Abstract
Objective
To assess the comparability of self-collected cervicovaginal samples and provider-collected cervical samples for the detection of human papillomavirus (HPV) DNA among Inuit women in Nunavik, Quebec, avoiding the use of liquid-based storage and transport of the self-collected samples.
Methods
Ninety-three women aged 18–69 years were recruited from a previously formed cohort on the natural history of HPV to this cross-sectional measurement study. This study utilized HPV DNA test results from 89 paired specimens collected by study participant and health provider with Dacron swabs. Samples were tested for 36 HPV types with the PGMY-primer PCR protocol and genotyping with the linear array method. Unweighted kappa statistics and McNemar tests were used to measure the agreement between sampling techniques.
Results
In the self-collected samples, 30 different HPV types were found, compared with 29 types found in the provider-collected samples. The prevalence of high-risk (HR) HPV was 38.2% in the self-collected samples and 28.1% in the provider-collected samples. The agreement between collection methods for the detection of HR-HPV DNA (85.4%) was good. HR-HPV and type-specific HPV 16/18 were as likely to be detected in the self-collected samples compared with the provider-obtained samples.
Conclusions
Women in this population were easily able to collect adequate cervicovaginal specimens for HPV testing. As self-sampling has a high recovery of HR-HPV and is comparable with provider-sampling, we conclude that self-sampling with dry storage and transport could be a good cervical cancer screening alternative for Inuit women in Nunavik who have traditionally avoided speculum examination.
INTRODUCTION
The discovery that persistent human papillomavirus (HPV) infection is a necessary cause of cervical cancer has led to the development of novel cervical cancer screening strategies based on HPV DNA testing. 1,2 HPV testing has several advantages over traditional cytology, such as a higher sensitivity to detect cervical cancer, greater reproducibility and potential for automated high-volume testing. 2,3 Another benefit of HPV testing is that it can be conducted on vaginal specimens collected by the patients themselves. Studies that have evaluated the accuracy of an HPV-positive sample to detect high-grade cervical disease have shown that the sensitivity of self-collected samples is high, but somewhat lower than provider-collected samples. 4–9 In the previous literature on the comparability of these sampling techniques to detect HPV infection, samples were usually preserved in a liquid storage system, but a dry system would be preferable, given that it is more straightforward for patients to use. 10–12 Self-sampling would also be useful to researchers conducting HPV natural history studies because it is less costly and samples would be easier to send via mail than samples in vials containing a liquid media.
Additionally, self-sampling has the potential to increase the number of women screened for cervical cancer by increasing the participation of screening non-responders. 13,14 As self-sampling is a less invasive method, it may be appealing to women who fear speculum examination conducted by a health provider because of previous abuse, 15 feelings of embarrassment, 16 perceived inconvenience, 17 and cultural and religious reasons. 18,19
The Quebec Inuit population, who have a higher cervical cancer incidence and mortality rate than the general population of Quebec, 20 could benefit from a screening programme that incorporates HPV testing of self-collected samples. The natural history of HPV has been studied in an ongoing cohort of Inuit women from Nunavik, the sub-arctic and arctic region of Northern Quebec. 21–23 Results of these studies reveal that Quebec Inuit women are at high risk of HPV infection and Pap smear coverage within the previous three years was estimated at 71%. Despite the potential for self-sampling to increase cervical cancer screening coverage in high-risk groups, no known published study has examined the accuracy of self-sampling in the Canadian Inuit population. The objective of this study was to assess the comparability of self-collected cervicovaginal samples and provider-collected cervical samples for the detection of HPV DNA among a sample of Inuit women participating in an ongoing cohort study in Nunavik, Quebec, avoiding the use of liquid-based storage and transport of the self-collected samples.
PATIENTS AND METHODS
Women were recruited to this study from a cohort formed between 2002 and 2007 that was comprised of 554 Inuit women aged 15 to 69 living in Nunavik, Quebec. 22 Nunavik's population, of which approximately 90% self-identify as Inuit, is distributed between 14 communities situated on the coasts of Hudson Bay, Hudson Strait and Ungava Bay. 24 Eligibility for the self-sampling sub-study and the cohort study were the same, except for the age criteria which was restricted to ages 18–69 years. 22 Recruitment for the self-sampling sub-study occurred between December 2007 and June 2010 in two communities of Ungava Bay, Nunavik by nurse practitioners. They systematically asked all non-enrolled cohort participants if they would like to participate in the study as they came to the clinic for any visit necessitating a Pap test.
