>75%). None of the six programmes that started more recently met the acceptable European Union target; in one of these (Czech Republic) the participation rate was not reported because women access the programme without a personal invitation letter.
Participation rates in 25 population-based European breast screening programmes
NA, not available
The data and rates in columns [3] to [7] refer to the reference period indicated in column [2] and the age range 50–69 years, unless a different age range is indicated in column [1]
No. of screening examinations reported in column [4] differs from respective number reported in Table 4 for programmes that used invitation cohort method (Belgium, Flanders; Denmark, Copenhagen; Italy; Poland; Switzerland, Fribourg; The Netherlands; UK, England). For explanation see Methods section
Calculations: Annual invitations [5] = [3]/no. of years in [2]; Annual examinations [6] = [4]/no. of years in [2]; Participation [7] = [6]/[5]
Average rates shown are based on data from the first screening round, 07/2001–09/2004 in Weser-Ems
Participation after invitation to attend screening for the first time: Italy: 40.7%; Portugal centre: 36.4%; Spain, Baleares 72.0%, Galicia: 76.2%, Navarra: 56.4%, Valencia: 80.3%; The Netherlands: 78.7%; UK England: 69.4%
July 2005-June 2007
Totals do not add up due to rounding of country data to the nearest digit
The participation rate calculated across all 25 programmes sending personal invitations was 53.4% (Table 5). Excluding Poland, the average participation rate was 66.4%.
Except for two regions in Spain (Baleares and Valencia), the programmes that provided data permitting separate calculation of participation after invitation to attend screening for the first time revealed lower participation rates for initial screening compared with the overall participation rate (initial and subsequent screening combined). The differences ranged from 3 to 33 percentage points (see Table 5, footnote).
Coverage by examination
The coverage by examination ranged from 28.4% in Italy to 92.1% in Navarra, Spain (Table 4). The overall coverage by examination calculated across all 26 programmes was 48.2%.
Discussion
A limited number of publications have presented data on the characteristics and performance of breast cancer screening programmes across Europe and internationally.4–10 These reports have been instrumental in demonstrating the need for uniform standards of reporting to improve the exchange of information and experience between programmes. The fourth edition of the European Guidelines for quality assurance in breast cancer screening and diagnosis recommends a comprehensive set of performance parameters and indicators for monitoring and evaluating any population-based breast cancer screening programme, but does not provide a means of routinely collecting the requisite data and uniformly generating indicators. The EUNICE data warehouse addresses this important need. This report demonstrates the feasibility of the EBCSM module on coverage and participation, and provides an overview of programme organization in 26 screening programmes in Europe. The results are relevant to the current discussion of the impact of breast cancer service screening programmes, and to the current preparations to update the first report on the implementation of cancer screening programmes in the European Union. 11
All of the programmes that participated in this survey are involved in the European Cancer Network (ECN), into which the former European Cancer Screening networks have been consolidated. In addition to collaboration in EUNICE, key projects in the ECN and the former European Union cancer screening networks have been the development and updating of the European Guidelines for quality assurance in breast, cervical and colorectal cancer screening and reporting on the implementation of cancer screening programmes in the European Union.1,2,11–14
Although 10 of the 26 European Union Member States with breast screening programmes are not represented in the current survey, the present results are consistent with the findings in the first report on implementation of cancer screening programmes in the European Union. This applies, for example, to the wide consensus in the European Union that breast cancer screening should be conducted in organized, population-based programmes, with personal invitations to each individual in the target population. The results presented here also show that despite wide agreement in Europe on additional policy aspects recommended by the Council of the European Union, such as the screening test (mammography), the target age range (50–69 years) and the screening interval (two years), there are still potentially significant differences in the way screening programmes are organized, particularly with regard to the volume and concentration of services and the size of target populations and screening programmes.
Professionals require a sufficient volume of tests to develop and maintain specialized skills in screening. The larger the testing volume of a screening unit, the shorter the time that will be required to accumulate sufficient data to reliably determine performance indicators, such as the rates of referral to surgery and detection of breast cancer, or the benign-to-malignant biopsy ratio. Delays in detecting potential problems necessarily also delay the time until corrective action can be taken. The present survey reveals a high, 27-fold variation between programmes in the estimated yearly number of examinations per screening unit, and a 20-fold variation in the estimated number of tests performed per mammography machine. This wide variation suggests that programmes with lower unit volumes may require additional efforts and resources to achieve and maintain appropriate quality.
Very large programmes, with millions of eligible participants, must also ensure the same high level of quality across a large number of screening units and mammography machines. Significant, sustainable resources for uniform, timely reporting of appropriate performance parameters and indicators are essential to reliably and promptly detect differences between screening units that may require further investigation and action.
