Abstract
Objective:
To provide current population-based prevalence and prenatal diagnosis rates (PND) for specified major congenital anomalies in England and Wales to enable monitoring of the Fetal Anomaly Screening Programme (FASP).
Design:
Secondary analysis of prospectively collected registry data.
Setting:
Seven multiple-source, population-based congenital anomaly registers, members of the British Isles Network of Congenital Anomaly Registers (BINOCAR) in 2005 and 2006.
Population:
2,883 births with congenital anomalies from a total of 601,545 live and stillbirths.
Main outcome measures:
PND and birth prevalence of selected congenital anomaly groups/subtypes (anencephaly, spina-bifida, serious cardiac, diaphragmatic hernia, gastroschisis, exomphalos, bilateral renal agenesis, lethal/severe skeletal dysplasia, cleft lip with or without cleft palate [CL + /-P]).
Results
Of the selected anomaly groups, the most frequently reported were serious cardiac (14.1 per 10,000 births [95% CI 13.0-15.2]) and CL + /-P (9.7 per 10,000 births [8.9-10.5]); the least frequent were bilateral renal agenesis and lethal/severe skeletal dysplasia (<1.5 per 10,000 births). The PND varied for different anomalies from 53.1% (95% CI 43.5-65.2) for serious cardiac anomalies to 99.6% (95% CI 97.9-100.0) for anencephaly. Least variation in PND rates was for anencephaly (range 98.9-100%) and gastroschisis (93.5-100%); greatest variation was for serious cardiac (43.5-65.2%) and lethal/severe skeletal dysplasias (50.0-100%).
Conclusions:
BINOCAR registers can, uniquely, provide contemporary data on PND and birth prevalence rates to enable monitoring of the ultrasound component of FASP at a national and regional level, allowing comparisons between populationstobemade, planningofresourcesfacilitatedand assistance for parents making informed decisions on whether to enter the screening programme.
Introduction
The use of antenatal ultrasound to visualize the fetus has developed since its introduction in the 1970s to become a routine part of pregnancy care. There have been several reports with recommendations about when and what to scan. In 1984 the Royal College of Obstetricians and Gynaecologists (RCOG) Working Party suggested a single routine scan at 16 to 18 weeks gestation 2 ; in 1997 the 18-20 week ‘anomaly’ scan was recommended 3 and in 2000 the RCOG generated a standardized protocol for the content of the scan with suggested detection rates for different anomalies available in an information leaflet for pregnant women. 4 In 2003, the first National Institute for Health and Clinical Excellence (NICE) antenatal guidelines recommended that women be offered an ultrasound scan to screen for structural anomalies. 5 The current (2008) NICE guidelines 6 state ‘ultrasound screening for fetal anomalies should be routinely offered, normally between 18 weeks 0 days and 20 weeks 6 days’. A survey of antenatal ultrasound practice in England in 2008 reported that all maternity units offered the second trimester anomaly scan to all women. 7
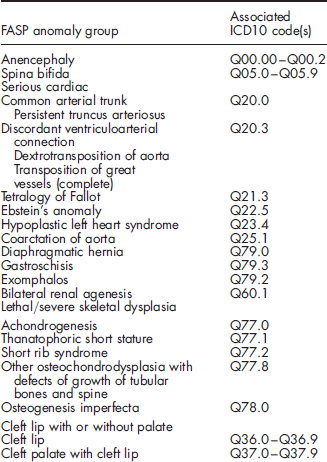
There is now an agreed policy for screening for fetal anomalies using ultrasound outlined by the Department of Health through the UK National Screening Committee (UK NSC), supported by the National Services Framework (NSF) and NICE. The NHS Fetal Anomaly Screening Programme (FASP; http://fetalanomaly.screening.nhs.uk/) has the remit to interpret this policy and provide a comprehensive service (which includes screening for Down's syndrome and structural congenital anomalies), for England. NHS FASP has initially chosen nine structural congenital anomalies or groups of anomalies and two chromosome anomalies (Trisomies 13 and18) that women should expect to be screened for at the anomaly scan as a baseline for audit. 8 The structural anomalies chosen are anencephaly, spina bifida, serious cardiac anomalies, diaphragmatic hernia, gastroschisis, exomphalos, bilateral renal agenesis, lethal or severe skeletal dysplasias, and cleft lip with or without cleft palate.
