Abstract
Background
Sending faecal occult blood tests (FOBT) by mail has been proposed both as a method to increase participation and a way to reduce staff costs in colorectal cancer screening.
Methods
Two multicentre randomized controlled trials (ISRCTN10351276) were performed: one randomly assigned 3196 individuals who had previously participated in colorectal screening to receive a FOBT kit at home or a standard invitation; in the second, 4219 people aged 50–69 years who did not respond to a screening invitation were either sent a FOBT or a standard recall letter. The cost per returned kit was calculated in each arm.
Results
Participation was higher with direct FOBT mailing in both trials: relative risk 1.11 (95% CI 1.06–1.17) and 1.36 (95% CI 1.16–1.60) for previous responders and non-responders, respectively. The cost per returned kit for previous responders ranged from 4.24€ to 16.10€, and from 3.29€ to 7.36€ with FOBT mailing and standard invitation, respectively, not including staff costs; for non-responders it ranged from 17.13€ to 46.80€, and from 7.36€ to 18.30€ with FOBT mailing and standard recall, respectively.
Conclusions
The FOBT mailing strategy modestly increased participation. This method can be used on a population of previous responders to reduce personnel costs and workload. When used as a reminder to non-responders, this method increases costs.
Introduction
The immunochemical FOBT does not need dietary restrictions; it is based on one simple stool sample and does not need any particular instructions for sampling; consequently its ease of use facilitates the direct mailing of the test kit to invite the screening population. Few published works have studied the effect of this strategy on participation and the results were contradictory.8–10 On the other hand, phenomena with a strong behavioural component such as participation in a screening programme are often context-specific. The only Italian study found no difference in participation rates between direct mailing of test kits and an invitation to pick up the test from the general practitioner (GP) or primary care clinic. 4
The direct mailing strategy has an effect also on the organizational costs of the screening programmes: on one hand, it increases the number, and consequently the cost, of test kits because many of those mailed are never returned; on the other hand, it reduces personnel costs by eliminating time-consuming face-to-face test pick-up.
The aim of this multicentric randomized trial is to compare the participation rates of direct FOBT mailing with those of a standard letter requesting the test be picked up at a primary care clinic, in two populations: a population of previous screening participants; and a non-respondent population, i.e. people who did not respond to the current screening round and should be recalled.
Organizational and economic consequences are also presented.
Materials and Methods
Setting
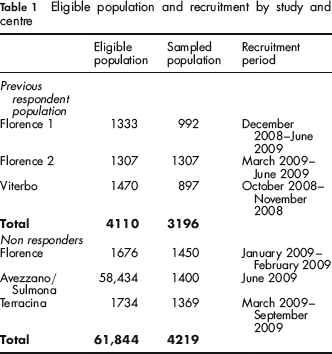
This multicentric study involved four organized screening programmes in three regions of Central Italy: Florence (Tuscany); Viterbo, Terracina (Lazio); and Avezzano/Sulmona (Abruzzo). The study was conducted from September 2008 to December 2009. The eligible population, sampled population and recruitment period by centre are reported in Table 1.
Eligible population and recruitment by study and centre
The study was financed with funds from a project of the Italian Ministry of Health aimed at supporting applied research for screening programmes (http://www.asrabruzzo.it/aree.php?id=46).
Population
The main characteristics of Italian screening programmes have already been described elsewhere.6,11
Briefly, colorectal cancer screening protocol addresses all residents aged 50–70 who are invited by mail every two years to perform a single immunochemical FOBT, without any dietary restrictions. Negative FOBT results are mailed back with a recommendation for biennial rescreening. Non-respondents to the first invitation receive a reminder, usually within six months.
Subjects who tested positive are referred for a complete colonoscopy. In the case of an incomplete colonoscopy, a double-contrast X-ray is then performed. Subjects with screen-detected neoplasms are referred for surgical or endoscopic treatment, and thereafter enrolled in a follow-up programme. A set of quality indicators is routinely monitored to control screening performance.6,11
In all the programmes involved, a latex agglutination FOBT (OC Sensor, Eiken, Tokyo, Japan) was adopted as a one-day screening test using a positivity threshold of 100 ng of Hb/mL of sample solution.
