Abstract
Objective
To assess the feasibility and acceptability of randomizing the phased introduction of the extension of the invited age range in the National Health Service (NHS) Breast Screening Programme in England from 50-70 to 47-73 years.
Setting
Six volunteer breast screening units (BSUs) in England.
Methods
Cluster-randomized trial of invitation versus no invitation for breast screening. Study participants: women aged 47-49 and 71-73 years in screening batches randomized between 1 June 2009 and 31 May 2010. Outcomes: workload, screening uptake among women invited, self-referrals among women not invited, and screening outcomes among women invited.
Results
A total of 312 screening batches (clusters) were randomized including 60,708 women. Screening uptake was 63% in women aged 47-49 and 62% in women aged 71-73. Those who attended screening in the younger age group were more likely to be recalled for assessment than older attendees (7.5% vs. 3.0%) but less likely to be diagnosed with breast cancer (0.5% vs. 1.1%). Among women not invited, 0.2% of those aged 47-49 and 6.8% of those aged 71-73 self-referred for screening. Despite the extra workload BSUs largely coped although there was some slippage in round lengths and other targets.
Conclusion
No major problems of feasibility or acceptability of randomization were found. This pilot study has informed the randomized phasing-in of the age extension across the whole of England.
Introduction
In 2007 the Cancer Reform Strategy (CRS) 5 announced that the age range for routine three-yearly screening in England would be extended from 50-70 to 47-73 years, i.e. providing one extra screen at age 47-49 and one extra screen at age 71-73 years. As, however, resources and capacity did not allow immediate extension of the age range across the whole country at the same time, the plan was to phase the age extension in over a number of years. This meant that, eventually, women would get 2 extra invitations for screening in their lifetime, the first one before age 50 and the last after age 70. In January 2011 it was announced in Improving Outcomes: A Strategy for Cancer 6 that phasing-in would not be fully implemented until 2016 at the earliest.
No major trial has looked at the added value of one extra screening invitation within an existing breast screening programme. The CRS stated that ‘the necessary phasing in of this expansion will be carefully considered to ensure that the most useful epidemiological data can be gathered to inform future decisions about the programme.’ Because of the excellent, and developing, NHS record linkage possibilities the phased age extension provides a unique opportunity to assess the effects of adding one extra screening invitation at younger ages and, separately, the effects of adding one extra invitation at older ages to the existing programme.
As there are sufficient resources during the phasing in for just some, but not all, women to be offered an extra invitation, then the best way of getting reliable evidence is to determine at random who will get an additional invitation, and then to use linkage of routine NHS medical records to assess the effects of an extra screen on a range of outcomes. The findings have the potential to inform future breast cancer screening policy both in the UK and elsewhere. To answer questions about the feasibility and acceptability of randomization, including its effects on workload, a pilot study was undertaken. We describe its conduct and findings.
Methods
Six volunteer BSUs were considered for inclusion in the study: Bolton Breast Unit (Royal Bolton Hospital NHS Foundation Trust); Cambridge Breast Unit (Cambridge University Hospitals NHS Foundation Trust); Warwickshire, Solihull & Coventry Breast Screening Service (University Hospitals Coventry and Warwickshire NHS Trust); The Jarvis Breast Screening Centre, Guildford (Surrey PCT); South East London (Kings) Breast Screening Service (King's College Hospital NHS Foundation Trust), and Manchester Breast Screening Service (University Hospital of South Manchester NHS Foundation Trust). Cambridge was withdrawn at the start of the study, however, when it became apparent that the method used by them to create screening invitation batches was not compatible with the randomization method. Five units remained.
As part of the operation of the routine breast screening programme, the National Health Application and Infrastructure Services ‘Exeter’ system regularly creates screening invitation batches for each BSU. A batch contains women aged 50-70 years who live in the same geographical locality and is used by the local BSU to invite those women for screening, generally mailing out invitations simultaneously to all women in a batch.
