Abstract
Objective
Analysing trends in population breast cancer mortality statistics appears a simple method of estimating the effectiveness of mammographic screening programmes. We reviewed such studies of population-based screening in Europe to assess their value.
Methods
A literature review identified 17 papers, of which 12 provided quantitative estimates of the impact of screening. Due to differences in comparisons and outcome measures, no pooled estimate of effectiveness was calculated.
Results
Comparisons included breast cancer mortality before and after the introduction of screening, trends in early and late starting areas and trends in age groups affected and unaffected by screening. Studies that calculated the percentage annual change after the start of screening found reductions of 1–9% per year (1%, 2.3–2.8% and 9% for those with adequate follow-up). Of studies that compared mortality in time periods before and after introduction of screening, three single country studies all had adequate follow-up and estimated mortality reductions ranging from 28% to 36%. Limitations of studies of population mortality rates include the inability to exclude deaths in women with breast cancer diagnosed before invitation to screening, diluting any observable impact of screening, and the gradual implementation of screening in a country or region.
Conclusions
Although analysing population breast cancer mortality rates over time can be a first step in examining changes following the introduction of screening, this method is of limited value for assessment of screening impact. Other methods and individual data are necessary to properly quantify the effect.
Introduction
Analysis of the trends in age-specific or age-standardized population mortality rates from breast cancer over time appear intuitively to be an attractively simple means of evaluating population-based service screening. However, for a number of reasons it is difficult to use such analyses to quantify the effectiveness of screening. Nevertheless the relative simplicity of an approach studying trends in population mortality rates has led to a large number of such studies. The aim of this paper is to review and summarize these studies from European countries, and to discuss their limitations.
Methods
We included as trend studies those that reported on trends in breast cancer mortality rates in a population as a whole in relation to the introduction and/or extent of population-based mammographic screening. Such studies can be, and usually are, based on aggregated data obtained from routine sources.
The search strategy (described in the Appendix to Broeders et al. 1 ) identified a total of 35 papers, of which three were excluded as the full paper was not available in English, four as they included only a time period before or shortly (less than three years) after the introduction of screening, three because they studied the effect of screening as a result of RCTs or pilot studies, two because they did not attempt to study the impact of mammographic screening, and two as the country or region studied did not have an organized screening programme. Five papers were excluded because they used non-mortality-based approaches such as simulation modelling, survival analysis or surrogate outcome measures, and two were effectively incidence-based mortality analyses.
We included three further papers which were not identified by the literature search or were published after it had been performed. We classified the resulting 17 papers into those that were only descriptions of the trend over time in breast cancer mortality in relation to the timing of the introduction of organized screening (Table 1), and those that included a more detailed analysis with the aim of quantifying the impact of screening on mortality (Table 2).