Design
Two independent randomized trials were designed and conducted (Figure 1). The first compared the response to two different screening invitation strategies, direct mailing of the FOBT kit or invitation to pick up the kit at the primary care office, in a population of previous participants i.e. persons who underwent colorectal cancer screening in the previous screening round (Figure 1) and were eligible for a new screening round according to organized screening guidelines as described above; Florence (two districts) and Viterbo recruited participants for this trial. The second compared participation after the two recall strategies of either the direct mailing of the kit or a standard recall letter to pick up the kit at a primary care clinic, in a population of non-responders to the first invitation (sent about three months before) and were eligible for a reminder letter according to the screening programme guidelines as described above; Florence, Terracina and Avezzano/Sulmona recruited participants for this trial (Figure 1).
Flow chart of the study design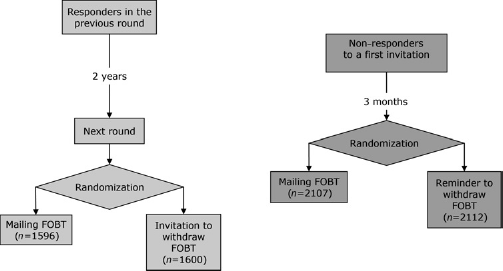
Sampling and randomization was performed using STATA 8.0 and using the first number drawn in the most recent National Lottery as the seed for the random number generator. In Florence, subjects from the same household could be identified, and were considered as a unique sampling unit (we decided not to account for this in the analysis, due to the extremely low number of these couples). Two samples of the same size were drawn and randomly assigned to one of the interventions for each centre and each trial from the lists of eligible subjects.
This trial was registered as International Standard Randomized Controlled Trial ISRCTN10351276.
Description of the intervention
In the control arm, people received by mail a letter that invited them to pick up the kit for the faecal occult blood test at a pre-fixed date at a primary care clinic. Invitation letters were signed by the GP or the screening programme medical director, according to standard local protocols.
Calendars were organized with an overbooking system; invited subjects could call a number to re-schedule the appointment if necessary. Health-care professionals delivered kits to invited subjects (mean number of kits delivered per hour = 20) and provided information on faecal sampling and returns.
People returned tests after faecal sampling directly in dedicated refrigerated containers at the primary care clinics.
In the intervention arm, as in the control arm, subjects received a mailing containing an invitation to participate in colorectal cancer screening signed by the general practitioner (GP) or the screening programme medical director.
The mailing also contained a FOBT kit (OC-Hemodia, Eiken, Tokyo, Japan), a pamphlet providing instructions on how to use the test, an envelope to return it in, a consent form, and an additional leaflet with general information about the colorectal screening programme. People in this group returned the test in the same way as the control arm subjects.
One centre (Florence District 1) mailed a reminder to both study arms to non-responders who had previously participated, 60 days after randomization.
Main outcomes
Participation in screening was measured as the proportion of people who returned a stool sample within 90 days of the mailing of the invitation letter or FOBT kit. Consequently, in the previous participant population, the outcome was measured before the standard recall, except in the District 1 centre in Florence. The analysis was intention to treat; in other words we used the total randomized population as the denominator in that arm and the number of returned samples as the numerator, regardless of how the kit was obtained.
Statistical analysis
We reported the relative risk of returning the test in the two arms with 95% confidence intervals (CI). The results were tested for heterogeneity; pooled results are reported only if the differences between centres were not significant.
In the group of non-respondents, the sample size was calculated to have a power of 95% to detect as significant a 4% difference in participation (alpha .05) with the hypothesis of a 10% participation rate in the control group. Each arm had a sample size of 1500. To reach this goal, we planned to randomize 700 people per arm per centre.
