Abstract
Objective
Early detection through mammographic screening and various treatment modalities of cancer may have changed life expectancy and cause-specific mortality of breast cancer patients. We aimed to determine the long-term cause of death patterns in screening-detected patients and clinically diagnosed patients in the Netherlands compared with the general population.
Methods
Using data from the Netherlands Cancer Registry and Statistics Netherlands of around 26,000 women, aged 50–75 at diagnosis and surgically treated for invasive breast cancer in 2004–2008, we compared patients with screening-detected and clinically diagnosed cancer for major causes of death until 2020. The expected number of all-cause and cause-specific deaths was calculated using rates of the general population.
Results
During the follow-up period, 4310 women died. The age-standardised all-cause mortality ratio for the screening-detected cancer group was 1.41 (95% confidence interval (95% CI), 1.37–1.46). A higher mortality ratio was observed for patients with clinically detected cancer: 2.27 (95% CI, 2.19–2.34). The observed versus expected breast cancer mortality ratio in the screening-detected patient group was 8.92 (95% CI, 8.45–9.40) and 20.23 (19.38–21.09) in the clinical group. Excess mortality was found for lung cancer in both patient groups, and small elevations for circulatory and respiratory disease in the clinically detected group.
Conclusion
Our results indicate that for the screening group no other causes of death but breast and lung cancer were prominent compared with the general population. The clinical group showed excess mortality for some other causes of death as well, suggesting a less healthy group compared with the general population.
After randomised trials and a range of observational studies showed mammographic screening to be effective in reducing breast cancer (BC) mortality in the long term, an increasing number of countries have introduced nationwide BC screening in the past decades.1,2 Most programmes involve invitation of the target age group starting from age 50 years. It is expected that due to early and less intensive treatment, mortality from complications and/or adverse long-term effects of the treatment (such as radiotherapy) is lower. 3 On the other hand, the reduction in BC deaths in this group may partly be compensated for by deaths from, for example, more frequent use of radiotherapy.4,5
In the Netherlands, the nationwide BC screening programme was stepwise implemented in the 1990s. Women aged 50 to 75 years are invited for a mammographic examination every two years. Within this setting, the primary objective of our study was to investigate the long-term cause of death patterns in screening-detected invasive BC patients compared with clinically diagnosed invasive BC patients. Patients were diagnosed in 2004–2008 and followed up until 2020. Our secondary objective was to assess excess mortality by comparing both BC patient groups with the general female population.
The study used data received from the Netherlands Cancer Registry and linked to Statistics Netherlands for vital status (including cause of death). By the end of 2019, 32% of the 26,166 patients had died. For cause-specific mortality the ICD-10 International Statistical Classification of Diseases and Related Health Problems was used. 6
Standardized cause-specific mortality ratios were estimated as observed/expected ratio. For the screening-detected BC group and the clinically diagnosed BC group, observed (O) numbers of the most common cause-specific deaths were tallied by 5-year age and period categories. Expected (E) numbers of deaths were calculated by multiplying the age-period specific death rates in the general population (available from Statistics Netherlands) by the follow-up time of both patient populations. The follow-up time starts at the year of diagnosis and ends at the year of death or the year of censoring. The 95% confidence interval (95% CI) of the O/E ratio was calculated using Byar's approach. 7
Of the registry-linked patients, 14,546 (55.6%) were screening-detected and 11,620 (44.4%) clinically detected. Patient groups differed significantly in age, stage at diagnosis and treatment (p < 0.0001). Mean patient age was 62.2 years (standard deviation (SD), 0.05) and 60.6 years (SD, 0.06), respectively. Compared with clinically detected BCs, screening-detected BCs were more likely to be stage I (63.0% vs. 34.3%), and less likely to be stage II (30.8% vs. 46.4%) or stage III (6.3% vs. 19.3%). Screening-detected BCs were more often treated with breast-conserving surgery (69.4% vs. 51.2%), which was also reflected in the applied radiation (73.5% vs. 67.4%).
