Abstract
Objective
To report and discuss the observation of three fragments on polymerase chain reaction (PCR) in routine carrier screening for fragile X.
Methods
From 2005 through 2010, 34,500 women underwent prenatal screening for fragile X. PCR was carried out to amplify the repeat segment. The resulting fragments were scanned by a genetic analyser.
Results
Three PCR peaks representing three different-sized fragments were found in 25 of the 34,500 women (1:1380 or 0.072%). Karyotype analysis was performed in 16 subjects. Full triple X was found in three women, while two had triple X mosaicism. Of the 16 karyotyped women, five (31%) had a finding of XXX (full or mosaic).
Conclusions
Triple X (full or mosaic) is the most frequently encountered mechanism responsible for three peaks on fragile X PCR testing.
INTRODUCTION
Fragile X syndrome (FX) is the most common form of single gene disorder associated with cognitive impairment. FX is caused by a trinucleotide repeat expansion (CGG) in the 5′-untranslated region of the fragile X mental retardation 1 (FMR1) gene located at Xq27.3. 1 The disorder is inherited as an X-linked dominant mode with reduced penetrance affecting both men and women. However, the degree of cognitive disability is typically more severe in men. Most wild-type alleles of the FMR-1 gene have a repeat number of 29–30 CGGs. 2–4 Individuals having 55–199 repeats are defined as carriers of a premutation. 5,6 One in 813 men and one in 259 women in the general population were found to carry the premutation. 7,8 A recent study from Israel that included 40,000 women tested in a population-based screening programme revealed a premutation carrier rate of one in 154. 9 This relatively high rate might be due to a referral bias. Affected individuals have at least 200 repeats and are defined as having full mutations. Often, full mutations appear as smears in different analyses. They are occasionally accompanied by additional bands of smaller molecular weight in the premutation range. Such ‘mosaics’ can be identified in 10–20% of affected individuals, 7 suggesting that large pre-to-full mutation expansions also occur during early embryogenesis and not only during female meiosis. 10,11 The mechanisms responsible for these observations include difficulties in replicating the GC-rich repetitive sequences, resulting in slipping, strand mispairing, premature termination, intramolecular recombination and unequal crossing over. The replication errors may result in increased or decreased numbers of repeats, although the latter have been rarely reported in FMR-1. 2,12
Adir et al. 13 reported on a Southern blot of a woman from a screening programme for fragile X syndrome. Because the upper band, which represents the allele with the higher number of CGG repeats, was more intense than the lower band, it was suspected that the amount of the DNA in the two FMR1 alleles was not equal. This observation suggested the possibility of aneuploidy involving the FMR1 gene, which would most likely be the result of aneuploidy involving the X chromosome. Analysing the karyotype resulted in the finding of a 47, XXX. However, the occurrence of three peaks on routine screening for FX has not been reported previously. Here, we describe 25 such cases and discuss possible causes in the context of instability mechanisms.
METHODS
From 2005 through 2010, 34,500 women participated in a prenatal screening programme for FX genotype status. The initial, routine analysis was performed by PCR amplification of the repeat region using the primers: 5′-GCTCAGCTCCGTTTCGGTTTCACTTCCGGT-3′ and 5′-AGCCCCGCACTTCCACCACCAGCTCCTCCA-3′. Amplification was carried out in a 10 µL reaction containing 50 ng of DNA, 4 pmol of each primer, 1.5 mmol/L dNTPs, in 1.5 mmol/L MgCl2 PCR buffer, with 0.4 U of Taq polymerase (Roche). After an initial denaturation of five minutes at 96°C, 14 cycles were performed (96°C for 1 minute, 60°C for 2 minutes and 72°C for 3 minutes), followed by 20 additional cycles (94°C for 45 seconds, 60°C for 1 minute and 72°C for 2 minutes), and a final extension of eight minutes at 72°C. PCR products were read using a genetic analyser (3010 xl, Applied Biosystems, Foster City, California, United States). 14,15 Samples with three peaks were retested with a second set of primers: 5′-GACGGAGGCGCCGCTGCCAGG-3′ and 5′-GGGGCCTGCCCTAGAGCCAAGTACCTT GT-3′ in order to reduce the chance of amplifying a different part of the genome as much as possible. Genetic counselling was offered to all women who had three peaks. Occasionally, additional tests such as karyotype in lymphocytes by cytogenetic analysis, or the FX status of the woman's parents were performed.