In the study of previous participants, a non-inferiority approach was used to determine the sample size, because the aim was to test a strategy able to reduce organizational and personnel costs. We chose a 5% decrease in participation as the maximum difference acceptable, we expected 75% participation in the control group and similar participation in the intervention group (75%); we estimated a sample size of 1100 subjects per arm to obtain 95% power with alpha .05. To ensure reaching these final sizes, we planned to randomize 450 people per arm per centre.
Cost analysis
We calculated the number of FOBT kits that needed to be mailed per returned test.
For the population of previous participants, we compared the cost per returned test in the intervention and in the control group for both subgroups (previous participants and non-respondents). Since the invitation strategy does not modify the accuracy of the test and consequently the health outcomes for those who decide to participate, we adopted a cost-minimization analysis per returned kit.
We calculated the cost of the direct mailing strategy as: (tube cost + kit mailing cost) * invitation/returned tests; the cost of the standard invitation strategy was: ((invitation mailing cost * invitation) + (cost of the tube * attendees))/returned tests.
We reported the extra costs of direct FOBT mailing as the threshold-cost of the organizational and administrative allowance having the tests picked up at the clinic per returned kit.
A similar analysis was performed for the non-responding population. Since this strategy does not eliminate organizational and personnel costs for kit pick-up at the clinic, which are required for the initial invitation, the cost per returned FOBT in the direct mailing arm had to be equal to or lesser than the costs in the standard recall arm to be acceptable.
The cost per person gained to screening was also calculated, where the increase in participation for the intervention arm was relevant.
Results
Previous participant population
Table 2 shows results in this population subgroup. Since for the trial on previous participant population superiority of the experimental strategy is evident in the pooled analysis and there is no heterogeneity among centres, we present the results in terms of relative risk of returning a FOBT instead of a non-inferiority analysis.
Number of recruited subjects, number of respondent subjects, and participation rates in control and intervention groups in a previous respondents population according to centres and in the overall series
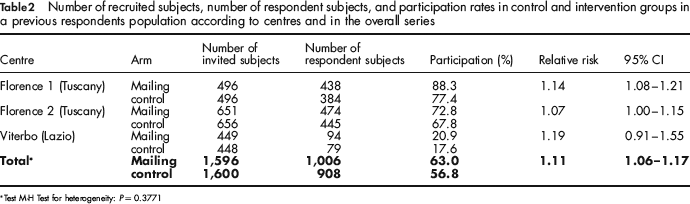
Test M-H Test for heterogeneity: P = 0.3771
In the Florence screening programme, a population of previous participants was recruited among residents of two urban districts: in the first (Florence 1), 496 subjects were randomized in the control group and 496 in the intervention arm. In the second district (Florence 2), 651 and 656 subjects were randomized in the control and intervention arms, respectively.
Participation in the first district was 88% (438/496) in the direct FOBT mailing group and 77% (384/496) in the control group. Relative risk was 1.14 (95% CI 1.08–1.21). In the second district in Florence, participation of invited subjects was 73% (474/651) in the intervention group and 68% (445/656) in the control group, with a relative risk of 1.07 (95% CI 1.00–1.15).
In the Viterbo screening programme, invited subject participation was 21% (94/449) in the intervention arm and 18% (79/448) among subjects who received the standard invitation letter (relative risk = 1.19, 95% CI 0.91–1.55).
Overall, as a consequence of the direct FOBT mailing we observed an 11% increase in the participation rate in the previous respondents population (RR: 1.11; 95% CI 1.06–1.17).
No statistically significant heterogeneity between the centres was observed (P = 0.38). Neither was the heterogeneity between genders or age groups (50–59 vs. 60–69) significant (P = 0.12 and P = 0.23, respectively), although a slightly higher RR in women (1.16; 95% CI 1.07–1.25) and in older people was detected (1.14; 95% CI 1.06–1.22).
The positivity rate was 5.1% (51/1006) in the mailing arm and 3.3% (30/908) in the conventional arm, the difference was border-line significant (P = 0.055).