The observed all-cause mortality in the screening-detected and the clinically diagnosed BC groups were respectively 1.41-fold and 2.27-fold higher than expected (see Table 1). In cancer mortality, the strongest increase was found for BC death, evidenced by O/E ratios of 8.92 (95% CI, 8.45−9.40), and 20.23 (95% CI, 19.38−21.09), respectively. In absolute terms, lung cancer was also a relatively common cause of death in both groups. The O/E ratios were 1.17 (95% CI, 1.04−1.31) and 1.21 (95% CI, 1.06−1.39), respectively. Among the other main causes of death, excess mortality was observed in the clinically diagnosed BC group for pancreatic and ovarian cancer (O/E = 1.46; 95% CI, 1.15–1.83 and O/E = 1.50; 95% CI, 1.16–1.92), myocardial infarction (O/E = 1.77; 95% CI, 1.46−2.11), and COPD (O/E = 1.22; 95% CI, 1.05−1.40). In the screening-detected BC group, the standardized cause-specific mortality ratios spread around unity.
Age and time adjusted mortality ratios of specific causes of death according to the ICD-10 in patients with screening and clinically detected breast cancer.
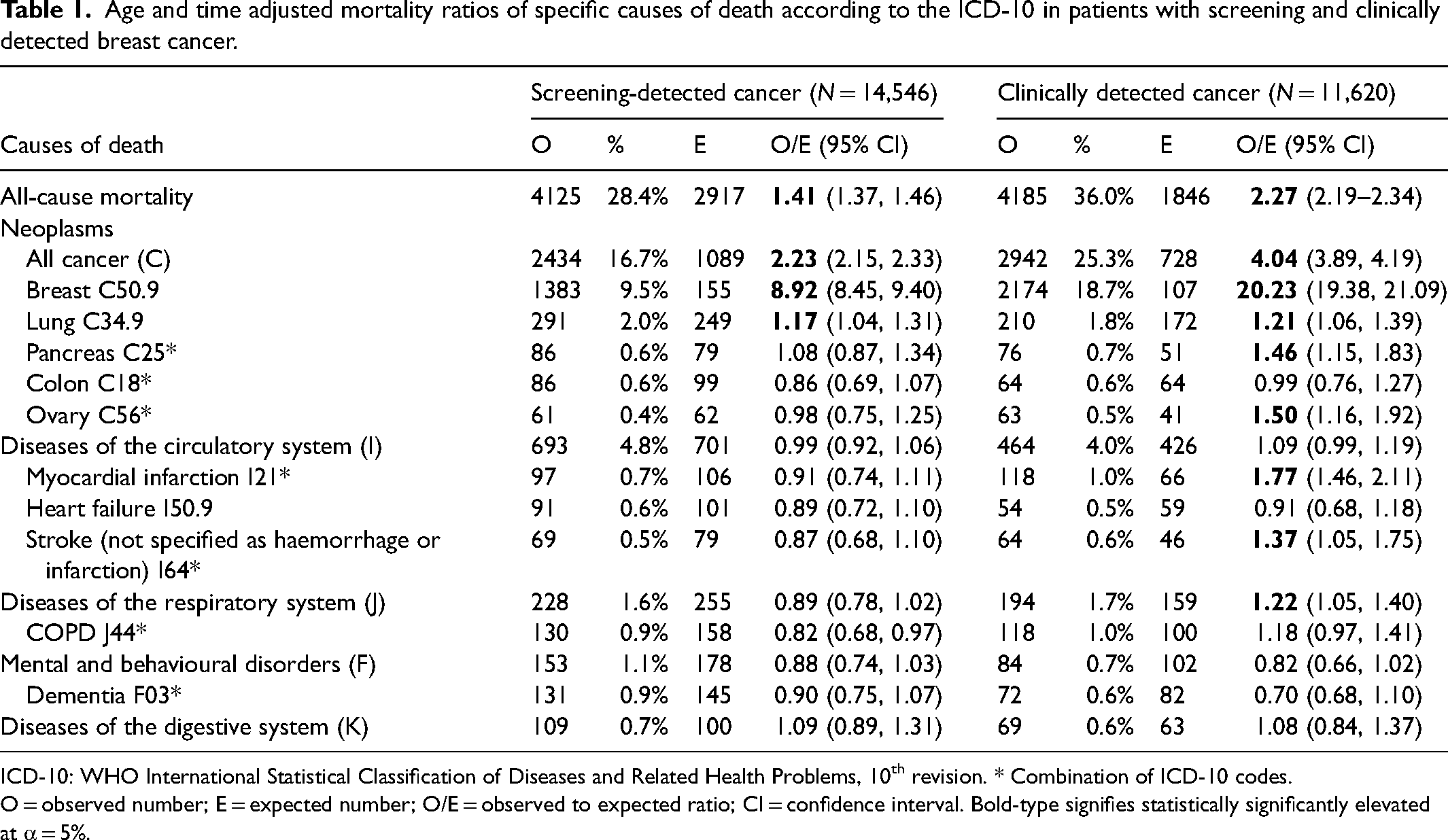
ICD-10: WHO International Statistical Classification of Diseases and Related Health Problems, 10th revision. * Combination of ICD-10 codes.
O = observed number; E = expected number; O/E = observed to expected ratio; CI = confidence interval. Bold-type signifies statistically significantly elevated at α = 5%.
As expected, BC mortality was the most prominent cause of death, almost 9 times more common in the screening-detected BC group compared to the general population and 20 times more common in the clinically diagnosed BC group. This difference between the groups is at least partly due to the more favourable stage distribution in the screening-detected group compared to the clinically diagnosed group.
In addition to BC mortality, lung cancer mortality is the only cause of death that was increased in both groups. This could be the consequence of radiation therapy to the breast, where the lungs are also within the radiation field. Radiation therapy was used in 74% of the patients in the screening-detected group and in 67% of the clinically diagnosed group (data not shown). However, the literature focusses more on factors such as smoking behaviour in combination with the radiation dose. 8 Damage to the heart (heart muscle, valves, etc.) may also be a long-term side effect of radiation. However, the O/E ratio was not increased in the screening-detected BC group. Excess mortality of the aforementioned causes of death as a result of radiation therapy is therefore not obvious.