Decision-makers, programme coordinators and scientists should be aware of the substantial differences in Europe in the extent to which target populations are actually exposed to screening. There is a nearly two-fold difference in the invitation coverage across the 25 programmes included in the survey that routinely sent personal invitations. Furthermore, there is a more than three-fold difference in the examination coverage in the 26 programmes included in the survey (Table 4). The low examination coverage in some programmes may be attributed, to a large extent, to the exclusion criteria in the case of the Hungarian programme (women with a mammogram in the previous 24 months were not eligible to attend), and the incomplete rollout of the very large screening programme in Italy and the pilot projects in Germany during the respective reference periods.
Low examination coverage should not, however, be misinterpreted as a reason to interrupt screening activities of appropriate quality, particularly in the rollout phase of programmes, because potentially high coverage in some regions will be masked by little or no coverage in regions that have not yet initiated or completed rollout. In general, the measureable impact of screening on a target population should be greater in a programme with higher examination coverage. In practice, the relationship between examination coverage and impact may not be proportional, but the importance of the degree to which a target population is actually exposed to the intended screening examination should not be overlooked when evaluating the impact of screening. The lower the examination coverage, the more difficult it will be to distinguish the impact of screening from other trends affecting the burden of breast cancer in the population, particularly when methods of analysis are used that do not distinguish carefully between those women who are exposed to the screening test, and those who are not.
Given the importance of maximizing the benefit of screening, while minimizing the negative effects, professionals responsible for the implementation of breast cancer screening programmes should make every reasonable effort to ensure that the screening examination is available to all eligible women. As pointed out in the European Guidelines, effective communication is crucial to the overall success of these activities. 1 , 2 Even if 90% of the target population is invited to screening, and the participation rate reaches the ‘acceptable ‘ target recommended in the European Union Guidelines (>70%), 3–4 out of 10 women will not have the mammographic examination during a given round of screening. Careful attention should therefore be paid not only to effective communication, enabling women to make an informed choice about attending screening, but also to technical and administrative aspects which ensure that all eligible women are reliably invited.
The present results show less variation between programmes in invitation coverage than in participation rates. The pronounced differences in participation rates underline the fact that the areas and target populations served by programmes may differ substantially with regard to the healthcare environment and the characteristics of the target population. Breast cancer awareness among women and the extent of opportunistic screening can strongly affect participation in population-based screening programmes. Low participation rates should also stimulate careful examination of organizational procedures. For example they may result from preselection of the invited population to only include women with previous tests or invitations. The comparatively low participation rates (<60%) in more recently established breast screening programmes are consistent with previous experience in the ECN, particularly in the initial rounds of programmes in areas with significant opportunistic screening activity. The potential impact of opportunistic screening on participation rates is also quite relevant in some Italian areas, especially in younger women. 15 Facilitating the switch to organized screening when eligible women seek an appointment for mammography outside the programme can help to improve participation. A point which is often overlooked is that organized programmes are usually subject to rigorous quality assurance, whereas opportunistic screening activities may not be. This illustrates the importance of informing general practitioners and office-based gynaecologists and radiologists about the programme, and involving them in communication with women. 1 , 2
While this overview of European breast screening programmes provides a useful snapshot of key aspects relevant to monitoring and evaluation, there are also limitations. Data were collected predominantly for the years 2005–2007, but significant changes in policies or performance of some programmes may have occurred subsequently. The use of digital technology, for example, which is now established in a number of programmes, was not yet widespread. The information provided in the report is derived from aggregated data. Though aggregated data permit some useful conclusions they are limited regarding the depth of analysis.
Conclusions
The feasibility of a web-based data warehouse (EBCSM) for standardized data collection, analysis and benchmarking of screening programmes in different countries has been demonstrated. The quality of data collection, and the validity and reliability of the information generated with the EBCSM database, is likely to improve if this resource is used on a regular basis to monitor regional and national programme performance, and to compare results between countries. The results presented here show a substantial difference in the extent to which eligible women are offered and participate in screening in European programmes; this should be taken into account when evaluating the impact of screening.
