Abstract
Magstream and OC Sensor quantitative immunochemical faecal occult blood tests (IFOBT) have shown better performances than guaiac (G) tests in colorectal cancer screening, however Magstream and OC Sensor have never been compared. We hypothesized that similar performances could be observed with Magstream and OC Sensors, provided a similar cut-off (expressed in concentration of haemoglobin in the stools) is used. We performed a literature-based indirect comparison between these tests, taking into account the cut-off, the number of samples, and the way they were combined (I2+: at least one positive sample of 2; I2++: both positive samples; I1: only one sample). Six studies conducted in general average-risk populations were included in this review. For each [test]*[cut-off], positivity rate (PR) decreased and predictive positive value (PPV) increased from I2+ to I1 and I2++. For similar PR, PPV with OC Sensor was greater than with Magstream. This could be due to factors other than the test, because PPVs associated with GFOBT in studies evaluating OC Sensor were greater than PPVs associated with GFOBT in the study evaluating Magstream. Direct comparison between Magstream and OC Sensor is needed to confirm the suspected superiority of OC Sensor.
Introduction
Material and Methods
A PubMed literature search was performed on 01/05/2011, using the keywords ‘immunochemical test and (threshold or cut-off) and colorectal cancer’, limited to articles in English, and excluding reviews and comments. Of the 38 articles retrieved, seven independent articles evaluated at least the performance of OC Sensor or Magstream in general average-risk populations, taking into account the cut-off and number of samples. 3 9 Of these one article was excluded because of a high positivity rate associated with GFOBT, suggesting a specific study population. 9 Details of the study characteristics and selection processes are given in the appendices.
We compared the positivity rates (PR), and positive predictive values (PPV) of FOBTs for detection of invasive cancers or advanced neoplasias (invasive cancers or high-risk adenomas) according to cut-off and number of samples.
When not published, PRs were derived from colonoscopy rates and global adherence to colonoscopy for a positive FOBT. Only the first rounds of screening results were included. Analysis considering the IFOBT as positive if both samples were positive was denoted as I2++, if at least one of two samples was positive as I2+, and when only one sample was performed as I1.
Results
Four studies evaluated the OC Sensor,3,4,6,7 and two the Magstream.5, 8 Three studies also used the GFOBT Hemoccult II. 3 5 Subjects included were aged 50-69 in the two Italian studies6, 7 and 50-74 in the two French studies and the two Dutch studies.3-5,8 Sex ratio was similar in all studies. Colonoscopy rates in case of a positive test varied from 79% to 94%.
For both Magstream and OC Sensor IFOBTs, using two samples led to a decrease in PR in I2++ analyses, but an increase in PR in I2+ analyses. The opposite was observed for PPV. Figures 1a and 1b plot the PPVs against the PR for detection of invasive cancers or advanced neoplasias. For the same PR, the PPVs for detection of invasive cancers for the Magstream in one study
5
were smaller than that of the OC Sensor, but were similar in another study.
8
In both studies the PPVs of Magstream for detection of advanced neoplasias were smaller than for the OC Sensor in all studies except one study conducted in Italy.