No inadequate tests were reported for any of the previous participants.
Non-responders
Table 3 shows the results in this subgroup. In particular, in the Florence programme, 722 non-responders were randomized in the intervention group and received a FOBT kit as a reminder. Of these, 142 (20%) returned their faecal sampling within 90 days; while 728 non-responders had been randomized to the control group and were mailed the standard recall letter to go to the clinic for kit pick-up, 127 of them (17%) returned a faecal sample.
Number of recruited subjects, number of respondent subjects, participation rates in control and intervention groups in a non-respondent population according to centres and in the overall series
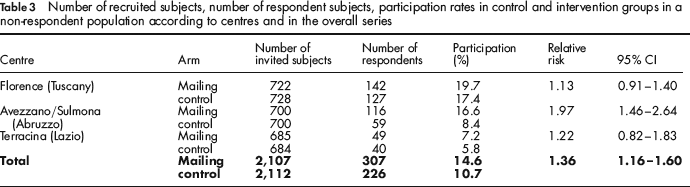
In conclusion, in the Florence programme, the direct mailing of FOBT kits produced a 13% increase in the number of returned tests within 90 days. The increase was not statistically significant (95% CI 0.91–1.40).
In the Avezzano/Sulmona screening programme, 700 subjects who did not respond to the first invitation were mailed a FOBT kit; 116 of them (17%) returned a faecal test.
In the control group, 700 subjects who did not respond to the first invitation were mailed a standard reminder and 59 (8%) returned a faecal sample. Relative risk was 1.97 (95% CI 1.46–2.64).
In the Terracina screening programme, the direct mailing of FOBT kits to people who did not respond to the first invitation produced a response rate of 7% (49/685) within 90 days. In the control group, 6% of non-respondents (40/684) who received a standard reminder returned a faecal sample. Relative risk was 1.22 (95% CI 0.82–1.83).
The heterogeneity between centres was significant (P = 0.01); the difference was completely due to the stronger effect observed in Avezzano/Sulmona. The overall heterogeneity between gender and age groups (50–59 vs. 60–69) was not significant (P = 0.16 and P = 0.3, respectively), but showed a slightly stronger effect in men (RR 1.56 95% CI 1.21–2.00) and older people (RR 1.47 95% CI 1.20–1.81).
No inadequate tests were registered in this study group either.
The positivity rate was 7.5% (23/307) in the mailing arm and 6.2% (14/226) in the conventional arm, and the difference was not statistically significant (P = 0.56).
Pooling the positivity rates observed in the two trials, there were significantly more positive results in the mailing arms than in the control arms: 5.6% (74/1313) and 3.7% (44/1134) respectively, relative risk 1.5 (95% CI 1.05–2.17).
Cost analysis
Previous participants population
In Florence, 1.3 invitations were needed to result in one completed test whereas in the direct mailing arm 1.4 invitations were sent for each FOBT returned in the standard screening protocol.
In Viterbo, the number of invitations needed per returned test was higher than in the other study groups (4.8 for intervention arm and 5.7 for control arm).
Since there cannot be significant differences between the two invitation strategies in terms of health outcome because the test is the same and the increase in participation rate was small, we adopted a cost-minimization analysis. We considered mailing costs of 1.97€ per tube, 1.40€ per kit and 0.95€ per standard invitation letter. Results of the cost analysis are shown in Table 4. For the direct mailing strategy, the cost per returned test was 4.24€ in Florence and 16.10€ in Viterbo.
Costs of the direct mailing and standard strategies according to the two population groups in the control and intervention arm (costs expressed in euro)
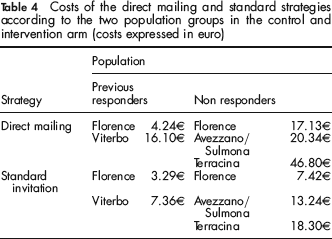
The total cost of the standard invitation strategy was 3.29€ in Florence and 7.36€ in Viterbo. Consequently, the direct mailing strategy may be convenient only if the total cost per FOBT kit of test pick-up/return at clinic is equal to or higher than 0.95€ in Florence and 8.74€ in Viterbo.