For this study batches now included women aged 47-73 years rather than just the routine 50-70 age group. The study was randomized by cluster. A flowchart of the phases of the pilot study is shown in Figure 1. For each batch it was determined randomly whether to invite for screening the women aged 47-49 years, or the women aged 71-73 years, in addition to all women aged 50-70 years. The random allocation of batches by a specially-written computer programme was done with equal (50/50) probability and no stratification. Women aged 50-70 were not study participants and were unaffected by the randomization process; their age group was already eligible for routine screening, and their invitations for screening continued as normal regardless of the age extension. Study participants were only the women aged 47-49 and 71-73 years in the randomized batches, where, in a given batch, one of the age groups was randomized to be invited for screening (randomized in) and the other age group was randomized not to be invited for screening (randomized out). Each participant entered the study on the date when the screening batch she was in was created and randomized. Women aged 4749 years in a batch where their age group was randomized not to be invited for screening could request to be screened. Women over 70 are already able to request screening every three years. Other than randomization, all other aspects of screening were conducted in exactly the same way as is normal in the NHSBSP, following all NHSBSP routine permissions and procedures.
1
Flow diagram of the progress through the stages of the age extension pilot study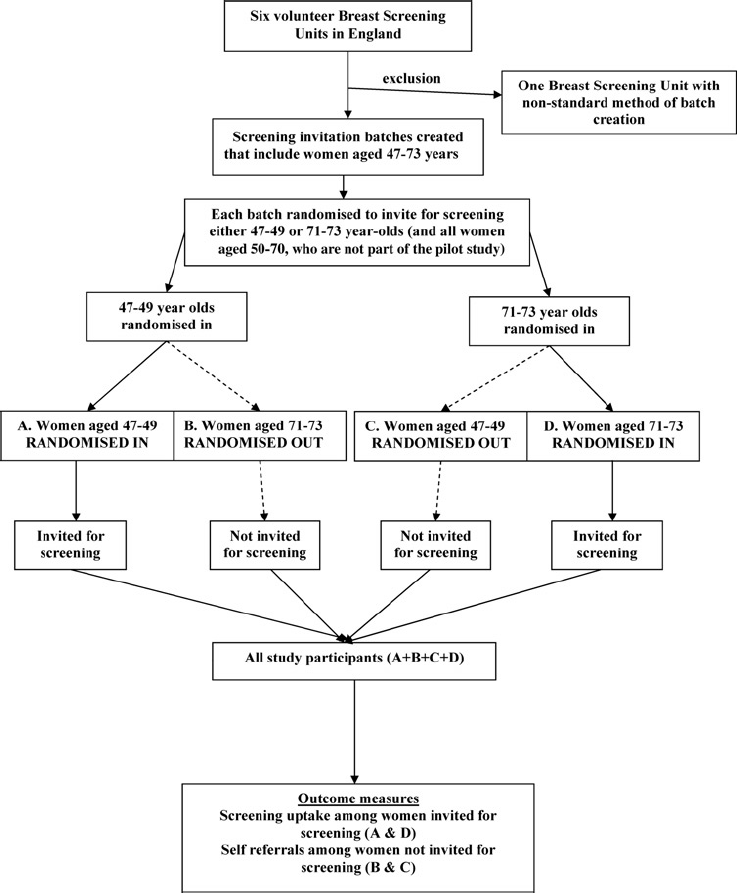
The pilot study included all screening batches created and randomized during the twelve-month period 1 June 200931 May 2010. Randomization of batches started in early June in Manchester, Coventry and Kings BSUs, in early July in Bolton and in August in Guildford. The delay in two sites starting randomization was due to the time required to obtain local management (R&D) approval.
Phone and issues logs, and suggested responses to common phone enquiries, were developed for use in the BSUs.
Data on study participants were extracted from the National Breast Screening System (NBSS) of each participating BSU. No patient-identifiable data were included. The data analysed here were downloaded from each unit in March 2011 allowing sufficient time from the date of batch creation for women to be invited for screening, attend their appointment, and (where applicable) to have complete follow-up data on the NBSS. Counts of self-referrals were also extracted. BSUs logged information on phone calls relating to the age extension, and any other workload issues.
