Abstract
Objectives
To estimate the impact of service mammography screening on breast cancer mortality using European incidence-based mortality (IBM) studies (or refined mortality studies). IBM studies include only breast cancer deaths occurring in women with breast cancer diagnosed after their first invitation to screening.
Methods
We conducted a literature review and identified 20 publications based on IBM studies. They were classified according to the method used for estimating the expected breast cancer mortality in the absence of screening: (1) women not yet invited; (2) historical data from the same region as well as from historical and current data from a region without screening; and (3) historical comparison group combined with data for non-participants.
Results
The estimated effect of mammography screening on breast cancer mortality varied across studies. The relative risks were 0.76–0.81 in group 1; 0.75–0.90 in group 2; and 0.52–0.89 in group 3. Study databases overlapped in both Swedish and Finnish studies, adjustment for lead time was not optimal in all studies, and some studies had other methodological limitations. There was less variability in the relative risks after allowing for the methodological shortcomings.
Conclusions
Based on evidence from the most methodologically sound IBM studies, the most likely impact of European service mammography screening programmes was a breast cancer mortality reduction of 26% (95% confidence interval 13–36%) among women invited for screening and followed up for 6–11 years.
Introduction
Mammography screening has been implemented in many European countries, based on the results of the randomized controlled trials. 1 However, the effect of screening on breast cancer mortality might differ between clinical trials and routine service screening programme where, for example, screening may be undertaken by initially less experienced personnel and there are differences between the populations screened. It is therefore important that the outcome of screening in routine health care, usually referred to as service screening, is monitored. 2 We aimed to provide an overview of IBM studies of service mammography screening in Europe.
Methods
We defined IBM studies as studies including only breast cancer deaths occurring in women targeted for screening, with breast cancer diagnosed after their first invitation to screening. We restricted our analysis to studies covering at least some of the age groups 50–69.
We conducted a literature review of service mammography screening and breast cancer mortality using PubMed (Appendix A). From 5009 abstracts, 122 were considered relevant by a documentalist (C Bellisario, CPO Torino) and an epidemiologist (P Armaroli, CPO Torino). Criteria for inclusion in the review were: (a) the study provided original data on a population-based screening programme in Europe; (b) a study outcome was breast cancer mortality; (c) the analysis included at least some of the age groups between 50 and 69; and (d) the study used IBM. In addition to the literature search, the Working Group added publications fulfilling the inclusion criteria but not identified by the search and new publications that became available after March 2011 (n = 4). Twenty publications were identified in total,3–22 one from Denmark, seven from Finland, two from Italy, one from Norway, one from Spain and eight from Sweden.
Four studies reported on data already used in other studies. Chen et al. 21 used some of the data reported by the Swedish Organised Service Screening Evaluation Group (SOSSEG), 18 and reported similar results, while the study by Baker et al. 22 used data presented by Duffy et al. 16 also with similar results. We therefore only considered the studies by SOSSEG and Duffy et al. 16 , 18 The data reported by Parvinen et al. 13 were used also by Wu et al. 20 and the resulting estimates were similar. We therefore included only Parvinen et al. 13 The two studies by Paci et al. 5 , 6 reported on the same data, but there was a difference in accrual versus follow-up period in Paci et al. 5 which might have caused a bias in that analysis. Therefore we only considered Paci et al. 6 in our review.
There was some overlap in the study populations in Finland and in Sweden. In Finland, approximately 50% of the study population in Hakama et al. 3 overlapped with Parvinen et al. 13 but in the latter study, the population was followed for nine years longer. In the 2002 Anttila et al. 4 study, 40% of the study population was also included in Hakama et al. 3 but followed up for five years longer in Anttila et al. In Sarkeala et al. 8 50% of the study population was also included in another article by the same researchers 9 and vice versa, both with follow up until the end of 2003. Most of the study population in Anttila et al. 19 was included in Sarkeala et al. 9 An overlap similar of that seen in Finland was found for the six Swedish publications, with the 13-area study 18 giving the best coverage of the Swedish data.
Statistical analysis
To evaluate the impact of screening, data for a non-screened comparison group are needed. For this purpose data from a geographic and/or historical comparison group are typically used. To avoid selection bias, we used invitation to screening instead of participation in screening as the primary exposure. With this intention-to-screen approach, the incidence-based breast cancer mortality in a population targeted by screening was compared with the incidence-based breast cancer mortality expected in the absence of screening, to produce the relative risk (RR) and 95% confidence interval (CI).