RESULTS
During a six-year period, we encountered three different-sized peaks on the gene analyser scan in 25 of 34,500 women screened for FX (0.072%). In most cases, one of the peaks was considerably shorter than the other two (Figure 1).
Three peaks in fragile X testing. One peak is usually considerably smaller than the other two (patient 20)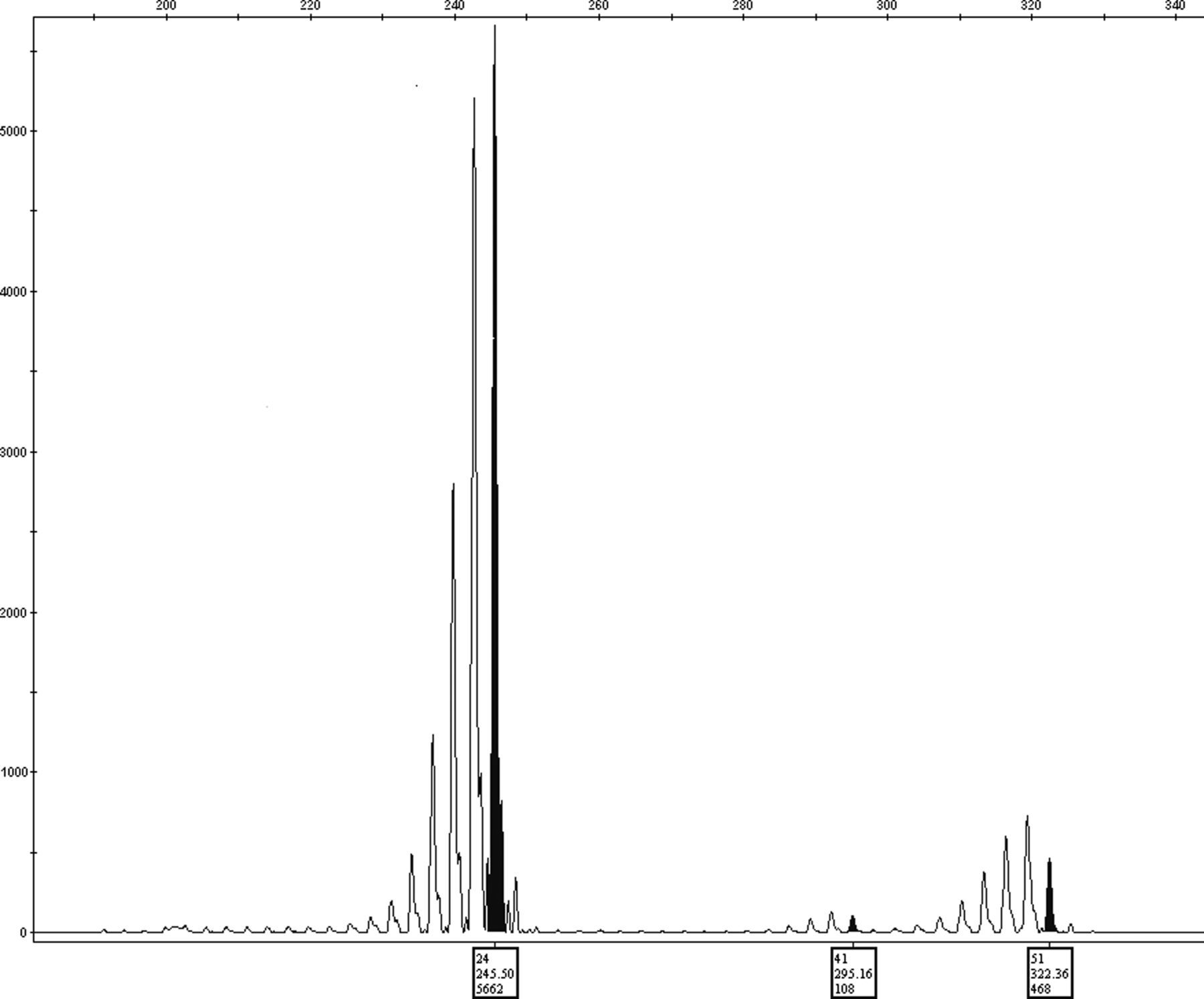
The height of the peak is determined by a few parameters, including the initial amount of DNA and the size of the amplified segment; the shorter segment being amplified better.