Outcome measures for the pilot study included screening uptake among women invited for screening; number of self-referrals among women not invited for screening; and workload associated with randomization. Proportions of women invited for screening who attended were tabulated, as were reasons for not attending. Numbers of self-referrals were also tabulated. Qualitative assessment was made of comments and opinions of screening unit staff on the workload associated with randomization, and of women on the acceptability of the process.
Ethical approval for the pilot study was received from Ealing and West London Research Ethics Committee March 2009 (REC Reference 09/H0710/2). In addition, BSUs were required to obtain local management (R&D) approval before starting to randomize batches. Subsequently further approval was obtained from the Research Ethics Committee and from the National Information Governance Board Ethics & Confidentiality Committee to proceed to the main trial, which is examining a range of outcomes of the age extension, and to include data from the pilot study in the main trial. Some results on screening outcome in the pilot study population are included here.
Results
Study participants
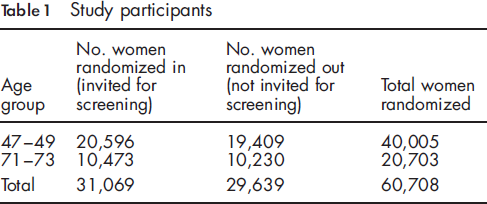
Number of study participants and mean number per batch, by breast screening unit
The mean number of women aged 47-49 or 71-73 randomized per batch was 195 although there was variation across breast screening units (Table 2). Half of the 195 randomized per batch (98) were randomized for screening invitation. The average number aged 50-70 per batch was 642, so, with an additional 98, the batch size increased by 15% to 740.
Screening uptake and non-attendance
Status on 31 March 2011 of women randomized for screening invitation, by breast screening unit (% (n))
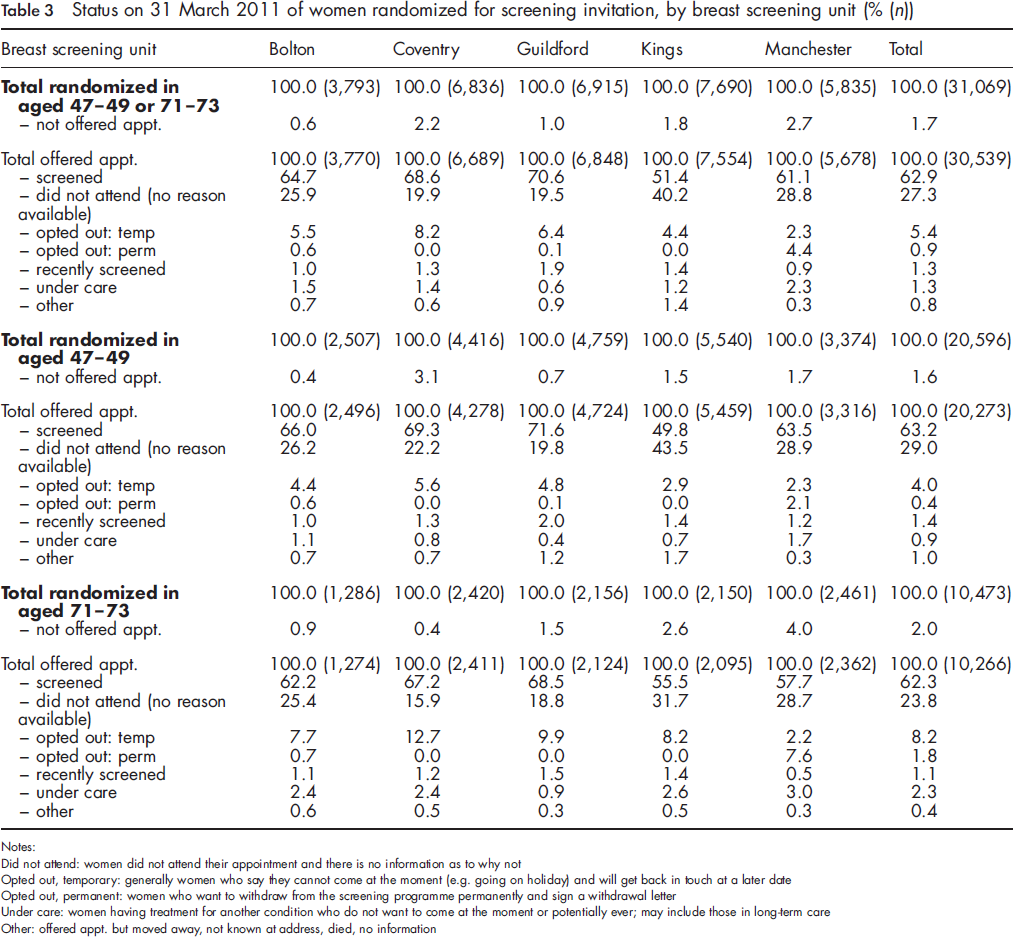
Notes:
Did not attend: women did not attend their appointment and there is no information as to why not
Opted out, temporary: generally women who say they cannot come at the moment (e.g. going on holiday) and will get back in touch at a later date
Opted out, permanent: women who want to withdraw from the screening programme permanently and sign a withdrawal letter
Under care: women having treatment for another condition who do not want to come at the moment or potentially ever; may include those in long-term care
Other: offered appt. but moved away, not known at address, died, no information