Estimation of expected breast cancer mortality in absence of screening
A key issue for each study was how the breast cancer mortality expected in the absence of screening was estimated. This had been done in three different ways:
Expected breast cancer mortality estimated from a cohort of women not yet invited to screening; Expected breast cancer mortality estimated from historical data from the same region as well as from historical and present data from a region without screening; Expected breast cancer mortality estimated from historical data from the same region, and if deemed necessary adjusted for changes in breast cancer mortality over time, based on changes in breast cancer mortality in non-participants.
By design, the study by Sarkeala et al. 8 belonged to group 3. However, when a comparison was made between women screened aged 50–69 with women screened at age 50–59 it was possible to include this study in group 2 as well. Part of Kalager et al. 10 had a very short and unbalanced follow-up period (with regards to calendar year). To report results for a long and balanced follow-up period, we used data available in the appendix of the article to reanalyse this study, 10 dividing the data into areas with long and balanced follow-up (area I versus areas V and VI) and areas with short and unbalanced follow-up (areas II-VI versus areas I-IV).
Adjustment for lead time
Breast cancer cases are diagnosed earlier in screened women than in non-screened women, as shown in the Figure 1. In studies where the follow-up period continued after the end of the accrual period (the period in which breast cancer cases are identified), more cases and thereby more breast cancer deaths will be included in the study group compared with the control group. The reduction in breast cancer mortality due to screening will therefore be underestimated. Adjustment for this often overlooked lead time bias was undertaken in most of these studies. Studies which include breast cancer cases for a certain age group (e.g. 50–69 years) and also breast cancer deaths for older age groups (e.g. 50–74) will similarly underestimate the reduction in breast cancer mortality due to this form of bias.
Accrual and follow-up periods and possible consequences for counting of screen-detected cases
When the most reliable studies were identified, a pooled estimate based on these studies was calculated using a fixed effects weighted average of the RRs on a logarithmic scale. 23
Results
Of the 20 publications, 16 were considered in the analysis,3,4,6–19 with re-analysis of two. 8 , 10 All of the studies focused on measuring the effect of an organized screening programme, that is, a screening service with a well-defined target population contacted by personal invitation.
Of the three studies in group 1, where expected breast cancer mortality was estimated from women not yet invited (Table 1), two were from Finland 3 , 4 and one from Italy. 6 In Hakama et al. 3 selected birth cohorts were invited before others to allow a subsequent comparison of death rates, and in this study all women were followed up individually. All three showed a reduction in breast cancer mortality, with RRs ranging from 0.76 to 0.81, each at borderline statistical significance. No adjustment for lead time was made in the Italian study.
Incidence-based breast cancer mortality studies with expected mortality estimated from women not yet invited
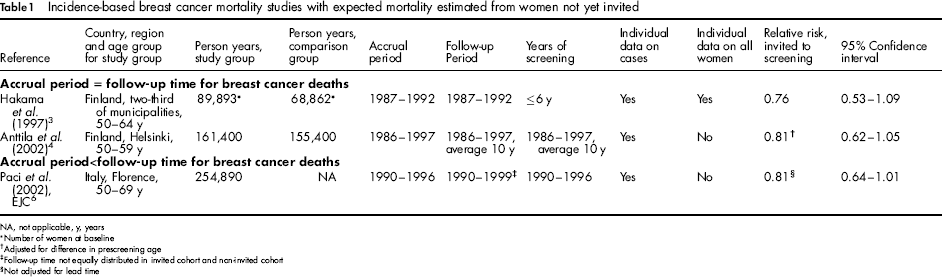
NA, not applicable, y, years
Number of women at baseline
Adjusted for difference in prescreening age
Follow-up time not equally distributed in invited cohort and non-invited cohort
Not adjusted for lead time
In total seven studies, including our re-analysis of Sarkeala et al. 8 were in group 2, where the expected breast cancer mortality was estimated from regional and historical comparison groups (Table 2). Three came from Sweden11,12,14and provided estimates based on both underlying cause of death and excess mortality. In order to provide comparable results, we only reported the estimates based on underlying cause of death, because this was the endpoint reported in the other studies. Two studies were from Finland, 8 , 13 and one each from Denmark 7 and Norway. 10 Individual follow-up of all women was undertaken in the Danish study only, 7 whereas the numbers of person-years at risk were estimated from population statistics in the other studies. The two studies 7 , 8 where no adjustment for lead time was necessary produced RRs of breast cancer mortality of 0.69 and 0.75. Breast cancer mortality was significantly decreased in the Danish study, RR = 0.75 (95% CI 0.63–0.89). 7 In the studies where the accrual period was shorter than the follow-up period,10–14 the RRs not adjusted for lead time ranged from 0.75 to 0.90. In the Kalager study 10 data from breast cancers diagnosed at age 50–69 and breast cancer deaths at age 50–79 showed a RR of 0.88 (95% CI 0.73–1.05). Dividing the data into areas with long and more balanced follow-up (area I versus areas V and VI) produced a RR of 0.81 (95% CI 0.62–1.05) whereas data from areas with short and unbalanced follow-up (areas II-VI versus areas I-IV) gave a RR of 0.99 (95% CI 0.71-1.37).