The sensitivity and specificity of ultrasound diagnosis of fetal anomaly will depend on a number of factors relating to, for example, the type of anomaly, scan duration, skill of the operator, type of machine, and maternal factors such as body mass index. 9 Parents need to know expected detection rates so that they can make an informed decision on whether to have an anomaly scan and clinicians need standards against which to compare their own performance. To assess performance of the fetal anomaly scan it is important that both prenatal detection and prevalence rates of specific anomalies be monitored at regional and national levels in the same way that laboratory performance of serum and combined screening is done (http://fetalanomaly.screening.nhs.uk/dqasspublications).
Compared with most health outcomes, specific congenital anomalies occur at low population prevalence. For rare anomalies, pooled data from large national cohorts are necessary to generate accurate estimates of prevalence. Cohorts generated from fetal medicine centres are unlikely to be population-based and therefore biased by referral patterns and variations in detection rates.
The British Isles Network of Congenital Anomaly Registers (BINOCAR; http://www.binocar.org/) is a network of regional and disease specific population-based registers across the British Isles, set up in 1996, partly in response to the recognition of the under ascertainment of anomalies to the National Congenital Anomaly System (NCAS). 10 The aim of BINOCAR is to provide continuous epidemiological monitoring of the frequency, nature, and outcome of congenital anomalies as well as research on possible causes. All births in Wales and 43% of births in England are covered by regional congenital anomaly registers. All the registers follow a common methodology for collection and coding of cases.
The aim of this paper is to provide contemporary population-based prenatal diagnosis and prevalence rates for specified major structural congenital anomalies amenable to detection by ultrasound at the 18 + 0 to 20 + 6 weeks gestation fetal anomaly screening scan to enable monitoring of the NHS FASP.
Methods
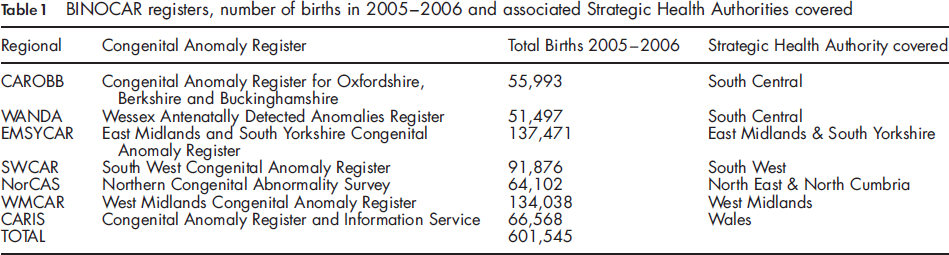
Seven BINOCAR regional population-based congenital anomaly registers in England and Wales contributed data to the study, a secondary analysis of prospectively collected registry data (Table 1).
BINOCAR registers, number of births in 2005-2006 and associated Strategic Health Authorities covered
Anonymized data on nine major structural congenital anomaly groups (Box 1) selected in discussion with the FASP were collated to calculate prenatal diagnosis and prevalence rates. Anomalies were coded according to the WHO International Classification of Diseases version 10 (ICD 10). Cases with one of the selected anomalies were included, whether as an isolated anomaly or with additional structural or chromosomal anomalies present.
The nine FASP anomalies agreed for study with associated ICD 10 code
Cases included all affected fetuses and infants notified within one year with a date of delivery/termination between 01 January 2005 and 31 December 2006. Cases were defined geographically by mother's place of residence at delivery.