The percentage who were screened varied across breast screening units from 51-71% of women offered an appointment. Uptake was higher in the younger as compared with the older age group (by 2-6 percentage points) in all but the inner London Kings BSU. The percentage not attending (with no information on why) varied from 20-40% with, unsurprisingly, the lowest percentage occurring in the units with the highest uptake. In three units the percentage not attending (with no information on why) was similar for younger and older women, while in the other two units the percentage was considerably higher among younger women (22% 47-49 vs. 16% 71-73; and 44% vs. 32%).
Self-referrals
Over the 12 months of the study only 31 women (0.2%) aged 47-49 who had been randomized out requested to be screened; no BSU had more than ten such women. A considerably higher number of women aged 71-73 (697, 6.8%) requested screening. These requests for screening in older women would have been expected in the absence of the pilot study.
Workload and other service issues
In two BSUs obtaining local management approval was a lengthy process. Other workload issues mentioned were: some GPs were not aware of the pilot study running in their area, and on occasion discouraged women outside the 50-70 age range from attending; one unit had problems enclosing the study patient information leaflet with screening invitations as the way it was folded meant envelopes now had to be filled manually rather than the process being fully automated as before; some sites mentioned a slightly longer mammogram reading time than usual in women aged 47-49, but others said reading times were no different from usual first screens. Extra assessment clinics were required as a consequence of the larger number of women being invited. Sites found the completion of the phone logs required for this pilot study time consuming.
Phone calls
Relatively few calls were logged from women in the new age groups, or from women aged 50-70 with queries relating to the age extension. As happens in all age groups, most calls were to change appointment. Some calls from women aged 47-49 indicated new concerns, for example, not wanting to be screened so young, suitability of screening women who were pregnant, breastfeeding or had breast implants. Some in this age group were puzzled as to why they had been invited, believing the age range to be 5070, and others as to why they had not been invited when friends of a similar age had been. Few calls were received from women aged 71-73. Some aged 50-70 were confused by the patient information leaflet which left them unsure why they had been invited; some in their early 50s were upset not yet to have received their first invitation when some women in their late 40s had already been invited.