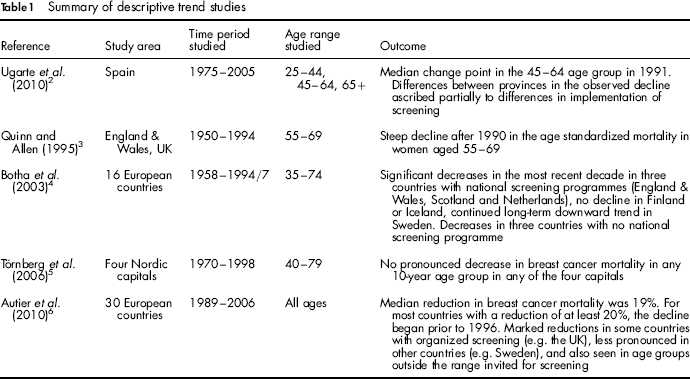
Summary of descriptive trend studies
Summary of trend studies that give a quantified estimate of the effect of screening
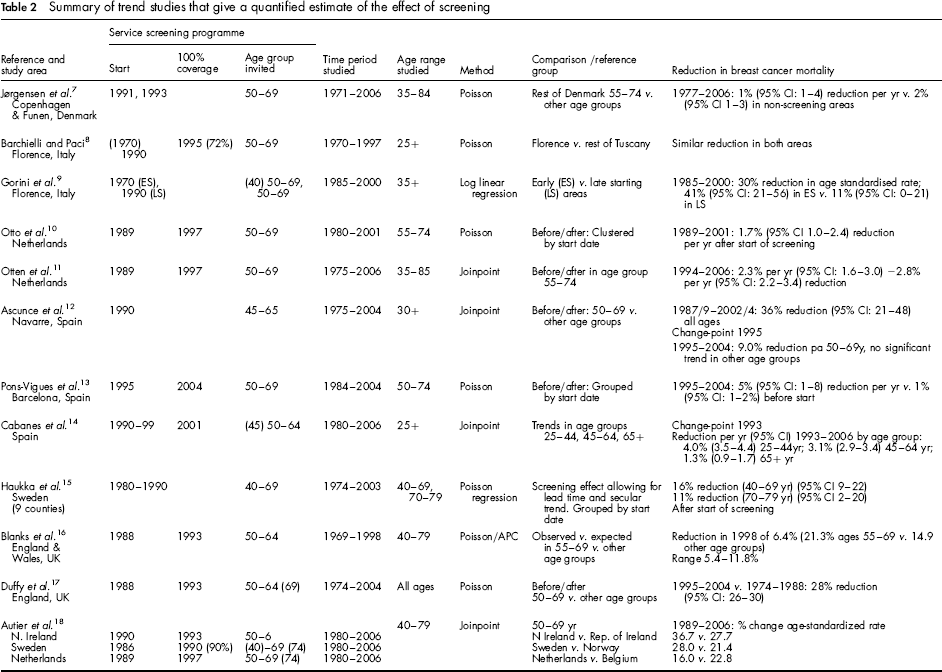
The latter category included 12 studies, which we classified according to country, time period and method of analysis (Table 2). The methods of analysis included Poisson regression (with or without age cohort modelling), and the use of joinpoint regression to identify ‘break points’ at which changes in mortality trends occurred.
We classified those studies with follow-up of 10 years or more after complete coverage of the target population by invitation as having adequate follow-up time in the screening period to observe the full effect of screening.
Comparisons included breast cancer mortality before and after the introduction of screening, trends in early and late starting areas and the trends in age groups affected and unaffected by screening. Outcomes were generally presented either as a percentage reduction in a time period after introduction of screening compared with a prescreening period, or as a percentage annual change after the start of screening. Due to differences in methodology, comparisons and outcome measures used in the studies, no attempt was made to produce a pooled estimate of the effect of screening.
Results
Descriptive studies
Of the five descriptive papers, two compared the breast cancer mortality trends in different regions or countries, and related these to differences in screening coverage/activity. 4 , 6 Botha et al. 4 studied breast cancer mortality trends in 16 European countries using the EUROCIM databases for the period 1958 (later in some countries) to 1994–1997 in the age-groups 35–49, 50–64 and 65–74. They noted significant decreases in the most recent decade in three countries with national screening programmes (England & Wales, Scotland and the Netherlands), although no decline was seen in Finland or Iceland. There was a continued longterm downward trend in Sweden. Decreases were also seen in three countries with no national screening programme (Slovakia, Spain and Switzerland).
Autier et al. 6 used the WHO mortality database to study breast cancer mortality 1989–2006 in 30 European countries. They used linear regression to analyse mortality in the periods 1989–2006 and 1999–2006, and estimated the annual percentage change for each country for the age groups <50, 50–69 and 70+. For the period 1989–2006 they found a median 19% reduction in breast cancer mortality at all ages (ranging from a 17% increase in Romania to a 45% reduction in Iceland), with the greatest reductions tending to be in those countries with higher initial mortality levels. For most of the 15 countries with reductions of at least 20% in this period, the year of the start of a decline was prior to 1996. The authors noted that while marked reductions were seen in some countries with organized screening (e.g. the UK), effects were less pronounced in other countries such as Sweden, and were also seen in age groups outside the range invited for screening.