Incidence-based breast cancer mortality studies with expected mortality estimated from regional and historical comparison group(s)
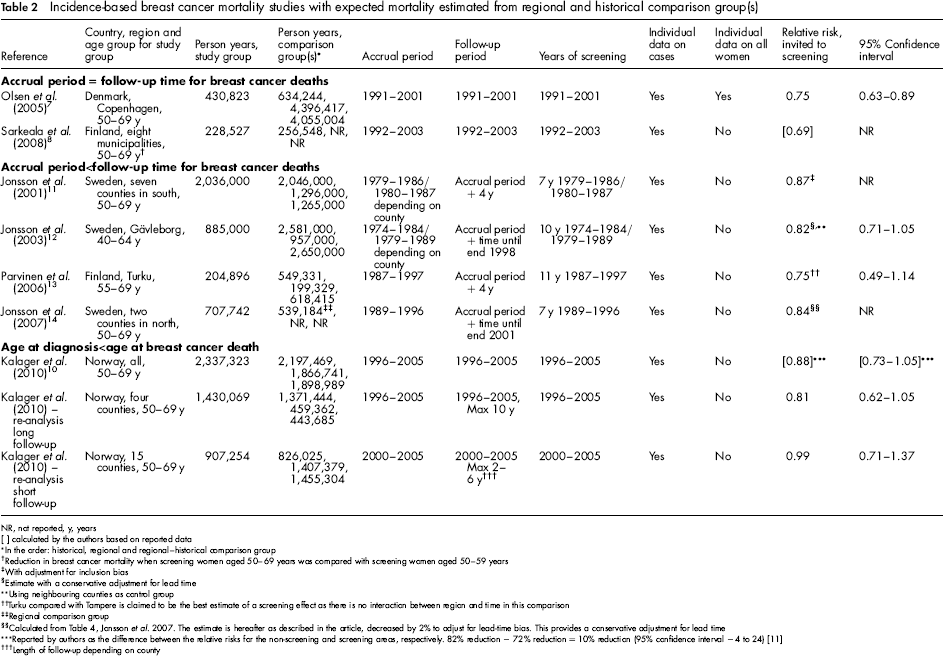
NR, not reported, y, years
[] calculated by the authors based on reported data
In the order: historical, regional and regional-historical comparison group
Reduction in breast cancer mortality when screening women aged 50–69 years was compared with screening women aged 50–59 years
With adjustment for inclusion bias
Estimate with a conservative adjustment for lead time
Using neighbouring counties as control group
Turku compared with Tampere is claimed to be the best estimate of a screening effect as there is no interaction between region and time in this comparison
Regional comparison group
Calculated from Table 4, Jonsson et al. 2007. The estimate is hereafter as described in the article, decreased by 2% to adjust for lead-time bias. This provides a conservative adjustment for lead time
Reported by authors as the difference between the relative risks for the non-screening and screening areas, respectively. 82% reduction -72% reduction = 10% reduction (95% confidence interval -4 to 24) [11]
Length of follow-up depending on county
There were seven studies in group 3, where the expected breast cancer mortality was estimated from a historical comparison group subsequently combined with data for non-participants. Three studies were from Sweden,15,16,18 three from Finland8,9,19 and one from Spain 17 (Table 3). In this group, all RRs were statistically significant ranging from 0.52 to 0.82. One study had an accrual period shorter than the follow-up period and used breast cancer cases from one age group and breast cancer deaths from an age group including older women. 19 This study reported a RR of 0.89.
