Abstract
Objective
To estimate the pre-eclampsia screening performance of PAPP-A (pregnancy-associated plasma protein-A) and ADAM12 (A Disintegrin And Metalloprotease 12) in the early second trimester of pregnancy.
Methods
Stored frozen serum samples from a previously published nested case-control study comprising 77 women who developed pre-eclampsia and 224 unaffected controls were thawed and assayed for PAPP-A and ADAM12. Levels were converted into multiple of the unaffected median (MoM) values.
Results
Neither PAPP-A or ADAM12 were significantly different in women who developed pre-eclamspia compared with unaffected controls (PAPP-A: 0.97 MoM [95% confidence interval 0.73 to 1.25], ADAM12: 1.01 MoM [0.91 to 1.13]).
Conclusion
Our results do not support the use of PAPP-A or ADAM12 in early second trimester antenatal screening for pre-eclampsia
INTRODUCTION
Pre-eclampsia remains an important cause of fetal loss, stillbirth and maternal morbidity and mortality. Screening women in the first half of pregnancy to identify those who are at a high risk of developing pre-eclampsia is useful if focusing research into methods of prevention and in identifying women who need special antenatal care.
PAPP-A (pregnancy-associated plasma protein-A), measured in the late
ADAM12 (A Disintegrin And Metalloprotease 12), measured in the late
The suggestion that PAPP-A and ADAM12 might be useful in screening for pre-eclamspia in the early second trimester of pregnancy prompted us to investigate this further. We used a case-control set of early second trimester serum samples previously used to determine the usefulness of the Down's syndrome Quadruple markers, endoglin and placental growth factor (PlGF) in screening for pre-eclampsia.
METHODS
We used a previously described dataset and stored serum samples relating to clinically confirmed pre-eclampsia (either (i) eclamptic fits, and/or (ii) raised blood pressure after 20 weeks, but not before, based on diastolic blood pressure ≥90 mmHg on two occasions at least one day apart, together with urinary proteinuria based on 24h urinary protein excretion ≥500 mg or at least two ‘abnormal’ spot urine tests classified as at least ‘ ++’ on different days) and five matched controls for each case for which Ethics Committee approval was obtained. 14 Second trimester serum markers for Down's syndrome had been measured as part of a previous study, and on grounds of economy, three of the five controls matched to each case were randomly selected for a second study in which endoglin and PlGF were measured. This same subset of pregnancies was used in this analysis. Serum samples were retrieved, thawed, and assayed blind to whether they were a case or not for PAPP-A and ADAM12 using time-resolved fluoro-immunoassay (AutoDELFIA Perkin ElmerTM [Life Sciences, USA]) at a 1 in 4 dilution. There were sufficient sera available for assay in 77 pre-eclampsia pregnancies, and 224 of the controls matched to these 77. The inter-assay coefficient of variation was 9.7% for ADAM12 and 9.4% for PAPP-A.
PAPP-A and ADAM12 concentrations were expressed as multiples of the median (MoM) in unaffected pregnancies of the same gestational age. This was done by performing a regression of the log median marker concentration against median gestational age in each completed week of gestation (weighted by the number of women in each week). Each observed concentration was divided by the regressed (i.e. expected) concentration for the same gestational age to obtain the MoM value. MoM values were then adjusted for maternal weight by performing a regression of the log median MoM values against median weight in categories of weight (weighted by the number of women in category) and dividing MoM values by the regressed value for the same weight. PAPP-A and ADAM12 MoM values in pre-eclampsia and unaffected pregnancies were compared for statistical significance using the Wilcoxon rank sum test.
RESULTS
Table 1 shows characteristics of women who developed pre-eclampsia and unaffected pregnancies. Figure 1 shows the PAPP-A levels (Figure 1a) and ADAM12 levels (Figure 1b) in the women who developed pre-eclampsia in relation to the median level in unaffected pregnancies, and lines indicating twice and half the median. In unaffected pregnancies PAPP-A increased by 22% per week of gestation (P < 0.001) and ADAM12 by 14% per week (P < 0.001). There is no indication that either the median PAPP-A or ADAM12 levels tended to be high or low in women who developed pre-eclampsia; 38 affected pregnancies were above the median, and 39 below the median for both. After converting into MoM values and adjusting for maternal weight the median PAPP-A in pregnancies that developed pre-eclampsia was 0.97 MoM (95% confidence interval 0.73 to 1.25) and the median ADAM12 was 1.01 MoM (0.91 to 1.13). Neither were statistically significantly different from the median MoM values in unaffected pregnancies (1.0; P = 0.774 and P = 0.597, respectively). There was no statistically significant difference in the median PAPP-A or ADAM12 MoM values in cases in which delivery was after 35 weeks' gestation and cases where delivery was at 36 weeks' gestation (0.79 and 1.04 respectively for PAPP-A, P = 0.558; 1.07 and 0.95 for ADAM12, P = 0.314). Similarly, there were no statistically significant differences in MoM values between parous and nulliparous cases (0.92 and 1.04, respectively for PAPP-A, P = 0.836; 1.07 and 0.99 for ADAM12, P = 0.607).