6
For both Magstream and OC Sensor, continuations between points for I1, I2+ or I2++ analyses were observed. The fitted curves showed slightly better performances (greater PPV for the same PR) with OC Sensor than Magstream.4,7
Comparison of efficiency of screening programmes according to test, number of samples and cut-off (a) Invasive cancers (b) Advanced neoplasias and (c) Figure legend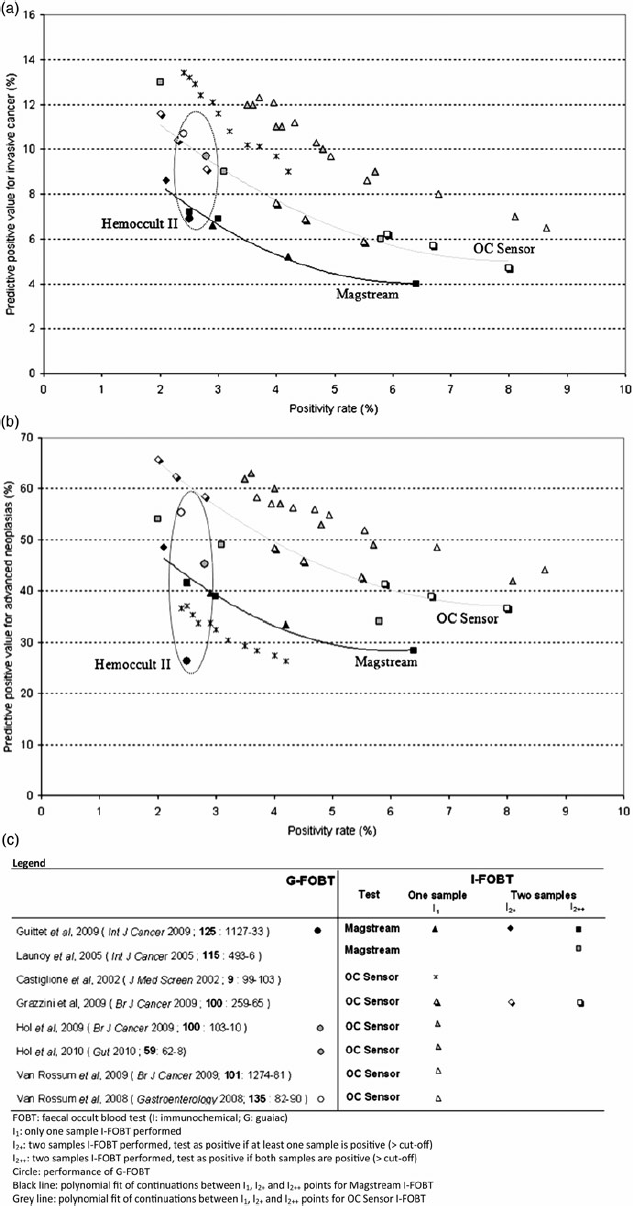
Discussion
This literature-based indirect comparison showed slightly better performances with OC Sensor than Magstream IFOBTs, especially for detection of advanced neoplasias. For each test, our results suggest that one-sample IFOBT (I1) could provide similar performances to two-sample IFOBT (I2+) in average-risk population provided that a different cut-off is chosen. This is in accordance with results of Park et al., 9 and is of importance because using only one sampling could improve participation and reduce costs.
Our approach does have several limits. First, the PR and PPV depend on the diagnostic accuracy of the test, but also on the prevalence of targeted lesions. Whilst colorectal cancer incidence is similar in the countries involved in these studies, prevalence of high-risk adenomas is unknown. Difference in prevalence could additionally result from difference in the definition of lesions. Not all studies included adenomas with villous patterns in the definition of advanced neoplasias, however, small adenomas with villous patterns represent only a minor portion of high-risk adenomas detected by FOBTs. 3 Furthermore, considerable variations are found in both colonoscopy results 10 and pathological assessment of advanced neoplasias. 11
It is not possible to determine which of the differences in global indicators of efficacy (such as PR or PPV) observed between tests were due to factors other than the performance of the test itself. Indeed considerable variations are described within one test, for both GFOBT and IFOBT. For example, in France, the PR of the GFOBT programme varied between departments from 1.9% to 3.7% in the first round. The PPV for detection of colorectal cancers (including intramucosal cancers) varied between 6.5% and 16.6%. 12 Similar variations were observed using the OC Sensor test in one Italian study, where four regions were included; PPV varied between 4.5% and 5.5% for detection of invasive cancers, and between 29.9% and 37.1% for detection of advanced neoplasias. 6 Differences between IFOBTs could be due to unmeasured confounding factors. Performance of GFOBT was higher in the studies evaluating OC Sensor in the Netherlands,3, 4 than in the study evaluating Magstream in France. 5 In addition, performance of the IFOBTs is affected by the delay between sampling and analysis and the temperature of storage. 13
The superiority of IFOBT over GFOBT is now proven, and the next step should be the comparison between IFOBTs. Our review demonstrates that indirect comparison between the crude performances lacks reliability. Direct comparisons of the performances of quantitative immunochemical FOBTs are necessary, using a paired design in which all subjects would undergo both IFOBTs, or a comparison of two groups of subjects with random allocation of IFOBTs. These comparisons should be designed and conducted to ensure that there is no confounding due to the analysis of tests (temperature and delay of storage), and standardize the definition of targeted lesions, including reproducibility of histological examination.