Non-responders
In Florence, 5.7 invitations had to be sent to the control group and 5.1 to the direct mailing group to receive a completed FOBT.
In Terracina, the number of invitations needed for a returned FOBT was 17.2 in the control group and 13.9 in the intervention group.
In the Avezzano screening programme, 11.9 standard invitations and six direct mailings were needed to receive one returned sample. Moreover, 12.3 direct mailings were needed for each additional returned test in the intervention arm.
Considering the following delivery costs (mailing tube, 1.97€, FOBT kit, 1.40€ and traditional invitation letter, 0.95€), we estimated the costs of a single returned test using the mailing strategy (see Table 4): 17.13€ in Florence, 46.80€ in Terracina, and 20.34€ in Avezzano/Sulmona. Since this strategy does not eliminate the front office activity necessary to send the FOBT to people who responded to the first invitation, mailing costs in the experimental arm must be equal to or less than the mailing costs in the standard invitation strategy. The costs of the standard recall strategy were lower in all centres: 7.42€ in Florence, 18.30€ in Terracina and 13.24€ in Avezzano/Sulmona.
Only in Avezzano/Sulmona, where the increase in participation rate in the mailing arm was relevant, we estimated costs per person gained to screening: ((1.97*0.834) + (1.40–0.95))/0.082 = 25.50€.
Discussion
We conducted a randomized trial to measure the effect of direct mailing of FOBT kits on participation in colorectal cancer screening.
The direct FOBT mailing strategy is actually an attempt to reduce the physical barriers to screening by providing the participants with a screening test with no effort on their part. At the same time it may reduce personnel costs and organizational workload.
Impact on participation
The results of this trial demonstrated a small increase in participation in a population of previous responders, consistent in the three centres, even if baseline compliance was extremely variable. In particular the low participation rate in Viterbo was a matter of concern. Two known factors may have contributed: 1) in the previous round the FOBT was delivered personally by the GP, but this strategy, which obtained a very high participation 12 , was dismissed for economic and organizational reasons and substituted with test pick-up directly by patients from health professionals at primary care offices; 2) in this centre, due to the organizational problems already mentioned, the interval between invitations was three years instead of the recommended two (Figure 1), so some people may have performed the test spontaneously.
The aim of this part of the study was to test if a screening programme that avoided direct picking-up staff costs would be at least as effective as the standard strategy. The results observed confirm that direct mailing can be used without any negative impact on participation.
On the other hand, the study on the non-respondents tested whether mailing the FOBT would increase participation compared with the standard recall letter.
In all centres, mailing the FOBT led to an increase in participation but the effect in each of the three centres varied; it was lower in Florence and Terracina and stronger in Avezzano/Sulmona. It is not surprising that this kind of strategy is context-dependent, but it is difficult to understand the differences that may exist between two rural areas less than 200 km apart, such as Terracina and Avezzano/Sulmona.
Differences between age groups and genders were slight in this study, as they were in the previous participant population. While a slight, not significant increase in the number of older participants was consistently observed in the two study groups, surprisingly, the gender effect modification was contrasting in the two study groups: a slightly stronger effect in women in the previous participant group and a slightly stronger effect in men in the non-responder group. All of these differences may be explained by random fluctuations.
A comparison with other studies on direct mailing is very difficult, given the many differences in their design and setting. Mahon 9 showed that mailing FOBT kits prior to office appointments was related to a strong increase in FOBT return rates, whereas Ore et al. 10 , in a population-based study, observed a mild effect in enhancing participation with this strategy. Church et al. 8 carried out a randomized trial consisting of direct mailing of FOBT kits among residents of an American community in which colorectal cancer screening was promoted. The authors showed a positive impact of the direct mailing strategy; Goldberg obtained similar results 13 but the findings (like those of the other studies) are not comparable with ours for the reasons mentioned above.
