Abstract
Objectives
To examine the optimal screening interval among the individuals who received a negative Epstein-Barr virus immunoglobulin A antibodies against viral capsid antigen (VCA-IgA) serum test result and who comprised the majority of the population screened for nasopharyngeal carcinoma (NPC).
Methods
Screening was performed in Sihui, Guangdong, China, offering a repeated screening for participants with an initial negative test either after 4-5 years in one centre (short interval centre), or 9-10 years in another (long interval centre). The characteristics and incidence rates (IRs) of interval NPCs (defined as cases diagnosed outside the screening protocol while within the screening interval) were compared between these two centres. Standard incidence ratios (SIRs) were also calculated using the general Sihui population as the reference.
Results
Seven interval NPCs were detected in the short interval centre (IR: 17.8/10 5 person-years) and 20 in the long interval centre (IR: 20.8/10 5 person-years during the first four years and 43.5/10 5 person-years during the remaining years). The SIR in the short interval centre was 0.43 (95% confidence interval [CI]: 0.17-0.89); SIR in the long interval centre was 0.47 (95% CI: 0.17-1.02) during the first four years and 0.90 (95% CI: 0.49-1.51) during the remaining years. No aggressive interval NPC was observed in the short interval centre; four were identified in the long interval centre.
Conclusions
The incidence of NPC, especially aggressive NPC, was low during the first few years after a negative screening; the incidence increased to the general population level afterwards. A screening interval of 4-5 years may therefore be more suitable than 9–10 years after a negative VCA-IgA test in NPC screening.
Introduction
Detection of immunoglobulin A antibodies against viral capsid antigen (VCA-IgA) and early antigen (EA-IgA) by immunoenzymic assay (IFA) has been recommended as the preferred mass screening test by the Chinese medical authorities, and is widely performed in NPC screening in NPC endemic areas in China. 6 This is supported by the well-established relation between EBV and NPC, together with a higher early detection rate and improved survival when using VCA-IgA as the primary screening marker.7–10
Previous reports have shown that VCA-IgA positive individuals have a significantly higher risk of NPC and a shorter time interval to develop NPC compared with VCA-IgA negative individuals.2,11 Different screening intervals should be considered for individuals with different serological tests of EBV. Determining the optimal screening interval is of great importance for cancer screening. 12 Intervals between different screening rounds influence various screening outcomes, such as the cancer detection rate, screening efficacy and cost-effectiveness. 13
Individuals with a negative initial VCA-IgA test comprise the great majority of the general population screened for NPC. 11 To our knowledge, no large-scale study has yet reported repeated screening among this population. In the present study, we used data from a screening cohort in Sihui, Guangdong province, China, to examine the optimal screening interval for such individuals.
Methods
Study Population
In 1987 and 1992, a mass screening programme for NPC was launched in Longjiang and Jingkou towns and Jianggu and Didou towns of Sihui by Sun Yat-Sen University Cancer Center (SYSUCC) and Sihui Cancer Institute. Residents aged between 30 and 59 were invited to participate; informed consent was obtained from all participants. In addition to 3 mL blood, information on socioeconomic status and demographics was also collected. Individuals with a VCA-IgA titre ≥1:5 were defined as seropositive and were further tested for EA-IgA. Blood samples were transported to SYSUCC and all tests were performed in the laboratory there. The inclusion and exclusion criteria and the detailed screening protocol have been described previously. 2 The screening programme was approved by the Institutional Ethics Review Board of Sun Yat-Sen Medical University Cancer Center (No. YP2009169).