Quinn and Allen 3 studied breast cancer mortality in England and Wales 1950–1994. They showed a steep decline after 1990 in the age standardized mortality in women aged 55–69, but concluded that the reduction was too soon after the introduction of screening in 1988–1990 to be wholly attributed to screening.
Törnberg et al. 5 studied time trends in breast cancer mortality in women aged 40–79 during the period 1970–1998 in the Nordic capitals, and related these to the introduction of mammography screening. They found no pronounced decrease in trend in any age group in any of the four capitals, where screening had been introduced from 1986 (in Helsinki), 1989 (in Stockholm), 1991–1994 (in Copenhagen) and 1996 (in Oslo). This was ascribed to the long-implementation period of screening in some places, and to the relatively short follow-up in some.
Ugarte et al. 2 studied spatio-temporal patterns in mortality trends in Spain 1975–2005 and suggested that differences between provinces in the observed decline could be partially explained by differences in implementation of screening.
Analytical studies
The 12 papers that quantified the effect of screening on breast cancer mortality are summarized in Table 2. Of these, three used joinpoint regression, and the remainder Poisson regression. Five papers studied the whole of an individual country (England (2) the Netherlands (2) and Spain), two studied the programme in Florence, Italy, two studied different regions in Spain and one studied two regions of Denmark. One paper included Northern Ireland, Netherlands and Sweden in comparison to the Republic of Ireland, Belgium/Flanders and Norway, respectively. The most recent paper studied nine counties in Sweden.
In the Netherlands, screening began for women aged 50–69 years in 1988–1989, with full coverage by 1997. Otto et al 10 used a Poisson regression model to study breast cancer mortality in women aged 55–74 between 1980 and 1999–2001, and found that mortality rates fell by 1.7% per year after the introduction of screening. Age-standardized mortality fell by 20% between 1986–88 and 2001. The authors attempted to study the confounding role of adjuvant therapy by grouping municipalities according to the date of starting screening, and demonstrated that in each of four clusters the turning point in mortality trends occurred at about the time of screening implementation. This study had limited follow-up (2–4 years) after full coverage had been reached, and the estimates of annual percentage change may have been diluted by the inclusion of the first years after the introduction of screening. More recently Otten et al. studied mortality trends to 2006, using joinpoint regression, and showed a significant 2.3–2.8% reduction per year in the age groups 55–64 and 65–74, respectively, after 1994. 11 At younger ages (45–54) there was an early decrease (1971–1980) with a declining trend also after 1992. The study therefore has at least 10 years follow-up after full coverage by screening, but the phased implementation of screening may have made it difficult to identify a joinpoint associated with screening impact.
In the UK, an early study used age period cohort modelling to compare breast cancer mortality between 1969 and 1998 in the age group 55–69 likely to be affected by screening and outside age groups where any reduction was likely to be due to changes in treatment. 16 The study estimated a 6.4% reduction due to screening in 1998 (21% reduction at ages 55–69 compared with 15% in other age groups). However, as the authors emphasized, this was too early to observe the full effect of screening, as the estimate relates to 1998 which is only five years after full screening coverage was reached. Duffy et al. 17 also fitted a Poisson regression model to breast cancer mortality rates in England for 1974–2004. They showed a significant 28% reduction in breast cancer mortality in the period 1995–2004 in the age group invited to screening (50–69) compared with the pre 1989 period relative to the change in other age groups. This study had follow-up of at least 10 years from full coverage, but the inclusion of the age group 50–54 in this analysis may have diluted any observed effect.