After eligibility was determined and the consent form completed, the nurse practitioner explained the self-collection procedure and participants were provided with a diagram. Consenting women were asked to collect a self-sample, unsupervised in the examination room. Women were asked to squat or put one foot up on a chair and insert a sterile 15 cm dry Dacron swab (Copan 159C) into the vagina, as high up as possible, to the vault and to rotate the swab three times. The swab was then to be inserted into the accompanying dry plastic tube. After the self-sample was collected, the nurse practitioner returned to the examination room to conduct a pelvic examination with direct cervical cell sampling. Ectocervical and endocervical cells were then collected with a Dacron swab by the nurse through direct cervical sampling, and were resuspended in 1.5 mL of a methanol-based liquid, PreservCyt (Cytyc Corporation, Boxborough, MA). Cell suspensions in PreservCyt and dry swabs were kept at 4°C until the samples were transported on wet ice to Montreal for HPV typing.
The protocol for cell preparation and HPV DNA detection by PCR amplification using PGMY09-PGMY11 consensus primers and Line Blot Assay (Roche Diagnostics) was followed as previously described. 22 HPV genotyping was accomplished with oligonucleotide probes to identify 36 genital HPV types. The protocol for the collection of cervical specimens by nurse practitioners has been used on this cohort since 2002, in addition to the cell preparation and HPV laboratory analysis protocol, and has proven successful, given the high adequacy of samples for analysis. 22
Samples were considered HPV-positive if they were positive for any of the 36 HPV types and also positive for β-globin (assessed quality of DNA sample). Samples were considered HPV-negative if they were negative for all HPV types. HPV types were classified as either high-risk (HR) or low-risk (LR) based on their oncogenic potential. Probable and possible HR types were grouped with HR types that have more established evidence for oncogenic potential: 16, 18, 26, 31, 33, 34, 35, 39, 45, 51, 52, 53, 56, 58, 59, 66, 67, 68, 69, 70, 73, and 82. 25 Unclassified types were grouped with low-risk types: 6, 11, 40, 42, 54, 55, 61, 62, 71, 72, 81, 83, 84, and 89. HPV types were also classified into papillomavirus species groupings (α1, α3, α5, α6, α7, α8, α9, α10, α11, α13 and α15) to examine species-specific agreement measures. 26 Finally, species were grouped based on their preference for specific niches in the genital tract; α3 and α15 categorized as vaginal species and α5, α6, α7, α9, and α11 categorized as cervical species. 27
The prevalence of HPV infection and the associated exact binomial 95% confidence intervals were calculated for any HPV infection, and type-, species-, niche- and risk-specific HPV infection. Concordance between self- and provider-collected specimens for the detection of any HPV DNA was calculated to assess the percentage of test results that were in agreement. The concordance between the two sampling strategies was also determined for the detection of any HPV DNA, HR-HPV DNA, LR-HPV DNA, HPV-16 or HPV-18, type-, species- and niche-specific HPV DNA. Unweighted kappa statistics (κ) and the associated 95% confidence intervals were calculated to determine the percent agreement between the two collection methods above the agreement expected by chance. Values of kappa were categorized based on the amount of agreement they suggest as follows: κ > 0.75 represents excellent agreement; 0.40 < κ < 0.75 represents fair to good agreement; and κ < 0.40 represents poor agreement. 28 McNemar's test was used to assess the split of discordant pairs. It tested whether the proportion of samples classified as positive by self-collection and negative by provider-collection were unequal to the proportion of samples classified as negative by self-collection and positive by provider-collection. The non-parametric Wilcoxon's signed rank test was used to compare the number of HPV types found by self- versus provider-collected specimens, classified as any HPV DNA, risk- and niche-specific HPV infection. Analysis was conducted in SAS version 9.2 and statistical significance was set at 5% for all tests. Written informed consent was obtained from all study participants with a standardized consent form at study entry. Ethical approval for this study was obtained from the McGill Institutional Review Board.