To achieve high levels of ascertainment and completeness, BINOCAR registers identify cases using multiple source notifications prospectively throughout pregnancy and confirmation or otherwise at delivery. These sources include laboratories, postmortem reports, parents, ultrasound departments, delivery suites, obstetric notes, neonatal units, child health systems, inpatient administration systems, and paediatric notes. All prenatally suspected and postnatally confirmed anomalies are recorded with the exception of some minor anomalies. 11 Prenatally suspected anomalies are followed up and the final diagnosis is recorded. The outcomes of affected pregnancies are also recorded including live births, stillbirths, fetuses terminated for fetal anomaly and losses less than 24 weeks. Cases can have single, multiple anomalies or a syndrome and each is coded using ICD 10.
The overall estimate for the prevalence of the FASP anomalies in the area covered by the BINOCAR registers was obtained by pooling individual data from all registers and calculating the overall prevalence. This is equivalent to weighting the estimates from each register by population size. This approach gives a direct estimate of the prevalence in the area covered by the registers. Total prevalence rates were calculated for each anomaly group including and excluding those with a chromosome anomaly. The presentation of these two rates is an attempt to reflect the clinical perspective at the time of the scan and the retrospective classification following further cytogenetic tests. Routinely collected denominator data on total births (live and stillbirths) were supplied by the Office for National Statistics.12,13
Prenatal diagnosis was defined as the diagnosis of any anomaly before delivery. In cases of isolated anomaly, the proportion of cases diagnosed prenatally will reflect the detection rate for that anomaly. However, in cases with two or more anomalies the detection rate is not specific to any individual anomaly.
Birth prevalence rates per 10,000 total births and the percentage of anomalies detected antenatally are presented with 95% confidence intervals (CIs). The range of variation for each of these measures across the regional congenital anomaly registers is provided.
Results
The birth population for this study was 601,545 live and stillbirths, this represents 47.9% of the total births in England and Wales (n = 1,255,492) during the same period. The coverage of the individual registers varied from 25,500 to 68,000 births per year (Table 1).
Total prevalence with and without chromosome anomalies for each FASP anomaly group: all BINOCAR registers combined, 2005-2006
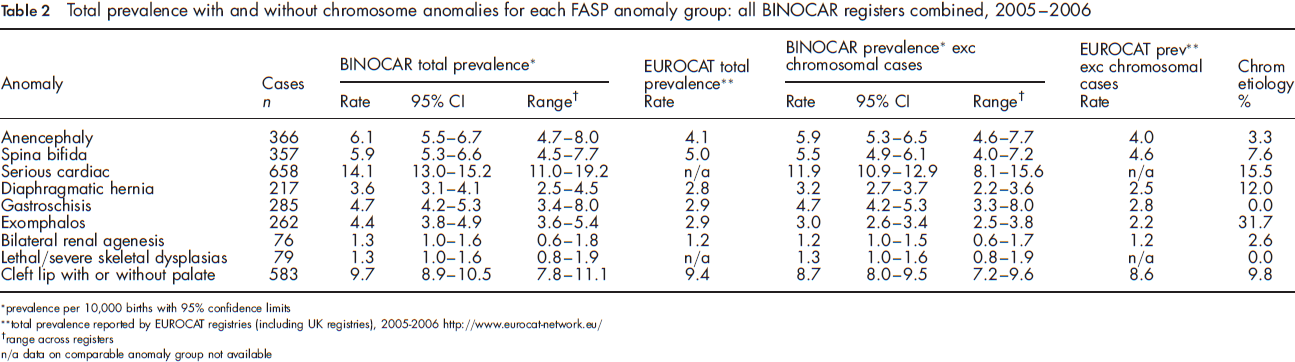
prevalence per 1 0,000 births with 95% confidence limits
total prevalence reported by EUROCAT registries (including UK registries), 2005-2006 http://www.eurocat-network.eu/
range across registers
n/a data on comparable anomaly group not available
The most frequent anomalies notified in this study were serious cardiac (14.1 per 10,000 births, 1 in 710 births [95% CI 13.0-15.2]) and cleft lip with or without palate (9.7 per 10,000 births, 1 in 1,032 [95% CI 8.9-10.5]).