Ascunce et al. 12 also used joinpoint regression to study breast cancer mortality rates between 1975 and 2004 in Navarre, Spain, where screening for women aged 45–65 was introduced in 1990, and showed a decrease of 9.0% per year in women aged 50–69 from 1995 onwards, with no significant trends in other age groups. They also performed an analysis excluding prevalent cancers, and found that the overall decrease in mortality in the age group 50–69 between the prescreening (1986–1990) and screening (1997–2001) periods rose from 35% to 42% with the exclusion of such cases. This study had 12 years of follow-up, and the joinpoint reflects the point in time when an effect of screening might be expected.
Pons-Vigues et al. 13 studied breast cancer mortality between 1984 and 2004 in Barcelona, where a screening programme was introduced between 1995 and 2004 for women aged 50–69 years. They used Poisson regression to compare rates in different districts according to year of implementation, also adjusting for socioeconomic status. Overall they found a reduction of 5% per year after the start of screening, with a relative risk of 0.83 (17% reduction) in the city as a whole after full implementation (relative to the prescreening period). However, follow-up did not extend beyond the year when full coverage by screening was reached, and the estimate of annual percentage change included the early years of the programme. Again the inclusion of the 50–54 age group may have diluted the results. Cabanes et al. 14 studied breast cancer mortality in the whole of Spain 1980–2006. Using change-point regression they identified a downturn in all age groups after 1992, but this was most pronounced in younger women aged 25–44, and was unlikely to be due to screening because 90% coverage was not achieved until 2001. There was therefore only five years follow-up from this time, and the change points identified did not relate to the introduction of screening.
In Italy, screening was introduced on a regional basis. Barchielli and Paci 8 studied breast cancer mortality trends in Tuscany between 1970 and 1997, comparing the Florence area (where screening was introduced in 1990 for women aged 50–69) with the rest of Tuscany. They found similar trends of decreasing mortality in both areas (although slightly more pronounced in Florence). Reductions were similar across age groups (including and excluding screening), and the results were interpreted as an effect of early detection (not necessarily screening) and improvements in treatment. The fact that 72% coverage in Florence was only achieved in 1995 means that the analysis is too early to have observed the full impact of screening, with only two years of follow-up after full coverage. In addition, the authors compared Florence with the rest of Tuscany using age standardized rates for all ages, rather than focusing on the age group where screening would have an impact.
Gorini et al. 9 compared breast cancer mortality in early and late starting areas in the province of Florence in 1985–2000, and showed that age standardized rates fell by 41% over this time period in the early starting areas compared with a reduction of 11% in the late starting areas, suggesting a 30% reduction due to screening. There were significant reductions of 61% and 45% in the early starting areas in the age groups 45–54 and 65–74, respectively. However, there was also a non-significant decrease in the age group 55–64.
More recently, Jørgensen et al. 7 studied the effect of organized mammography in Denmark where screening was introduced in Copenhagen in 1991 and Funen in 1993. Mortality was examined between 1971 and 2006, and the reduction from 1997 onwards in these two areas was compared with the rest of Denmark. In the age group 55–74 the reduction was 1% per year in the screened areas and 2% per year in the unscreened areas. However, mortality in the prescreening period was higher in Copenhagen and Funen combined than in the rest of Denmark, and by 2006 had fallen to a similar level to that in the rest of the country. Autier et al. 18 compared breast cancer mortality 1980–2006 in three pairs of neighbouring European countries with different implementation of screening, using joinpoint analysis. 18 The authors present only summary rates and percentage reductions without confidence intervals. Although they observed no difference within each country pair in the change in mortality at all ages 1989–2006, the reduction at ages 50–69 in both Northern Ireland and the Netherlands, where screening started early, was greater than that in the neighbouring country.
The inclusion of rates in women from age 50 may have diluted the findings, the joinpoints identified do not relate to the introduction of screening and do not appear to be used in the interpretation of the results.