RESULTS
Of the 109 eligible cohort participants who were approached to participate in this study, 93 women (85.3%) agreed to participate. Selected demographic characteristics are displayed in Table 1. HPV DNA laboratory results were available for 92 participants. One self-sample did not have a matching clinician-sample and two samples, both obtained through self-sampling were found to be inadequate for HPV analysis as they lacked β-globin amplification, leaving 89 sample pairs that could be compared.
Baseline characteristics of study participants (n = 93)
a At time of self-sampling study entry
b Median: 31.71, Range: 18–62
Figure 1 displays the overall HPV infection prevalence and the prevalence of HPV infection when HPV is grouped by risk, HPV-16 and HPV-18, and papillomavirus species. The overall prevalence and the prevalence of each papillomavirus species was higher in self-collected samples than provider-collected samples, but the 95% confidence intervals overlapped. The presence of any HPV DNA was detected in either single or multiple infections in 57.30% of the self-collected samples and 38.20% of the provider-collected samples. Of the 36 distinct HPV types that could be detected, 30 were detected by self-sampling and 29 were detected by provider-sampling.
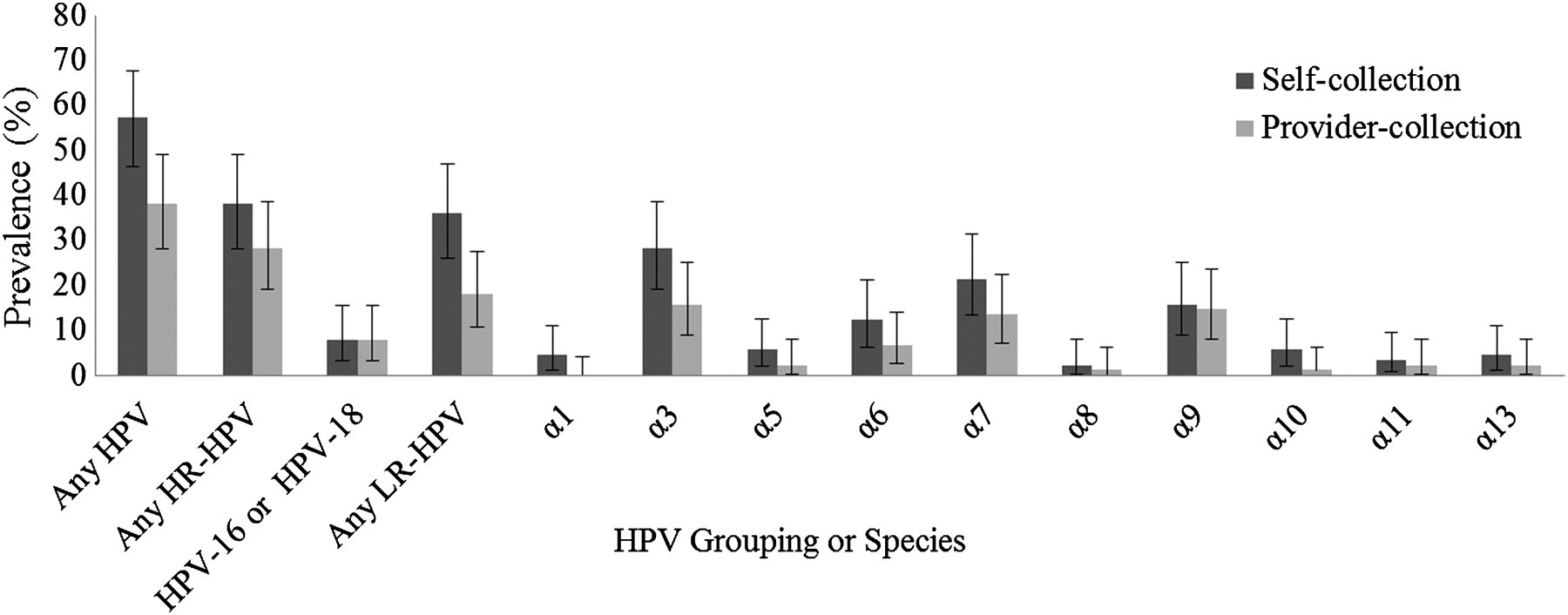
Prevalence estimates and associated 95% CI for each HPV type grouping and species by self-collection and provider-collection (n = 89)
The prevalence of HR-HPV DNA was 38.2% in self-collected samples and 28.1% in provider-collected samples. Of the 22 distinct HR-HPV types that were analysed in this study, 18 types were detected in the self-collected samples and 19 types were detected in the provider-collected samples. Figure 2 displays the type-specific prevalence of each HR and LR type detected by self-and provider-collection. The presence of HPV 16 or HPV 18 was detected in 7.9% of study participants by both sampling methods. The prevalence of LR-HPV DNA was 36.0% in self-collection samples and 18.0% in provider-collected samples. Of the 14 LR-HPV types that were analysed, 12 were detected in the self-collected samples and 10 were detected in the nurse-collected samples.