The observed numbers of death from diseases of the circulatory and respiratory system in the screening-detected BC group correspond to the expected numbers in the general population. In this sense, the screening group appears to be ‘as healthy’ as the general population. In the clinically diagnosed group, however, deaths from respiratory disease are more common compared to the general population. Also, deaths from myocardial infarction, stroke, and of the malignant neoplasms of pancreatic and ovarian cancer are more common in the clinically diagnosed group than in the general population. This excess mortality does not seem to be the result of the applied treatments. A potential explanation is that the clinically diagnosed BC patients are a less healthy group of BC patients. The decision not to participate in the population screening for BC could possibly be associated with general health. However, it is important to note that the case mix of patients in terms of stage at diagnosis, socioeconomic deprivation and treatment received could also have been different between the screening-detected and clinically diagnosed groups.
In conclusion, for the screening-detected group, no other causes of death than BC are increased compared to the general population. The clinically diagnosed group shows an excess mortality for several causes of death and could therefore represent a less healthy group compared to the general population. It is not possible to determine from the data whether these patients were already ‘less healthy’ at the time of diagnosis or whether their condition deteriorated afterwards.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Centre for Population Screening (CvB) of the Dutch National Institute for Public Health and the Environment (RIVM). The CvB was not in any way involved in the study design, analysis or interpretation of the data, nor in the writing of the report or in the decision to submit the article for publication.