Haukka et al. 15 studied breast cancer mortality in nine counties in Sweden using data from the NORDCAN database 1974–2003. They fitted a Poisson regression model including factors for secular trend and screening effect based on the start of screening in each county. The results are difficult to interpret as no detailed data are provided on the fitted model. The model appears to include an adjustment to take account of the lead time before the effect of screening becomes apparent. The authors estimated a 16% reduction due to screening in women aged 40–69, with an 11% reduction at ages 70–79. However it is not clear from the paper at what time point these reductions are estimated. Again the age range is not focused on that which would be affected by screening.
Discussion
Of the papers identified that attempted to use trends in breast cancer mortality to quantify the effect of screening, some estimated the annual percentage change in mortality, while others presented a comparison between average levels of mortality in two distinct time periods. Of the former, estimates ranged from reductions of 1.0% to 9.0% per year; for those studies with adequate follow-up the estimates were 1.0%, 2.3–2.8% and 9.0% per year over a 10–12 year period. Of the three studies comparing time periods within a single country,9,12,17 all had long enough follow-up, and the estimates of mortality reduction compared with a prescreening period ranged from 28% to 36%.
Population mortality rates include deaths in women with breast cancer diagnosed before the introduction of screening, which will dilute any observable impact of screening. Rates in the youngest age groups invited to screening will always be diluted due to deaths in cases diagnosed at ages below the screening age range. In comparison, the advantage of individual-based studies is that they allow ‘refined mortality’ that excludes such deaths to be evaluated. 1 The majority of studies included a period immediately or soon after the start of screening and inclusion of this period is likely to reduce any observed effect per annum. Studies that include the youngest age group invited to screening are also likely to underestimate the effectiveness of screening because there should be at least 3–4 years after first invitation to screening before any impact of screening can be seen.
Several papers did attempt to limit the dilution due to the inclusion of deaths in prevalent cases by restricting the age group to five years above that invited for screening, and/or by including a time lag between the start of screening and the period in which mortality was studied. Nevertheless this is unlikely to take account of this dilution completely.
Many studies had limited follow-up after the time at which full screening coverage was achieved, including two of the studies reporting only a small or no mortality reduction. 8 , 16 Many studies included age groups outside the screening age range and compared trends in these age groups with those in the age group(s) likely to be affected by screening. However, factors other than screening, such as treatment, might differ between these groups, which mean that these comparisons should be interpreted with caution. Ascunce et al. 12 note that the large reduction in mortality noted for their screening age group is unlikely to be entirely due to screening, and cite the contemporaneous advent of adjuvant hormonal therapy as a possible confounder.
A number of studies used joinpoint regression to attempt to identify the time at which changes in breast cancer mortality occurred, but the joinpoints identified did not always correspond to the point in time at which screening would be expected to have an impact, emphasizing the difficulty in accounting for factors other than screening that will have influenced mortality. The more gradual impact due to phased implementation of screening means that it will be more difficult to identify a single time point at which mortality rates change, although three studies attempted to group areas by start date.10,13,15 Individual data would allow all women's exposure to be measured from the correct point in time.
Opportunistic screening prior to the introduction of an organized programme will dilute any observed effect of organized screening on mortality trends, as has been reported for Norway 19 . Changes in treatment over time will also make trend studies difficult to interpret. None of the studies were able to quantify the effect of opportunistic screening, or of changes in treatment (other than by grouping areas according to the start date of screening). In addition, there may be inaccuracies in cause of death coding for official statistics (although such inaccuracies will also affect other types of studies as well as trend studies).
Several studies noted larger reductions in countries or regions with the highest initial mortality rates, emphasizing the need to take into consideration the different levels of mortality in the period before implementation of screening. The fact that some geographical comparisons of trends have tended to identify similar trends in areas with different implementation of screening may reflect the difficulty in taking account of differences in the background levels and changes in these between different areas, and also in identifying the correct point in time from which to measure rates of decline. This highlights the difficulty in accounting for the effect of other influences on breast cancer mortality, and the need for a more rigorous approach to the evaluation of service screening.