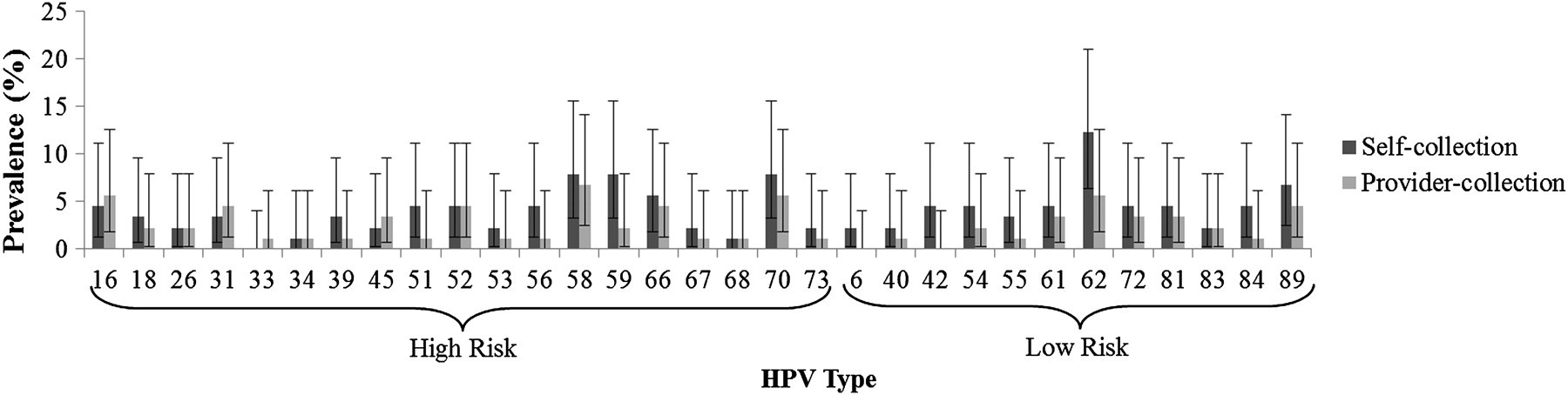
Prevalence estimates and associated 95% CI for HR-HPV and LR-HPV types detected by self-collection and provider-collection (n = 89)
The agreement between sampling methods for the detection of any HPV DNA was good, with a concordance of 76.4% (68 of 89 pairs) and an unweighted kappa statistic of 0.54 (95% CI: 0.39–0.70) (Table 2). The agreement of sampling methods for the detection of any HR-HPV DNA (85.4% agreement, κ [95% CI]: 0.67 [0.52–0.83]) and HPV16/18 (95.5% agreement, κ [95% CI]: 0.69 [0.40–0.98]) was higher than for the detection of any LR-HPV DNA (79.8% agreement, κ [95% CI]: 0.51 [0.33–0.69]). Among the discordant results for any HPV, HR-HPV DNA and LR-HPV DNA detection, the self-collected samples were more likely to be positive than the provider-collected samples (P value = 0.0002, 0.02 and 0.0002 respectively). The sampling methods were not found to be significantly different in the classification of samples as positive for HPV16/18 (P value = 1.00).