In the present study, we focused on participants with a negative VCA-IgA test result in the initial screening round. According to the screening protocol, the screening intervals for individuals with a negative VCA-IgA test were 4-5 years in Jianggu and Didou towns (the ‘short interval centre’) and 9-10 years in Longjiang and Jingkou towns (the ‘long interval centre’). Accordingly, all participants with an initial negative VCA-IgA test were invited to participate in rescreening in 1996 (Figure 1). The rescreening started on 1 July 1996 and was completed on 31 December 1997. All screening participants were followed from the initial screening until a diagnosis of NPC, death, migration or end of 1997, whichever came first, through record linkages to the Cancer Register, Cause of Death Register, the Rosters of Village Committee and the local Public Security Bureau in Sihui. The completeness of Sihui Cancer Register has been described in detail previously.1,2 The clinical characteristics of NPCs were collected through medical record review. Information on histopathology, symptoms, lymph node status, distant metastasis and treatment was extracted from the medical records. Pathologists who performed the assessment were blinded to screening status. In total, 90% (45/50) NPCs had retrievable medical records and 98% (49/50) were pathologically confirmed.
The trial flow diagram of the screening study for NPC in Sihui, Guangdong, China. NPC: nasopharyngeal carcinoma, VCA-IgA: immunoglobulin A antibodies against viral capsid antigen, EBV: Epstein-Barr virus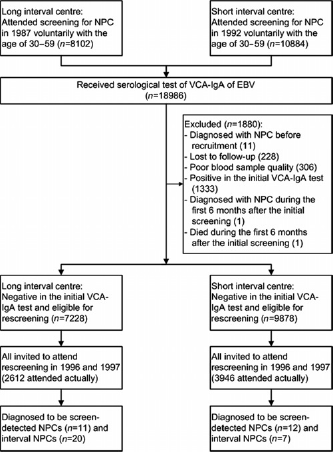
Definition of Interval Cancers, Screen-Detected Cancers And Aggressive Interval Cancers
Interval cancer was defined as any NPC diagnosed outside the screening protocol but within the screening interval. 14 In the present study, NPCs diagnosed more than six months after the initial screening and before the rescreening were defined as interval cancers. NPCs diagnosed within first six months after the initial screening were excluded, given the potential influence of initial screening on these cases. Interval cancers in the long interval centre were divided into two subgroups: cancers detected during the first four years and the remaining years, in order to compare with the short interval centre. NPCs diagnosed during the rescreening were considered to be screen-detected cancers.
Aggressive interval cancer was defined as an interval cancer that had at least one of the following characteristics at diagnosis: nodal size >6 cm, supraclavicular node metastases, involvement of skull base and/or cranial nerves, or multiple neck node metastases with one node > 4 cm.15,16
Statistical Analysis
Chi-square test and Kruskal–Wallis test were used to compare the baseline characteristics of screening participants between the two screening interval groups. Using the indirect method of standardization, we calculated the standardized incidence ratios (SIRs) for different screening intervals.17,18 The 95% confidence intervals (CIs) of SIR were calculated using the method proposed by Bailar and Ederer 19 , assuming a Poisson distribution. The expected number of NPCs was calculated by multiplying the person-years at risk accumulated in each group by the age-, sex-and calendar-year-specific incidence rate of NPC in the entire Sihui population.
All statistical tests were two-sided, and a P value of <0.05 was considered statistically significant. All analyses were conducted using SAS 9.2 (SAS Institute, Cary, NC, USA).
Results
Cancer Detection Rate
Of the 18986 participants in the NPC screening programme in Sihui, 17106 had a negative VCA-IgA test and were therefore included in the present study, including 9878 from the short interval centre and 7228 from the long interval centre (Table 1). Participants were more likely to be women in general. Compared with the short interval centre, participants were slightly younger and more likely to be men in long interval centre. A total of 3946 (39.9%) participants in the short interval centre and 2612 (36.1%) in the long interval centre attended rescreening. Up to 31 December 1997, 19 NPCs were detected in the short interval centre, including 12 screen-detected cancers (detection rate: 0.12%) and 31 NPCs were detected in the long interval centre, including 11 screen-detected cancers (detection rate: 0.15%) (Table 2).