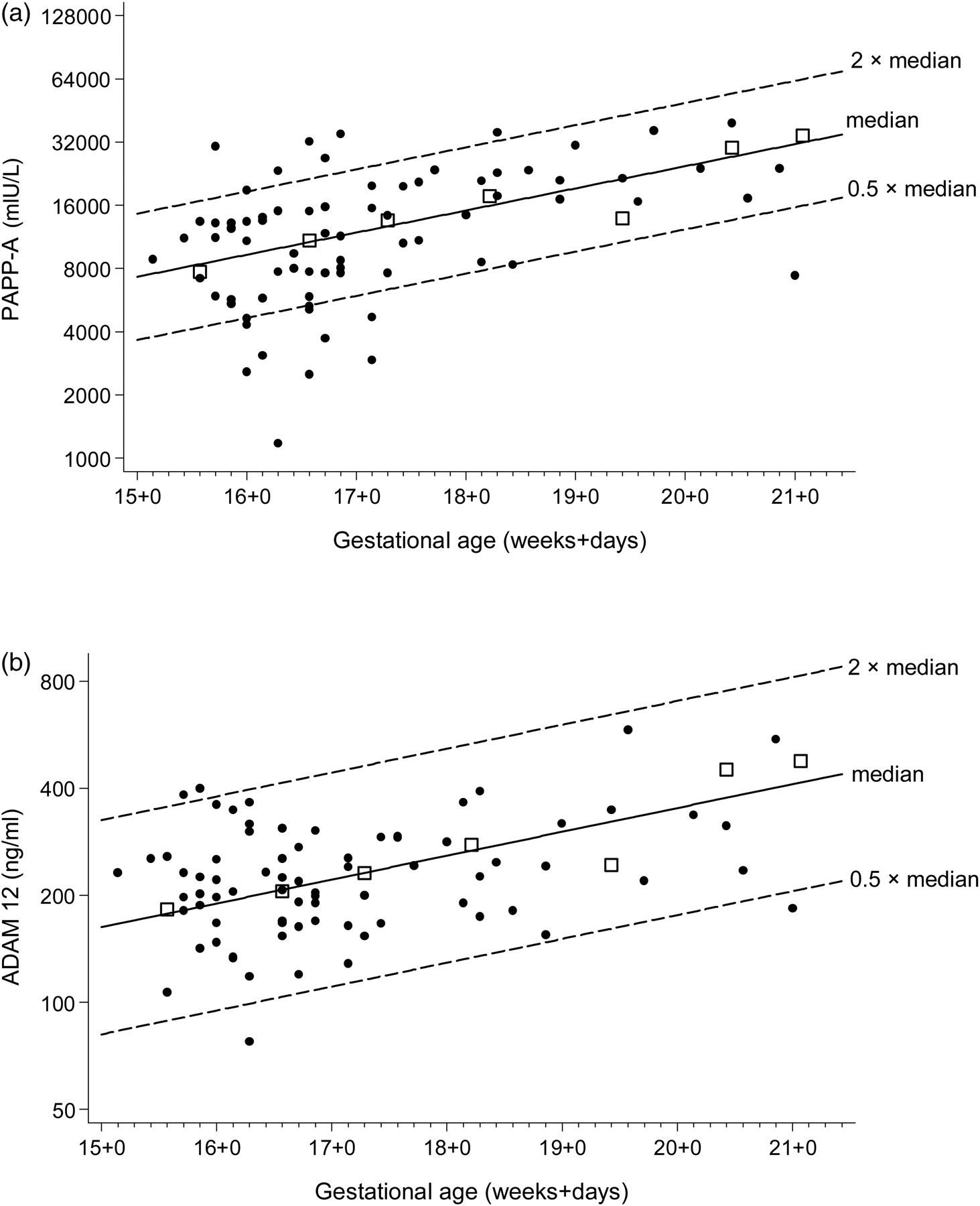
PAPP-A (a) and ADAM12 (b) according to gestational age in pregnancies that develop pre-eclampsia and unaffected pregnancies. Squares are weekly medians in unaffected pregnancies, dots are affected pregnancies
Maternal characteristics of pre-eclamptic and unaffected pregnancies
EDD: expected date of delivery
DISCUSSION
This is the largest study, based on 77 affected pregnancies, of early second trimester measurements of PAPP-A and ADAM12 in pregnancies that developed pre-eclamspia. The results indicate that measurements of PAPP-A and ADAM12 in the early second trimester of pregnancy are not useful in screening for the development of pre-eclampsia. The results differ from those of D'Anna et al. (2011) based on 40 affected pregnancies with a median PAPP-A MoM of 0.30 and a median ADAM12 MoM of 0.28 in pregnancies that developed pre-eclampsia (both P < 0.001) 10 and with those of Bersinger et al. (2004) with median PAPP-A levels in 28 affected pregnancies about 60% lower than in unaffected controls (P < 0.0001). 9
We have no explanation for why, unlike the results of D'Anna et al. and Bersinger et al., our study shows no material difference in PAPP-A and ADAM12 in pre-eclampsia and unaffected pregnancies. The explanation is unlikely to be due to using frozen serum samples because while the samples used in the study went through three freeze-thaw cycles, Cowans et al. showed that first trimester PAPP-A levels are not significantly altered following six freeze-thaw cycles 15 and that first trimester ADAM12 levels are not altered following three freeze-thaw cycles. 16 Even though there are differences between studies in the definition of pre-eclampsia, our definition was at least as strict as others. Consequently, if anything, our results would have shown a larger effect if one were present. Different assays are unlikely to be an explanation, since within each study cases and controls were tested using the same assay. Other differences between studies such as the exclusion of smokers and diabetics in the D'Anna study 10 do not provide an explanation because these exclusions applied to both cases and controls. The discrepancy between studies can only be resolved with evidence provided from further studies.
We conclude, on the balance of the evidence, that the use of PAPP-A and ADAM12 is probably of no value in early second trimester screening for the development of pre-eclampsia.