EUNICE WORKING GROUP
The following authors were responsible for data collection from participating screening programmes or for data verification:
Belgium: Van Hal G 1 , Martens P 2 ; Czech Republic: Majek O 3 , Danes J 4 ; Denmark: von Euler-Chelpin M 5 ; Estonia: Aasmaa A 6 ; Finland: Anttila A 7 ; Germany: Becker N 8 ; Hungary: Pentek Z 9 , Budai A 9 ;Madai S 10 ; Ireland: Fitzpatrick P,16 Mooney T16; Italy: Zappa M 11 , Ventura L 11 ; Luxembourg: Scharpantgen A 12 ; Norway: Hofvind S 13 ; Poland: Seroczynski P 14 ; Portugal: Morais A 15 , Rodrigues V 15 , Bento M J 15 , Gomes de Carvalho J 15 †; Spain: Natal C17, Prieto M17,Sánchez-Contador Escudero C18, Zubizarreta Alberti R19, Fernández Llanes S B19, Ascunce N20, Ederra Sanza M20, Sarriugarte Irigoien G21, Salas Trejo D22; Ibáñez Cabanell J22; Sweden: Wiege M23; Ohlsson G23, Tornberg S24, Korzeniewska M25; Switzerland: de Wolf C26; The Netherlands: Fracheboud J27; United Kingdom: Patnick J28, Lancucki L28; IARC: Ducarroz S.29, Suonio E.29
† Dr J Gomes de Carvalho now deceased
Affiliations:
1 Voorzitter Consortium, Centrum voor Kankerpreventie Antwerpen, Belgium
2 Vzw Vroegtijdige Opsporing van Borstklierkanker, Brugge, Belgium
3 Institute of Biostatistics and Analyses, Masaryk University, Brno, Czech Republic
4 Department of Radiology, First Faculty of Medicine, Charles University in Prague, Czech Republic
5 Centre for Epidemiology and Screening, University of Copenhagen, Denmark
6 Epidemiology and Biostatistics Department-National Institute for Health Development, Tallinn, Estonia
7 Mass Screening Registry and Finnish Cancer Registry, Helsinki, Finland
8 German Cancer Research Center, Division of Cancer Epidemiology, Heidelberg, Germany
9 National Mammography Screening Evaluation Committee (NMSEC), Budapest, Hungary
10 MaMMa Healthcare Co., Budapest, Hungary
11 National Centre of Screening Monitoring, Florence, Italy
12 Programme Mammographic, Direction de la Sante, Luxembourg
13 The Cancer Registry of Norway, Oslo, Norway
14 Asseco Poland S.A.
15 Liga Portuguesa Contra o Cancro, Lisbon, Portugal
16BreastCheck, National Cancer Screening Service, Dublin, Ireland
17Subdirección de Gestion Clínica y Calidad. Servicio de Salud del Principado de Asturias, Spain
18Dirección General de Salud Pública y Participation. Conselleria de Salut i Consum, Baleares, Spain
19Dirección General de Innovación y Gestión de la Salud Publica, Consellería de Sanidade, Galicia, Spain
20Instituto de Salud Publica, Breast Cancer Screening Program, Navarra, Spain
21Programa Detección Precoz Cáncer de Mama, Direccion Territorial de Sanidad de Bizkaia, Bilbao, Spain
22General Directorate Research and Public Health & Centre for Public Health Research (CSISP), Valencia, Spain
23Mälarsjukhuset, Eskilstuna, Sweden
24Cancer Screening Unit, Stockholm, Sweden
25Brostenheten Vásteras Central Hospital, Västmanland, Sweden
26Swiss federation of breast cancer screening, Bern, Switzerland
27Department of Public Health, NETB, Erasmus University Medical Centre, Rotterdam, The Netherlands
28NHS Cancer Screening Programmes, Sheffield, UK
29Quality Assurance Group, Early Detection and Prevention Section, International Agency for Research on Cancer, Lyon, France
Footnotes
Acknowledgements
Financial support for this publication was provided by the Italian ‘National Centre for Screening Monitoring’ (![]() ) within the EUROSCREEN Project. The EUNICE breast cancer screening survey was financially supported by the European Commission (grant agreements No. 2004114, European Network for Information on Cancer, and No. 2006322, European Cancer Network). Financial support from the European Union Public Health Programme (project no. 2006322, European Cooperation on Development and Implementation of Cancer Screening and Prevention Guidelines [ECCG-ECN]) was also provided for the preparation of this report. Many thanks to Roberta Castagno and Denise Casella (CPO Piemonte) for their assistance during the project and the preparation of this report. The authors thank Tracy Lignini (IARC) for assistance in the preparation of the manuscript. A special acknowledgment to Adam Svobodnik, formerly at Masarick University, Brno (Czech Republic), for providing great support to the project and hosting, with Sylvia Madai, MaMMa Healthcare, Budapest (Hungary), the project meetings. Finally, thanks to Ahti Anttila for his encouragement and for contributing to refining and dissemination of survey methods and results within the Eurocourse project.
) within the EUROSCREEN Project. The EUNICE breast cancer screening survey was financially supported by the European Commission (grant agreements No. 2004114, European Network for Information on Cancer, and No. 2006322, European Cancer Network). Financial support from the European Union Public Health Programme (project no. 2006322, European Cooperation on Development and Implementation of Cancer Screening and Prevention Guidelines [ECCG-ECN]) was also provided for the preparation of this report. Many thanks to Roberta Castagno and Denise Casella (CPO Piemonte) for their assistance during the project and the preparation of this report. The authors thank Tracy Lignini (IARC) for assistance in the preparation of the manuscript. A special acknowledgment to Adam Svobodnik, formerly at Masarick University, Brno (Czech Republic), for providing great support to the project and hosting, with Sylvia Madai, MaMMa Healthcare, Budapest (Hungary), the project meetings. Finally, thanks to Ahti Anttila for his encouragement and for contributing to refining and dissemination of survey methods and results within the Eurocourse project.