Baseline characteristics of the participants with a negative VCA-IgA test in NPC screening in Sihui, Guangdong, China
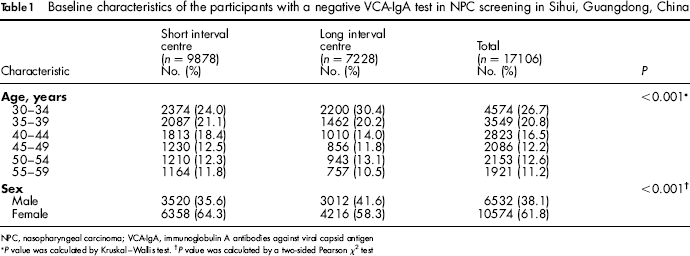
NPC, nasopharyngeal carcinoma; VCA-IgA, immunoglobulin A antibodies against viral capsid antigen
P value was calculated by Kruskal-Wallis test.
P value was calculated by a two-sided Pearson χ 2 test
Number and detection rate of screen-detected NPCs, interval NPCs and aggressive interval NPCs in Sihui, Guangdong, China
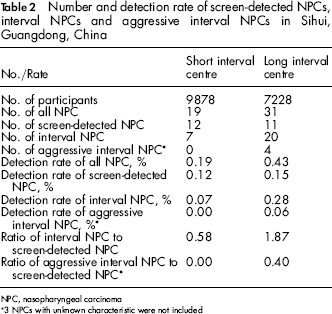
NPC, nasopharyngeal carcinoma
3 NPCs with unknown characteristic were not included
Identified Interval Cancers
Seven interval cancers were diagnosed in the short interval centre (none of which were aggressive interval cancers), leading to a detection rate of 0.07% for interval cancer. Twenty interval cancers were diagnosed in the long interval centre, including four aggressive interval cancers, resulting in a detection rate of 0.28% for interval cancer and 0.06% for aggressive interval cancer. The ratios of interval cancer to screen-detected cancer (i.e. the detection rate of interval cancer divided by the detection rate of screen-detected cancer) were 0.58 in the short interval centre and 1.87 in the long interval centre. The ratios of aggressive interval cancer to screen-detected cancer (i.e. the detection rate of aggressive interval cancer divided by the detection rate of screen-detected cancer) in these two groups were zero and 0.40, respectively.
Interval Cancers Per Screening Interval
The annual numbers of interval cancers and aggressive interval cancers in these two interval groups are presented in Table 3. In the long interval centre, a clear increase of interval cancer diagnosis, as well as aggressive interval cancer diagnosis, was noted over time. During the first four years, six interval cancers were detected; 14 interval cancers were detected during the remaining years. Aggressive interval cancers (n = 4) were diagnosed in the second, sixth, seventh and ninth year of the screening interval in this group.
Number of interval NPCs and aggressive interval NPCs per year more than six months after the initial NPC screening in Sihui, Guangdong, China
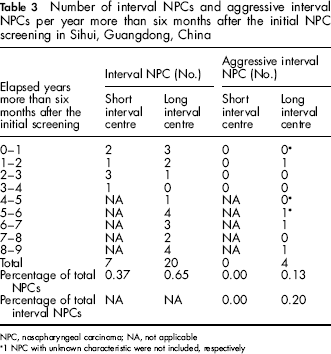
NPC, nasopharyngeal carcinoma; NA, not applicable
1 NPC with unknown characteristic were not included, respectively
Standardized Incidence Ratios of Interval Cancers
In total, 39 429 person-years were accumulated in the short interval centre, giving an incidence rate of interval cancer of 17.8/10 5 person-years and a SIR of 0.43 (95% CI: 0.17-0.89) (Table 4). During the first four years in the long interval centre, a total of 28835 person-years were accumulated, yielding an incidence rate of interval cancer of 20.8/10 5 person-years and a SIR of 0.47 (95% CI: 0.17-1.02); 32196 person-years were accumulated during the remaining years in the long interval centre, yielding an incidence rate of interval cancer of 43.5/10 5 person-years and a SIR of 0.90 (95% CI: 0.49-1.51).