Incidence-based breast cancer mortality studies with expected mortality estimated from historical comparison group and non-participants *
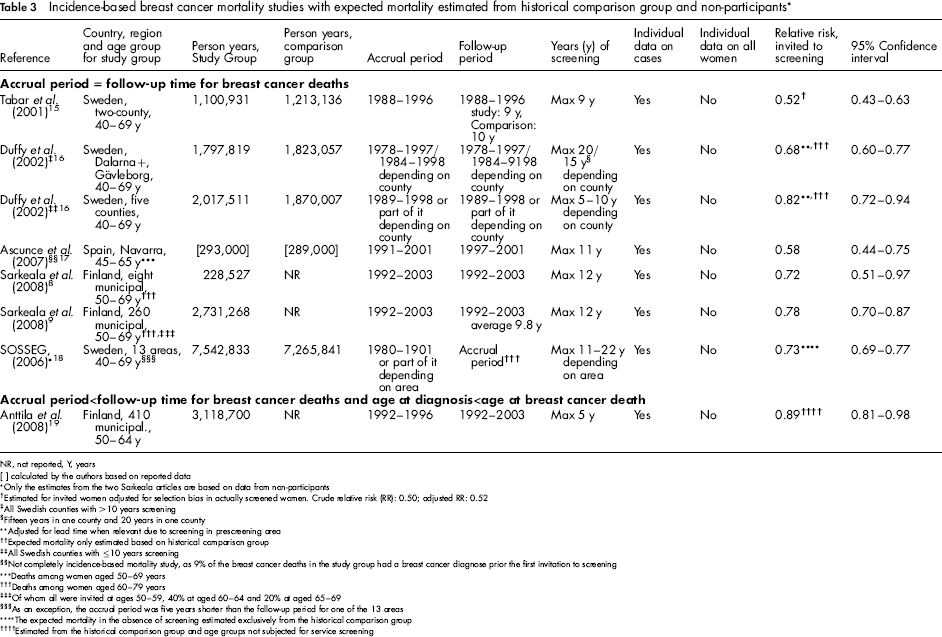
NR, not reported, Y, years
[] calculated by the authors based on reported data
Only the estimates from the two Sarkeala articles are based on data from non-participants
Estimated for invited women adjusted for selection bias in actually screened women. Crude relative risk (RR): 0.50; adjusted RR: 0.52
All Swedish counties with >10 years screening
Fifteen years in one county and 20 years in one county
Adjusted for lead time when relevant due to screening in prescreening area
††Expected mortality only estimated based on historical comparison group
All Swedish counties with ≤10 years screening
Not completely incidence-based mortality study, as 9% of the breast cancer deaths in the study group had a breast cancer diagnose prior the first invitation to screening
Deaths among women aged 50–69 years
Deaths among women aged 60–79 years
Of whom all were invited at ages 50–59, 40% at aged 60–64 and 20% at aged 65–69
As an exception, the accrual period was five years shorter than the follow-up period for one of the 13 areas
The expected mortality in the absence of screening estimated exclusively from the historical comparison group
Estimated from the historical comparison group and age groups not subjected for service screening
The studies with the most unbiased comparisons are those able to control for changes over time without introducing a healthy user bias and where the accrual period is similar to the follow-up time for breast cancer deaths (those in the top half of Tables 1 and 2). The most reliable estimate of the breast cancer mortality reduction among women invited to screening is, therefore, based on these studies, i.e. Hakama et al. 3 Anttila et al. 4 Olsen et al. 7 and our re-analysis of Sarkeala et al. 8 Due to overlap between Hakama et al. 3 and Anttila et al. 4 both should not be included in a pooled estimate. As Hakama et al. 3 included most of the age group 50–69, we chose to include this study in the pooled estimate. Our best estimate of the breast cancer mortality reduction among women invited to screening, based on Hakama et al. 3 Olsen et al. 7 and our re-analysis of Sarkeala et al. 8 was a combined RR estimate of 0.74 (95%CI 0.64–0.87). In Hakama et al. 3 the participation rate was 88% in the first round, in Sarkeala et al. 8 the proportion of person-years among participants out of all person-years in the cohort screened at age 50–69 was 85%, whereas in Olsen et al. 7 the participation rate in the first round was 71%. As women included in these three studies were followed for 6–11 years, this is a reliable estimate of the breast cancer mortality reduction after 6–11 years of follow-up for women offered screening at age 50–69.
Discussion
The effect of mammography screening on breast cancer mortality differed between the studies. The RRs were 0.76 -0.81 in group 1; 0.75 -0.90 in group 2 and 0.52 to 0.89 in group 3. However, there was less variability in the RRs after allowing for the methodological limitations of the studies.