Footnotes
Appendix
Selection process

| 1: | Berchi C, Guittet L, Bouvier V, et al. Cost-effectiveness analysis of the optimal threshold of an automated immunochemical test for colorectal cancer screening: performances of immunochemical colorectal cancer screening. Int J Technol Assess Health Care 2010; |
Excluded, same data as study #12 |
| 2: | Castiglione G, Grazzini G, Miccinesi G, et al. Basic variables at different positivity thresholds of a quantitative immunochemical test for faecal occult blood. J Med Screen 2002; |
Included |
| 3: | Castiglione G, Zappa M, Grazzini G, et al. Screening for colorectal cancer by faecal occult blood test: comparison of immunochemical tests. J Med Screen 2000; |
Excluded, data also included in study #2 |
| 4: | Castiglione G, Zappa M, Grazzini G, et al. Immunochemical vs. guaiac faecal occult blood tests in a population-based screening programme for colorectal cancer. Br J Cancer 1996; |
Excluded, HemeSelect test (qualitative version of Magstream) |
| 5: | Chen LS, Liao CS, Chang SH, et al. Cost-effectiveness analysis for determining optimal cut-off of immunochemical faecal occult blood test for population-based colorectal cancer screening (KCIS 16). J Med Screen 2007; |
Excluded, insufficient data |
| 6: | Crotta S, Castiglione G, Grazzini G, et al. Feasibility study of colorectal cancer screening by immunochemical faecal occult blood testing: results in a northern Italian community. Eur J Gastroenterol Hepatol 2004; |
Excluded, OC Hemodia 1 sample at 100 ng/ml threshold |
| 7: | Edwards JE, Williams JA, Thomas DW. Experiences in providing a screening service for colorectal cancer from a pathology laboratory. Pathology 2004; |
Excluded, non targeted I-FOBT |
| 8: | Fenocchi E, Martínez L, Tolve J, et al. Screening for colorectal cancer in Uruguay with an immunochemical faecal occult blood test. Eur J Cancer Prev. 2006; |
OC Hemodia 1 sample at 100 ng/ml threshold |
| 9: | Fraser CG, Mathew CM, McKay K, et al. Automated immunochemical quantitation of haemoglobin in faeces collected on cards for screening for colorectal cancer. Gut 2008; |
Excluded, immunochemical test performed on gaiac cards |
| 10: | Fu WP, Kam MH, Ling WM, et al. Screening for colorectal cancer using a quantitative immunochemical faecal occult blood test: a feasibility study in an Asian population. Tech Coloproctol 2009; |
Excluded, neither variation in threshold nor number of samples |
| 11: | Grazzini G, Visioli CB, Zorzi M, et al. Immunochemical faecal occult blood test: number of samples and positivity cutoff. What is the best strategy for colorectal cancer screening? Br J Cancer 2009; |
Included |
| 12: | Guittet L, Bouvier V, Mariotte N, et al. Performance of immunochemical faecal occult blood test in colorectal cancer screening in average-risk population according to positivity threshold and number of samples. Int J Cancer 2009; |
Included |
| 13: | Guittet L, Bouvier V, Mariotte N, et al. Comparison of a guaiac based and an immunochemical faecal occult blood test in screening for colorectal cancer in a general average risk population. Gut 2007; |
Excluded, preliminary analysis of study #12 |
| 14: | Haug U, Hundt S, Brenner H. Quantitative immunochemical fecal occult blood testing for colorectal adenoma detection: evaluation in the target population of screening and comparison with qualitative tests. Am J Gastroenterol. 2010; |
Excluded, non targeted I-FOBT |
| 15: | Hoepffner N, Shastri YM, Hanisch E, et al. Comparative evaluation of a new bedside faecal occult blood test in a prospective multicentre study. Aliment Pharmacol Ther 2006; |
Excluded, non targeted I-FOBT |
| 16: | Hol L, Wilschut JA, van Ballegooijen M, et al. Screening for colorectal cancer: random comparison of guaiac and immunochemical faecal occult blood testing at different cut-off levels. Br J Cancer 2009; |
Included |
| 17: | Launoy GD, Bertrand HJ, Berchi C, et al. Evaluation of an immunochemical fecal occult blood test with automated reading in screening for colorectal cancer in a general average-risk population. Int J Cancer 2005; |
Included |
| 18: | Levi Z, Rozen P, Hazazi R, et al. Sensitivity, but not specificity, of a quantitative immunochemical fecal occult blood test for neoplasia is slightly increased by the use of low-dose aspirin, NSAIDs, and anticoagulants. Am J Gastroenterol 2009; |
Excluded, not in average-risk general population |
| 19: | Levi Z, Rozen P, Hazazi R, et al. A quantitative immunochemical fecal occult blood test for colorectal neoplasia. Ann Intern Med 2007; |
Excluded, not in average-risk general population |
| 20: | Levi Z, Rozen P, Hazazi R, et al. Can quantification of faecal occult blood predetermine the need for colonoscopy in patients at risk for non-syndromic familial colorectal cancer? Aliment Pharmacol Ther 2006; |