Agreement between self- and provider-collected samples for the detection of any HPV, any HR-HPV, any LR-HPV, HPV-16 or HPV-18, and by HPV species and type (n = 89)
a % of samples in agreement
b agreement above that excepted by chance
Species α9 (genotypes related to HPV-16) was detected with excellent agreement between sampling methods with a κ of 0.87 (95% CI: 0.72–1.00) and a concordance of 96.6%. The concordance between sampling methods for species α7 (genotypes related to HPV-18) was 87.6% (κ [95% CI]: 0.58 [0.36–0.80]). There was a concordance for the detection of species α3 (LR genotypes) of 85.4% (kappa of 0.58 [95% CI: 0.39–0.78]). Among the discordant pairs there was a significant difference in the detection rates of species α3 genotypes (vaginal niche preference) by the two sampling methods (P = 0.003), which is attributable to the higher proportion of self-collected samples found to be positive for these genotypes. All other papillomavirus species were also detected more frequently in self-collected samples compared with provider-collected samples, but these differences did not reach statistical significance.
Type-specific agreement is presented in Table 2, where it can be seen that type-specific concordance is high, ranging from 93% to 100%. The kappas comparing the detection of HPV-16 and HPV-18 between self-and provider-collection were 0.65 (95% CI: 0.28–1.00) and 0.80 (95% CI: 0.40–1.00), respectively. Positivity of the self-collected samples only was more frequently encountered than the opposite.
The number of types detected in the samples collected by self-sampling was greater than the number found by provider-sampling for any HPV and when HPV types were categorized by risk and niche preference (Table 3).
Mean and median number of HPV types detected per self-collected samples and provider-collected samples (n = 89)
DISCUSSION
The HPV prevalences determined by provider-sampling were similar to those reported for the cohort at baseline. A higher prevalence of any HPV, HR- and LR-HPV was detected by self-sampling than provider-sampling. We also observed higher species-specific prevalence in the self-collected specimens, but we did not find any significant differences in the HPV point prevalences found by the two sampling methods. Using McNemar's test, a systematic over-identification of any HPV, HR-HPV and LR-HPV by self-sampling among the discordant specimen pairs was found. Consistent with this higher systematic detection of HPV in self-collected samples, a marginally higher number of HPV types were detected in these samples.
Self-collected specimens were always collected before provider-collected specimens, as in the current literature. 6,7,9,29–31 Despite this, it is unlikely that a reduced recovery of exfoliated cells by provider-sampling is responsible for the higher detection of HPV in self-collected samples, as a randomized trial of sampling methods found that the detection of HR-HPV was not dependent on the order of sample collection and more frequent sampling has not been shown to influence detection rates of HPV. 32,33 The results of a cohort study of female university students, which suggest that newly acquired HPV infection may be detected in vulvovaginal sites before it is detected in the cervix, may partially explain the higher prevalence in cervicovaginal samples. 34
Although a higher detection of any-HPV, 6,29,30,35 HR-HPV types 6,8,29,30,36 and LR-HPV 29 with self-sampling has been found in the previous literature, it was an unexpected finding in this study. Our study protocol was one of the first self-sampling study protocols to use dry tubes to store self-collected swabs and liquid transport medium to store provider-collected samples. 37 A recent study investigating the use of dry swab samples found that HPV detection in the dry samples was slightly lower than in the wet samples (both samples collected by a clinician). 38 Our results suggest that HPV detection in dry samples is high and comparable with wet samples collected by nurse practitioners, and thus dry swabs may be useful in clinical practice and research settings.
The agreement in detection for any HPV 7,8,30,35 and HR-HPV 7,29,31,32,35 between self-collected and provider-collected samples was comparable, but slightly lower compared to studies with similar sampling techniques and laboratory protocol, which can be explained by the increased detection of HPV in self-collected samples. Petignat et al. 29 note that differences in agreement may exist between studies because women were given different sample collection instructions, as there is no standardized approach to self-sampling. The lower agreement found for the detection of LR-types (κ: 0.51) compared with HR-types (κ: 0.67), which was found in this study and others, was due to a higher detection of LR-HPV types in the self-collected samples. 6,29,35 Species α3 and α15, which contain low-risk types, have been shown to preferentially infect the keratinized tissue found in the vagina, whereas high-risk types have been shown to infect the whole genital tract equally. 27 Our results agree with this research, as types in species α3 were less likely to be detected in the cervical samples.