Incidence rates and standardized incidence ratios (SIRs) of interval NPC during follow-up of the NPC screening in Sihui, Guangdong, China
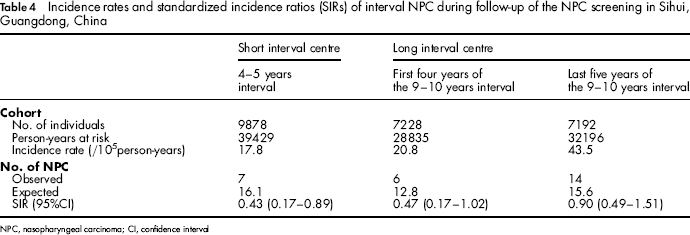
NPC, nasopharyngeal carcinoma; CI, confidence interval
Discussion
Although the screening protocol for NPCs has been developed for many years, the optimal screening interval is not clear, especially for participants with a negative VCA-IgA test. Using Markov chain models with the parameters of a hypothetical screening trial in Hong Kong, Chen et al. 20 suggested that there was no substantial difference between triennial and sexennial serum screening of VCA-IgA, and that in terms of cost-effectiveness, the sexennial VCA-IgA test combined with the triennial indirect mirror examination was sufficient. The screening strategy for NPC may be unsatisfactory, given a lack of consideration that individuals with different VCA-IgA titres have different risks and incubation time for NPC. Cheuk et al. 21 also used the Markov chain model to compare four NPC screening strategies with different screening intervals, and their results showed that the quadrennial screening for individuals with a negative EBV test and annual screening for individuals with a positive EBV test offered the greatest screening efficacy. However, this screening strategy was limited to participants with a family history of NPC rather than the general population. In southern China, individuals with different VCA-IgA titres in the initial screening round were allocated to different rescreening intervals, although the evidence for this guidance was lacking. 6 Usually, individuals with a positive VCA-IgA test would undertake shorter screening intervals than those with a negative test. In our study, we first compared the incidence rate of interval cancers among individuals with an initial negative VCA-IgA test in two screening centres with a repeated screening conducted after a 4-5 year interval and a 9-10 year interval, in order to explore the optimal screening interval for this population.
Interval cancers may be detected either because of rapid tumour growth or an inadequacy in the screening process. These cancers are inevitable in a cancer screening programme, but their numbers should be kept as low as possible in order to avoid decreasing the screening efficacy. 22 The incidence rate of interval cancer may shed light on the appropriateness of a screening interval. 14 Our results showed that the risk of NPC was 57% lower in the short interval centre, and 53% lower during the first four years in the long interval centre, compared with the general population, but there was no difference during the remaining years in the long interval centre compared with the general population in Sihui.
One explanation for the relatively high incidence rate of interval cancers during the last five years in the long interval centre may be that EBV antibody is a short-term indicator for NPC. Previous studies, including our own data, have illustrated that the serological levels of EBV antibodies are significantly associated with the risk of NPC, with the most prominent association observed during the first five years after the EBV test.2,23 A positive (although weaker) association was still noted more than five years after the EBV test. Similarly, the protective effect of negative EBV antibodies for NPC may also be short-termed. Ji et al. 11 reported that the NPC risk in EBV seronegative individuals was markedly reduced only during the first two years after test, but rose drastically after four years.
The proper length of screening interval should be evaluated not only by the incidence rate of interval cancers, but also taking into account the characteristics of the interval cancers. In our study, aggressive interval cancer was defined according to clinical symptoms and physical examination results extracted from the medical records. Before 1992, NPC tumour stage was largely determined by symptoms and X-ray examination; after 1992 CT scan was used. Consequently, we were not able to compare the tumour stage data directly for the different time periods in these two screening interval groups.