The least common of the selected anomaly groups notified were bilateral renal agenesis and lethal/severe skeletal dysplasia, occurring at birth prevalence rates of less than 1.5 per 10,000 births.
The anomaly group that showed most variation in total prevalence across the registers was the bilateral renal agenesis group which varied from 0.6-1.8 per 10,000 total births, and the least variation was for cleft lip with or without palate (7.8-11.1 per 10,000 births respectively). The anomalies most commonly associated with a chromosome anomaly were exomphalos (31.7%) and serious cardiac (15.5%). There were no reported cases of gastroschisis or lethal/severe skeletal dysplasia associated with chromosomal anomalies.
Percentage of total prenatally diagnosed at any gestation and <24 weeks (where gestation of diagnosis is known) for each FASP anomaly group: data from 6 of the 7 BINOCAR registers combined, 2005-2006
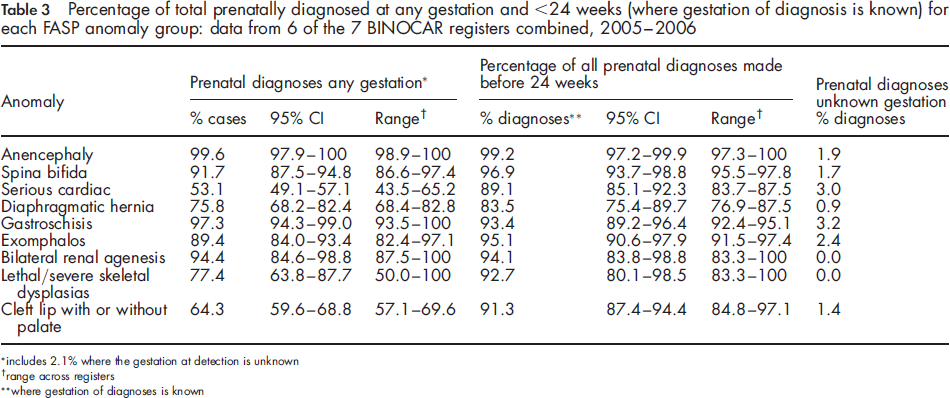
includes 2.1% where the gestation at detection is unknown
range across registers
where gestation of diagnoses is known
Discussion
This is the first study to provide contemporary population-based prenatal diagnosis and prevalence rates for selected major structural congenital anomalies, amenable to prenatal diagnosis, for a large proportion of England and all of Wales.
In this benchmark study, overall prenatal diagnosis and total prevalence rates are provided, i.e. the individual anomalies may have been isolated, associated with other structural anomalies, or with chromosome anomalies. This pooled prevalence will support planning for services (ultrasound, fetal medicine, and paediatric) and the identification of trends and patterns of disease. The information on prenatal diagnosis will facilitate the development of prenatal diagnostic standards for performance management. These data will enable women to exercise informed consent when making a decision on whether to have an anomaly scan.