Summary of the women with three peaks on fragile X screening
In two of the remaining nine cases, the women's parents consented to be tested for FX, and both revealed two peaks in the mother and one peak in the father. The fetus was tested in two cases. The first test was performed due to a fragment size of 58 in the woman's FX screening test. The second woman had amniocentesis due to maternal anxiety. Both of her parents had been tested for FX previously. In those two cases, a normal karyotype was found. Both fetuses were females and two peaks were seen.
DISCUSSION
Usually, FX PCR testing yields one or two fragments according to whether the female tested is heterozygous or homozygous for CGG repeats. In this study, PCR analysis revealed three peaks in 25 cases. We found only one proven mechanism to explain this rare observation: a complete or mosaic triple X that was seen in metaphase karyotype in five of 16 women (31%) who consented to additional genetic analysis. Triple X is an unexpected diagnosis for the woman, and generates some anxiety, although it imposes no adverse implications for the pregnancy.
Conversely, there is also a small chance for some triple X individuals to have only two or even a single peak due to homozygosity. A recent publication described a woman with fragile X and XXX, with two alleles showing a full mutation of 580 repeats. 16 The most plausible mechanism for the remaining cases is a low-level mosaicism involving the sex chromosomes that is not detected by cytogenetic analysis. This hypothesis is supported by the fact that in most cases, one of the peaks was considerably shorter than the other two. If, as we suggested, the third peak represents low-level mosaicism and a correspondingly low amount of DNA, this explains why the representative peak is shorter than the others.
Numerous reports describe low-level mosaicism that is revealed only by molecular–cytogenetic measurements. 17–19 Awareness of this phenomenon has increased recently due to microarray CGH testing, which finds a low percentage of mosaicism in many samples. 20–24
Other theoretical explanations for three peaks include duplication of part of the X chromosome containing the fragile X site with a different fragment size. Under such a scenario, a male fetus would have an increased risk for chromosome X-related disorders, and duplication of the fragile X site elsewhere in the genome and not on the X chromosome.
Due to the possibilities mentioned above, we offer additional genetic investigation to all women in whom we find three peaks. This includes genetic counselling, followed by a karyotype. If the woman's karyotype is not triple X, FX testing of her parents is indicated. The finding of three peaks in the maternal mother or two peaks in the maternal father is consistent with duplication. While a result showing three peaks in the maternal mother would necessitate further investigation, two peaks in a ‘normal’ maternal father would suggest a benign prognosis. If three peaks are found in the maternal mother or if the maternal parents are unavailable, we suggest PCR FX testing on male fetuses to rule out two peaks, which may indicate duplication. If two peaks are detected in a male fetus, we offer microarray CGH for the fetus, to rule out duplication. An alternative approach would be to offer all women with three peaks, in whom the cytogenetic examination did not reveal triple X, microarray CGH in order to search for low grade mosaicism or duplication.
CONCLUSION
The appearance of three peaks in FX screening is a very rare phenomenon. Almost a third of the cases can be accounted for by the triple X syndrome. There is no conclusive explanation for the remaining cases, but very low level mosaicism of the X chromosome, undetectable by conventional cytogenetic testing is the most likely aetiology.
Footnotes
ACKNOWLEDGEMENTS
The authors thank F Schreiber MS for editorial assistance.