Although only infections in the transformation zone of the cervix lead to cervical cancer, the epithelial surfaces of both the vagina and cervix are prone to infection by HPV. It is possible that many of the HR-HPV infections detected in self-sampling are vaginal and may never infect the cervix, thereby reducing the specificity of self-sampling and leading to unnecessary follow-up of false-positives. However, it seems that self-sampling detects a pattern of infection that resembles the one in the cervix, as the type-specific agreement between methods was high and samples were concordant by type. Additionally, self-sampling is as good as provider-sampling for the detection of HPV-16, HPV-18, species α7 and species α9. As self-sampling has a high recovery of HR-HPV, is comparable with provider-sampling and has been shown to be an acceptable form of sampling, 39 we can conclude that self-sampling with dry storage and transport could be a good cervical cancer screening alternative for Inuit women in Nunavik who have traditionally avoided speculum examination.
The main limitation of this study is the small sample size, given that paired HPV results were only available for 89 women. Few cytological abnormalities were found in this small sample, so we were unable to compare the sensitivity and specificity of self-collected samples to detect precursor lesions. Using disease endpoints would have been ideal, but it was felt that the use of virological endpoints would demonstrate that self-samples could be adequately collected in this population (98% of samples were adequate) and act as first step to determine the feasibility and practicality of self-sampling in this population. Although laboratory personnel could not be blinded to sampling method, it should not be considered a serious limitation as the linear array test is an automated objective molecular test.
Another limitation of our study was that although the samples did not need to be refrigerated or transported in dry ice, 37 self- and provider-obtained samples were put with all other biological samples collected in the clinics, following the protocol of refrigeration that is used at the Nunavik health centres. This was done to simplify the transport process from these remote communities to a large urban centre for analysis and to avoid overburdening staff, as research in the Northern context greatly depends on their continued collaboration and support. Additionally, there is a high turnover rate of healthcare staff in these communities and so it was thought that a simplified research protocol would have a greater chance of sustainability and uptake.
Despite the usefulness of a mail-based system of sampling kit distribution and return for the purposes of screening and research alike, we did not aim to assess the adequacy of samples returned through this type of system. In the framework of the North, it was thought that a clinic-based system would be more conducive to increasing sample return given the proximity of residents to the health centres in each community. The adequacy of dry self-collected samples collected outside the clinic would need to be investigated before they are used in a study or screening protocol. Additionally, nurse practitioners explained to the study participants how to collect a sample and so the proportion of adequate samples may be lower when collected outside the clinic in a mail-return protocol.
The strengths of this study include a standardized protocol with PCR, so we were able to describe type-specific and species-specific prevalence and agreement. This is one of the few studies to report agreement on LR-HPV, which although may be less important for screening, good detection of LR-HPV is vital to studies on the natural history and transmission of HPV. 29,35 In our protocol, Dacron swabs were used for both self-collection and provider-collection, increasing the comparability of results from each collection site. The Dacron swab is particularly useful as a self-sampling device as it is easy to manipulate, 35 does not require any other devices for collection, 40 can be easily processed in the same manner as clinician-obtained samples 35 and has a faster processing time compared with other methods. 32 To ensure that differences in detection rates are not due to transient infections we collected both samples on the same day, unlike some studies. 30,35 Although self-sampling has been studied in a variety of other populations, this study is the first to look at the comparability of self-sampling in Inuit women and one of the first to use dry-swabs. Given that this population is at high risk for HPV and cervical cancer, research on novel screening methods is highly relevant. Further research should examine the logistical feasibility of implementing self-sampling into the screening programme for this population and determining if it will increase uptake.
DECLARATION OF INTERESTS
Franco has served as consultant to Roche, Qiagen, and Gen-Probe. F Coutlée has served as consultant for Qiagen and has a research project funded by Roche Diagnostics.
Footnotes
ACKNOWLEDGEMENTS
Funding for this research was provided in part by the Canadian Cancer Society and a Team Grant on HPV Infection and Associated Diseases from the Canadian Institutes of Health Research (CIHR). H Cerigo was supported by a student fellowship from CIHR. P Brassard was supported by a clinician scientist career award from the Fonds de Recherche en Santé du Quebec (FRSQ). The authors thank the participating communities, Tulattavik Health Centre, collaborating nurse practitioners, and the Nunavik Regional Board of Health and Social Services for making this research possible.