A 25% statistical significant reduction in breast cancer mortality was found in the single study from Denmark. 7 It included three comparison groups, incorporating both historical control within the same region and geographic control from both the historical and the screening periods. All data were based on individuals. A similar method was used in the single study from Norway, 10 although with person years at risk based on routine statistics rather than individual follow-up. In the latter study, results were reported based on breast cancer cases diagnosed at age 50–69 and subsequent breast cancer deaths among these cases. Due to lead time, the mortality reduction due to screening would be underestimated. Overall, this study showed only a 12% statistically non-significant reduction in breast cancer mortality. There was, however, a considerable difference between counties within Norway. In counties with long and calendar year balanced observations, there was a 19% reduction in breast cancer deaths, whereas no material impact of screening was seen in counties with shorter and calendar year unbalanced follow up. The single study from Spain 17 did not allow for a possible non-screening related underlying trend because only a historical comparison group was included. This might partly explain the large reduction of 42%. A 19% reduction in breast cancer mortality was observed in the Italian study, 6 where the follow-up period was three years longer than the accrual period and where no adjustment for lead time was undertaken.
Of the overlapping Finnish studies, Hakama et al. 3 was the most methodologically robust, because the comparison group was most unlikely to be biased. However, the data covered the period 1987–1992, and concern has been raised about the relevance of older data for evaluating the potential impact of mammography screening in more recent times. Anttila et al. 19 was the largest study but it had an accrual period shorter than the follow-up and used different age groups for breast cancer diagnoses and breast cancer deaths. The Sarkeala et al. studies 8 , 9 were the most recent and only had minor overlap with the Hakama et al. Sarkeala et al. 9 only used a historical comparison group, making it difficult to distinguish between a mortality reduction due to screening and other temporal changes, such as improvements in treatment. In order to use both a regional control group and a historical control group, we made a separate comparison between the data for women offered screening at ages 50–69 with those offered screening only at ages 50–59, assuming that the methodology for estimating the expected numbers in the absence of screening had been the same for the two groups. Using this approach we found a 31% reduction in breast cancer mortality among women offered screening at age 50–69 compared with women offered screening at age 50–59. This was calculated by dividing the RR for screening at 50–69, by that for screening at 50–59 in Table 2 of Sarkeala, 8 giving an RR of 0.69. One might reasonably expect that the reduction in breast cancer mortality among women offered screening at age 50–69 compared with women not offered screening at all would be at least 31%, consistent with the 25% reduction reported by Parvinen et al. 13
Of the overlapping Swedish studies, the SOSSEG study covering 13 Swedish areas was the most comprehensive. 18 It showed a 27% highly statistically significant reduction in breast cancer mortality. However, it would have been useful if allowance were made for the underlying trend in the absence of screening, as in some other Swedish studies.11,12,14 All of these studies did, however, have an accrual period shorter than the follow-up period, and the adjustment used for lead time bias was probably conservative.
There are several potential limitations to IBM studies. The incidence-based breast cancer deaths were in all studies identified from linkages between screening registers, cancer registers and cause of death registers. Data on person years at risk should in principle also be derived from a linkage of incidence and population data, as only women at risk of breast cancer at the time of first invitation to screening can contribute person years at risk. The person years at risk were, however, estimated based on routine statistics data in all but the Hakama et al. 3 and Olsen et al. 7 studies. This should be taken into account in the interpretation of the results, as the outcome of this type of study can be sensitive to relatively minor compromises in the methodology. 24
Another limitation is that although the studies by Jonsson et al.11,12,14 used both regional and historical comparison groups, we did not emphasize these results in the summary due to concerns that the lead time adjustments might be too small. Furthermore, breast cancer screening in Europe is normally undertaken in organized programmes. In the reported studies, the estimated breast cancer mortality in the absence of screening represented the breast cancer mortality in the absence of an organized programme. Only limited opportunistic screening took place in countries and/or regions where organized breast cancer screening started early. This was the case in Denmark, Spain, Finland and Sweden. In Norway, however, organized screening started only in 1996, and opportunistic screening was widespread before this point in time. 25 This may have affected the Norwegian data even in the counties with a relatively long follow up and balanced data.
CONCLUSION
Although data from observational studies should be interpreted with caution, the outcomes of the reported IBM studies were compatible when differences in methodology and local circumstances were taken into account. Based on evidence from the most reliable studies, a breast cancer mortality reduction of 26% after 6–11 years of follow-up seems to be the likely impact of the European service mammography screening programmes offered to women aged 50–69.