CONCLUSION
The majority of studies suggested reductions in breast cancer mortality as a result of mammographic screening. Although the varied methodology of these studies means that it is difficult to produce a pooled estimate of mortality reduction, the majority of studies that estimated the annual percentage change in mortality found reductions ranging from 1% to 9% per year for approximately 10 years after the introduction of screening, while those that compared mortality in two distinct time periods within a single country found reductions of 28–36% in the postscreening period compared with the prescreening period. The study and analysis of population breast cancer mortality rates can be a first step in evaluating the impact of screening on mortality, but other methods possibly based on individual data (either by a cohort or a case control approach) and excluding deaths in cases diagnosed before the start of screening are necessary to reliably quantify the estimated benefit. Where analyses of trends are performed, they should ideally be restricted to the age ranges likely to demonstrate a benefit from screening, attempt to exclude time periods where dilution due to deaths in women diagnosed pre-invitation will be evident, and should attempt to take account of past underlying trends in breast cancer mortality.
EUROSCREEN WORKING GROUP
Coordinators
Members
Ancelle-Park, R (F) 1 , Armaroli P (I) 2 , Ascunce N (E) 3 , Bisanti, L (I) 4 , Bellisario C (I) 2 , Broeders M (NL) 5 , Cogo C (I) 6 , De Koning H (NL) 7 , Duffy SW (UK) 8 , Frigerio A (I) 2 , Giordano L (I) 2 , Hofvind S (N) 9 , Jonsson H (S) 10 , Lynge E (DK) 11 , Massat N (UK) 8 , Miccinesi G (I) 12 , Moss S (UK) 8 , Naldoni C (I) 13 , Njor S (DK) 11 , Nystrom l (S) 14 , Paap E (NL) 5 , Paci E (I) 12 , Patnick J (UK) 15 , Ponti A (I) 2 , Puliti D (I) 12 , Segnan N (I) 2 , Von Karsa L (D) 16 ,Törnberg S (S) 17 , Zappa M (I) 12 , Zorzi M (I) 6
Affiliations
1
Ministère du travail de l'emploi et de la santé, Paris, France
2
CPO-Piedmont, Turin, Italy
3
Navarra Breast Cancer Screening Programme, Pamplona, Spain
4
S.C. Epidemiologia, ASL di Milano, Italy
5
Radboud University Nijmegen Medical Centre & National Expert and Training Centre for Breast Cancer Screening, Nijmegen, The Netherlands
6
Veneto Tumor Registry, Padua, Italy
7
Erasmus MC, Dept. Of Public Health Rotterdam, The Netherlands
8
Wolfson Institute of Preventive Medicine, Queen Mary University of London, United Kingdom
9
Cancer Registry of Norway, Research Department and Oslo and Akershus University College of Applied Science, Oslo, Norway
10
Department of Radiation Sciences, Oncology, Umeå University, Umeå, Sweden
11
Centre for Epidemiology and Screening, Universtiy of Copenhagen, Denmark
12
ISPO Cancer Research and Prevention Institute, Florence, Italy
13
Regional Cancer Screening Center, Emilia-Romagna Region, Bologna, Italy
14
Department of Public Health and Clinical Medicine, Division of Epidemiology and Global Health, Umeå University, Umeå, Sweden
15
NHS Cancer Screening Programmes and Oxford University, United Kingdom
16
International Agency for Research on Cancer, Lyon, France
17
Stockholm Cancer Screening, Stockholm, Sweden
Footnotes
Acknowledgments
Financial support was provided by the National Monitoring Italian Centre (ONS) to host the EUROSCREEN meetings in Florence in November 2010 and in March 2011 and the supplement publication and the National Expert and Training Centre for Breast Cancer Screening, Nijmegen, the Netherlands to host a meeting of the EUROSCREEN mortality working group in July 2011.