Aggressive NPCs are often incurable due to the high possibility of loco-regional recurrence and long-distance metastasis among these cases. 24 In the long interval centre, a high percentage of aggressive interval NPCs (20%) was observed, but no aggressive interval NPCs were detected in the short interval centre. Even in the long interval centre, only one aggressive interval cancer was detected during the first four years, with the other three detected during the remaining years of the screening interval. These aggressive interval cancers could be considered as cancers that might have been detected with more favourable characteristics if a shorter screening interval were adopted. 14 The results suggest that the 4-5 year screening interval may be more suitable for individuals with initially negative EBV antibodies in the NPC high-risk area.
Levels of EBV antibodies fluctuate. 2 Seroconversion from VCA-IgA negative to positive has been suggested as a key step in the pathogenesis of NPC, indicating EBV re-activation or re-infection. 25 We found a 6.6% (260/3946) seroconversion rate in the short interval centre, and a 6.8% (178/2612) seroconversion rate in the long interval centre. At rescreening, there were more individuals with high titres (> 1:20) in the long interval centre compared with the short interval centre (data not shown). Due to the striking dose-response relationship between VCA-IgA titres and NPC risk2,11,26,27, it is likely that individuals with negative VCA-IgA titres who attended rescreening after 9-10 years may have a higher incidence of NPC following the rescreening, compared with individuals attended rescreening after 4-5 years.
In addition to EBV related antibodies, EBV DNA can also be detected, using realtime quantitative polymerase chain reaction, in plasma and serum from NPC patients. EBV DNA showed a more specific marker for clinical diagnosis, monitoring treatment results and prediction prognosis of NPC patients.28–30 However, the role of EBV DNA detection as a screening test for NPC remains inconclusive. These short EBV DNA fragments are mainly released into the circulation by apoptosis of cancer cells, rather than by active viral re-activation, 31 suggesting that it may not be a suitable preliminary screening marker for high-risk population or for determining early stage NPC. A pilot NPC screening study in Taiwan showed that EBV DNA had low sensitivity to detect early stage NPC patients from high-risk families, and the virus load did not correlate with the EBV serological results. 32 Given the complementary characteristics between EBV DNA and serological antibodies, EBV DNA testing might be used in the future to improve screening effectiveness based on serological antibodies.
To our knowledge, this is the first report on interval cancers for different screening intervals of NPC. Using the age-, sex- and calendar-year-specific incidence rate of NPC in the entire Sihui population as a standard, we can compare directly the SIRs for two different screening intervals. An important limitation of our study is that the rescreening participation rate was low in both screening centres. The low compliance with rescreening might decrease the detection rates for NPCs and increase the ratio of interval to screen-detected cancers. Furthermore, a comprehensive cost-effectiveness analysis of the optimal screening interval should be conducted.
Conclusion
Our study shows that the risk of NPC was low in the 4–5 year interval group, and increased to the general population level during the last five years of the 9-10 year screening interval. More aggressive interval NPCs were observed in the 9–10 year interval group than the 4-5 year interval group. Our findings suggest that a screening interval of 4-5 years may be more suitable than 9-10 years for individuals with a negative initial VCA-IgA screening test results.
Footnotes
This work was supported by the Seventh (PI: Bao-Xiang Ou), Eighth (PI: Yin-Tang Wu), Ninth (969060103, PI: Teng-Bo Huang), Tenth (2001BA703B07, PI: Ming-Huang Hong) Chinese National Programs for Science and Technology Development and Eleventh (2006BAI02A11, PI: Ming-Huang Hong) National Science and Technology Support Program of China. The sponsors had no involvement in the design of the study; the collection, analysis and interpretation of the data; the writing of the manuscript; or the decision to submit the manuscript for publication. We thank all staff involved in recruiting the screening participants and running the screening tests, and staff in Sihui Cancer Registry for their help with data linkage and follow-up.