Screening outcomes
Screening outcomes of women screened
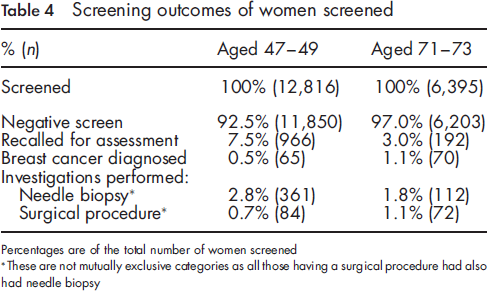
Percentages are of the total number of women screened
These are not mutually exclusive categories as all those having a surgical procedure had also had needle biopsy
Discussion
The randomized phasing-in of the age extension within the NHSBSP was found to be largely acceptable and practical. There had been some concern that many younger women randomized not to be invited for screening would request screening, and that the extra workload associated with randomization would be difficult to manage. There was no indication that the first issue materialized, with only 0.2% of women aged 47-49 requesting screening. It is, however, possible that self-referrals may change over time as general awareness of the age extension grows. The concern about workload was less straightforward. As the extension of the age range is government policy, and therefore is proceeding regardless of the randomization, the only workload issues of relevance here were those associated with randomization. The main such issue is that breast screening units knew only after creating and randomizing a batch whether the younger or older women were to be invited for screening. Because the number of 47-49 year olds in a batch is roughly twice the number of 71-73 year olds, and the two age groups have differing needs (for example, in the proportion recalled for assessment), which age group is randomized to be invited for screening has important workload implications. However, the units managed with no major problems. All units met the national standard to send results to at least 90% of eligible women within two weeks of screening. Three units maintained their round lengths (interval between previous screening and next first offered appointment), while two units slipped on this in one or two three-month periods. Most units failed in at least some three-month periods to achieve a waiting time of less than three weeks from screening to attending assessment for 90% of eligible women; several units reported that this was not related to the age extension but had been a previously existing problem.
The higher recall for assessment rate, higher needle biopsy rate and lower breast cancer diagnosis rate seen in the 47-49 age group, as compared with the 71-73 age group, was expected. A similar pattern is observed within the 50-70 age range routinely invited for screening in the NHSBSP, with higher recall and needle biopsy rates and a lower cancer diagnosis rate in women aged 50-52 compared with those aged 65-70. 7
On the basis of anecdotal feedback, comments, and phone calls, women in both age groups were generally glad to be invited for an extra screen, and no major problems were evident in terms of the acceptability of randomization. Some women aged 47-49 said they were too young to be screened. It is of concern that screening uptake was only 63% in women aged 47-49; 29% of this age group did not attend (with no information on the reason why), 4% opted out temporarily, 1.4% had been recently screened, 0.9% were under care, and 0.4% had opted out permanently. The inner London unit had a particularly high percentage not attending (with no information on why). If this unit is excluded from the calculations overall uptake in women aged 47-49 in the remaining four units was 68% and non-attendance (with no information why) 23%. The uptake across the five screening units, which ranged from 50-72% in women aged 47-49, was 1-12 percentage points lower than the uptake in women aged 50-52 invited routinely in these units in 2009/10 (personal communication with pilot BSUs). It is notable that 62% of the women aged 71-73 who were offered an appointment attended for screening, with the proportion varying between units from 56-69%. This is lower by 3-12 percentage points than the uptake of women invited routinely at ages 65-70 in these units. The uptake of 62% among women aged 71-73 in our study demonstrates the difference between actively inviting women in this age group and simply telling them they can request screening once they are over 70, as only 7% of the women aged 71-73 who were randomized out requested to be screened. This figure compares with a recent estimate that 11% of women aged 71-74 in England self-referred for breast screening 2005-08. 8 The finding of a low rate of self-referrals is particularly important given the increase in breast cancer incidence with age, and the general lack of knowledge among women of this increasing risk. 9
The BSUs volunteered to participate in this study, thereby indicating their willingness to pioneer the age extension and to take on a greater workload in advance of other sites; they are thus unlikely to be representative of all BSUs nationally. However, importantly, they contributed a range of experiences to the study as they varied in size, and included inner city and ethnically diverse areas as well as smaller and more prosperous towns.
Randomized phasing-in of the breast screening age extension is now proceeding across England. 10 The experience of the pilot study has helped the full roll out of the age extension which began in 2010, ensuring problems could be anticipated and ironed out in advance, and guidance provided. Drawing on the pilot study, a guide to implementing the age extension was distributed to all breast screening units in England that were preparing to extend the screening age range. 11 Implementation of the randomized phasing-in of the age extension of screening was discussed at regional meetings of NHS breast screening units in England held during the summer of 2010.
Conclusions
No significant problems of feasibility or acceptability of randomization were demonstrated. Useful lessons were learnt which have informed the planning and implementation of randomized phasing-in of the age extension across the whole of England.
Footnotes
Acknowledgements
This study would not have been possible without the input of all staff in each of the pilot breast screening units.