EUROSCREEN WORKING GROUP
Coordinators:
Members:
Ancelle-Park R (F) 1 , Armaroli P (I) 2 , Ascunce N (E) 3 , Bisanti L (I) 4 , Bellisario C (I) 2 , Broeders M (NL) 5 , Cogo C (I) 6 ,De Koning H (NL) 7 , Duffy SW (UK) 8 , Frigerio A (I) 2 , Giordano L (I) 2 , Hofvind S (N) 9 , Jonsson H (S) 10 , Lynge E (DK) 11 , Massat N (UK) 8 , Miccinesi G (I) 12 , Moss S (UK) 8 , Naldoni C (I) 13 , Njor S (DK) 11 , Nystrom L (S) 14 , Paap E (NL) 5 , Paci E (I) 12 , Patnick J (UK) 15 , Ponti A (I) 2 , Puliti D (I) 12 , Segnan N (I) 2 , Von Karsa L (D) 16 ,Tornberg S (S) 17 , Zappa M (I) 12 , Zorzi M (I) 6
Affiliations:
1
Ministère du travail de l'emploi et de la santé, Paris, France
2
CPO-Piedmont, Turin, Italy
3
Navarra Breast Cancer Screening Programme, Pamplona, Spain
4
S.C. Epidemiologia, ASL di Milano, Italy
5
Radboud University Nijmegen Medical Centre & National Expert and Training Centre for Breast Cancer Screening, Nijmegen, The Netherlands
6
Veneto Tumor Registry, Padua, Italy
7
Erasmus MC, Dept. of Public Health Rotterdam, The Netherlands
8
Wolfson Institute of Preventive Medicine, Queen Mary University of London, UK
9
Cancer Registry of Norway, Research Department and Oslo and Akershus University College of Applied Science, Oslo, Norway
10
Department of Radiation Sciences, Oncology, Umeå University, Umeå, Sweden
11
Centre for Epidemiology and Screening, Universtiy of Copenhagen, Denmark
12
ISPO Cancer Research and Prevention Institute, Florence, Italy
13
Regional Cancer Screening Center, Emilia-Romagna Region, Bologna, Italy
14
Department of Public Health and Clinical Medicine, Division of Epidemiology and Global Health, Umeå University, Umeå, Sweden
15
NHS Cancer Screening Programmes and Oxford University, UK
16
International Agency for Research on Cancer, Lyon, France
17
Stockholm Cancer Screening, Stockholm, Sweden
Footnotes
Acknowledgements
Financial support was provided by the National Monitoring Italian Centre (ONS) to host the EUROSCREEN meetings in Florence in November 2010 and in March 2011 and the supplement publication and the National Expert and Training Centre for Breast Cancer Screening, Nijmegen, The Netherlands to host a meeting of the EUROSCREEN mortality working group in July 2011.
Search Strategy
We performed the following search strategies on Pubmed:
(‘Breast Neoplasms/mortality*’[MeSH Terms] OR breast cancer mortality OR ‘Mortality’[MeSH Terms]) AND (‘mass screening’[MeSH Terms] OR screening) AND (‘mammography’[MeSH Terms] OR mammography) The search strategy retrieved a total of 2432 papers. After inspection of titles and abstract, according to the criteria's described in the article, we judged 98 papers as relevant. (effect* OR evaluation OR impact OR trend) AND (service screening OR programme screening OR mass screening) AND breast cancer AND (mortality OR survival) The search strategy retrieved a total of 1649 papers. After inspection of titles and abstract, according to the criteria's described in the article, we judged 16 papers as relevant. ‘Breast Neoplasms/mortality*’[MeSH Terms] AND ‘mass screening’[MeSH Terms] AND (‘Mortality/trends’[MeSH Terms] OR ‘Survival Analysis’[MeSH Terms] OR ‘Survival Rate/trends’[MeSH Terms]) The search strategy retrieved a total of 187 papers. After inspection of titles and abstract, according to the criteria's described in the article, we judged two papers as relevant. Function ‘Related articles’ of this model study Otten JDM, Broeders MJM, Fracheboud J, Otto SJ, de Koning HJ, Verbeek ALM. Impressive time-related influence of the Dutch screening programme on breast cancer incidence and mortality, 1975–2006. Int J Cancer 2008;
A total of 741 papers were retrieved. After inspection of titles and abstract, according to the criteria's described in the article, we judged eight papers as relevant.