In our study no inadequate samples were registered. This result is not surprising if we consider that the proportion of inadequate tests was very low (<1%) in Italian surveys. 6 Inadequate tests are essentially due to incorrect sampling by the subject and their proportion represents an indirect estimate of the quality of the informational material. Actually, the sampling procedure is very simple and very little instruction is necessary to ensure good quality samplings. In our study, people recruited in all study arms were provided with a leaflet prepared according to recommendations of the Italian Working Group on Communication. 14
We found a higher positivity rate in the mailing arms than in the control arms. This is surprising since the difference in participation is small and the prevalence of bleeding certainly could not have influenced the mode of delivery, even if we cannot exclude a stronger self-selection of symptomatic patients in the intervention arms. The way of returning the samples was identical for the two arms, but it could be that the time it took to return the sample was systematically shorter in the mailing arm, which better preserved the hemoglobin. 15 Given the study design, we can exclude seasonal variations in positivity rates. 16 On the other hand, we can argue that the accompanying letter, with very short written instructions and sent only to participants in the mailing arms, may have positively influenced their behavior in returning samples.
Organizational and cost analysis
As far as cost analysis is concerned, it is clear that a direct mailing strategy may be cost-effective in high participation settings because the expense of mailing tubes that were never returned negatively influenced the cost/benefit balance. In the population of previous participants, the extra cost per returned kit observed in the direct mailing arm, at least in Florence, was lower than the personnel costs for face-to-face picking up and returning the tests.
It is therefore evident that this strategy is more costly for non-respondents than for previous participants. Furthermore, a strategy using direct mailing only for reminders does not eliminate front office activity. Consequently, we considered the best strategy the one with lower invitation costs per returned test. In all three centres the cost of direct mailing was much higher than the standard recall. On the other hand, in Avezzano/Sulmona, the mailing strategy in the group of non-responders resulted in higher participation than could be obtained with a standard reminder. In this case, a cost-minimization approach was not appropriate, thus we estimated the cost per subject gained to screening, which was more than 25€. According to a previous Italian study 17 , this estimate is much higher than the cost of the entire screening process per participant subject. This cost also overcomes the predicted cost-effectiveness threshold of an endeavour to increase participation. 18
Limitations
One of the limitations of this study is a consequence of the outcome definition: success was defined as the FOBT being returned within 90 days. This time cut-off was necessary to standardize the trial procedures, but differences between the two arms may have changed if we had observed a longer period. In particular, our study was not designed to see the effect of a recall after the first invitation to a population of previous participants, since the recall letter is usually mailed more than three months after the first invitation. A recall letter to people who received the test at home may be highly effective as a reminder and will probably increase cost-effectiveness. In fact, among previous participants, the highest participation was registered in Florence District 1 (about 90%) and these results are probably due to the reminder strategy adopted for this population. Also in the non-responders group, a third reminder to people who had already received a kit at home may be cost-effective, but given the trial design it was not fair to compare a three-contact strategy with a two-contact strategy (control); in addition, three letters proposing the same offer to the control group may be considered persecutory.
A second limitation of this study is that we did not compare a third strategy now emerging in FOBT-based colorectal cancer screening in Italy: the use of pharmacies as test providers.19,20 Unfortunately, when the trial was being planned, only a few programmes had been starting using this strategy and it was not feasible to run a trial in that setting.
Conclusions
In our context, the strategy of mailing FOBT kits had a modest effect on increasing screening participation. This method can be used with previous responders without any loss in participation, where it may be useful to minimize costs and the workload associated with kit pick-up/return at the clinic, while at the same time reducing the waste of consumables by mailing kits only to those who are likely to participate.
Moreover, the direct mailing of FOBT kits produced a slight effect on participation when used as a reminder to non-responders following the standard invitation letter, but with higher costs per invited person.