Excluded, not in average-risk general population |
| 21: | Levi Z, Vilkin A, Niv Y. Esophago-gastro-duodenoscopy is not indicated in patients with positive immunochemical test and nonexplanatory colonoscopy. Eur J Gastroenterol Hepatol 2010; |
Excluded, only FOBT positive patients with negative colonoscopy |
| 22: | Levi Z, Birkenfeld S, vilkin A, et al. A higher detection rate for colorectal cancer and advanced adenomatous polyp for screening with immunochemical fecal occult blood test then guaiac fecal occult blood test, despite lower compliance. A prospective, controlled, feasibility study. Int J Cancer 2011; |
Excluded, predictive positive value and positivity rates not provided |
| 23: | Liedenbaum MH, van Rijn AF, de Vries AH, et al. Using CT colonography as a triage technique after a positive faecal occult blood test in colorectal cancer screening. Gut 2009; |
Excluded, only FOBT positive patients (guaiac of immunochemical) were included |
| 24: | Nakama H, Zhang B, Zhang X. Evaluation of the optimum cut-off point in immunochemical occult blood testing in screening for colorectal cancer. Eur J Cancer 2001; |
Excluded, four samples of OC Sensor, subject with familial history of cancer included, and insufficient data |
| 25: | Oono Y, Iriguchi Y, Doi Y, et al. A retrospective study of immunochemical fecal occult blood testing for colorectal cancer detection. Clin Chim Acta 2010; |
Excluded, only symptomatic cases of colorectal cancer |
| 26: | Park DI, Ryu S, Kim YH, et al. Comparison of guaiac-based and quantitative immunochemical fecal occult blood testing in a population at average risk undergoing colorectal cancer screening. Am J Gastroenterol 2010; |
Secondarily excluded because of high positivity rate of G-FOBT, and small sample (770 tests) |
| 27: | Parra-Blanco A, Gimeno-Garcia AZ, Quintero E, et al. Diagnostic accuracy of immunochemical versus guaiac faecal occult blood tests for colorectal cancer screening. J Gastroenterol 2010; |
Excluded, non targeted I-FOBT |
| 28: | Rozen P, Comaneshter D, Levi Z, et al. Cumulative evaluation of a quantitative immunochemical fecal occult blood test to determine its optimal clinical use. Cancer 2010; |
Excluded, not in average-risk general population |
| 29: | Rozen P, Levi Z, Hazazi R, et al. Identification of colorectal adenomas by a quantitative immunochemical faecal occult blood screening test depends on adenoma characteristics, development threshold used and number of tests performed. Aliment Pharmacol Ther 2009; |
Excluded, not in average-risk general population |
| 30: | Rozen P, Levi Z, Hazazi R, et al. Quantitative colonoscopic evaluation of relative efficiencies of an immunochemical faecal occult blood test and a sensitive guaiac test for detecting significant colorectal neoplasms. Aliment Pharmacol Ther 2009; |
Excluded, not in average-risk general population |
| 31: | Shastri YM, Loitsch S, Hoepffner N, et al. Comparison of an established simple office-based immunological FOBT with fecal tumor pyruvate kinase type M2 (M2-PK) for colorectal cancer screening: prospective multicenter study. Am J Gastroenterol 2008; |
Excluded, non targeted I-FOBT |
| 32: | Terhaar Sive Droste JS, Oort FA, van der Hulst RW, et al. Higher immunochemical test cut-off levels: lower positivity rates but still acceptable detection rates for early stage colorectal cancers. Cancer Epidemio Biomarkers Prev 2011; |
Excluded, not in average-risk general population |
| 33: | van Rossum LG, van Rijn AF, Laheij RJ, et al. Cutoff value determines the performance of a semi-quantitative immunochemical faecal occult blood test in a colorectal cancer screening programme. Br J Cancer 2009; |
Included |
| 34: | van Rossum LG, van Rijn AF, Laheij RJ, et al. Random comparison of guaiac and immunochemical fecal occult blood tests for colorectal cancer in a screening population. Gastroenterology 2008; |
Excluded, same data of study #33 |
| 35: | Vilkin A, Rozen P, Levi Z, et al. Performance characteristics and evaluation of an automated-developed and quantitative, immunochemical, fecal occult blood screening test. Am J Gastroenterol 2005; |
Excluded, not in average-risk general population |
| 36: | Whitlock EP, Lin J, Liles E, et al. Screening for colorectal cancer: an updated systematic review [internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2008 Oct | Excluded, review |
| 37: | Wong WM, Lam SK, Cheung KL, et al. Evaluation of an automated immunochemical fecal occult blood test for colorectal neoplasia detection in a Chinese population. Cancer 2003; |
Excluded, not in average-risk general population |
| 38: | Zorzi M, Fedato C, Grazzini G, et al. High sensitivity of five colorectal screening programmes with faecal immunochemical test in the Veneto Region, Italy. Gut 2010 Dec 30 | Excluded, only sensitivity of the test estimated, at a single threshold |