The birth prevalence for the nine selected anomaly groups range from 1.3 to 14.1 per 10,000 births within the regional study populations. These rates are in line with those from other European population-based registries reported by EUROCAT and collected using similar standardized methodologies. 14 In contrast, data presented by the International Clearinghouse for Birth Defects show a wider range of birth prevalence, reflecting a less standardized approach to congenital anomaly data collection and the wide ranging variation in the provision of antenatal services and cultural and policy differences. 15
This analysis has five major strengths. Firstly, the data presented are from high quality population-based registers. Fetal medicine services are generally structured around regional centres which regularly report their individual service antenatal detection rates. Such data, from single or multicentre sites, present prenatal diagnosis for a selected (pre-screened), often high risk population and may generate artificially high detection rates. Larger population-based series minimize potential selection bias. Secondly, BINOCAR registers use multiple-source notifications, i.e. notifications relating to the same case are received from multiple sources, which maximize the details available and improve ascertainment for rare outcomes. The NCAS (recently under review and no longer issuing reports) 16 has used a predominantly single source for their notifications, leading to low levels of ascertainment and high levels of under-reporting. 10 The third strength is that all the BINOCAR registries use a standardized methodology and minimum data set ensuring high quality, comparable data. Fourth, birth prevalence and detection rates in smaller populations are likely to show more variation and to have wider estimates. This study overcomes this problem by combining data from regional registers and providing results for a large proportion of England and Wales. Finally, all outcomes of affected pregnancies are recorded by the BINOCAR registries, including terminations of pregnancy (TOP) and late fetal losses. For significant structural anomalies, where cases are detected prenatally and outcomes are poor, a system that fails to record TOPs and late fetal losses will underestimate the associated birth prevalence and present inaccurate prenatal detection rates.
There are some acknowledged limitations in this initial attempt to provide prevalence and prenatal detection rates for England and Wales. Although the whole of Wales is covered by a congenital anomaly register the whole of England is not, and so the data are not truly ‘national’ but the best approximation possible to date. Although the data are contemporary, the study is based on two years of data alone and does not provide any information about trends over time. Levels of ascertainment of cases diagnosed in the post-neonatal period vary between registries depending upon their established notification networks; low levels will cause an underestimation of the birth prevalence and overestimation of the proportion of cases diagnosed prenatally. However, this problem has been minimized in this study by the selection of the anomaly groups. These have been restricted to nine, selected because they are associated with significant mortality or morbidity, or require immediate postnatal support, and have high prenatal detection rates (> 50%). 8 Some of the anomaly groups chosen have a low prevalence, for example within our data bilateral renal agenesis occurred in 1 in 7,692 births. NHS Regions seem to be the most appropriate population size to monitor the performance of screening programmes for fetal anomalies as screening services are already structured around this geography and prenatal detection and prevalence rates in smaller populations are likely to show large annual variation.
In this study cases were included whether they occurred in isolation or were associated with other anomalies, with and without a chromosome anomaly; the suspicion on scan leading to the prenatal diagnosis of a case may not have been due to the identification of the anomaly listed but to an associated anomaly. In future analyses further subdivision into isolated and non-isolated anomaly, with and without chromosome anomalies may be helpful.
All the contributing registries ascertain anomalies identified prenatally, at birth and in early childhood. For those anomalies obvious at delivery (anencephaly, spina bifida, gastroschisis, exomphalos, severe skeletal dysplasias and cleft lip and palate), ascertainment is likely to be high, but for those ‘hidden’ defects, such as more minor cardiac, the ascertainment at birth may be lower leading to falsely high detection rates for these anomalies. The availability and uptake of fetal and neonatal postmortem examinations may also affect ascertainment, for those ‘hidden’ anomalies. Real geographical variations have been noted in a number of anomaly groups, e.g. a north-south gradient for gastroschisis cases and a west-east 17 gradient for neural tube defects. 18
A small proportion of women decline the fetal anomaly scan and some present too late in pregnancy for the scan to be performed. This ultimately leads to an underestimation of the prenatal detection rates for the screened population because the total birth denominator is larger than the actual screened population.
The prenatal detection of anomalies is not limited to the anomaly scan alone; anomalies may be detected as part of other screening programmes or diagnostic tests. Some structural anomalies such as anencephaly are amenable to detection by ultrasound at earlier gestations (at the time of the dating scan/combined screening). For all anomaly groups the majority of prenatal diagnoses were made before 24 weeks gestation.
Cases with underlying chromosomal anomalies may be diagnosed prenatally by invasive testing and cytogenetic analysis following high risk screening results before the detection of specific anomalies is possible, for example Trisomy 18 diagnosed by Chorion Villous Sampling before ultrasound diagnosis of exomphalos.
Prenatal diagnosis of structural congenital anomalies is now a major feature of antenatal care. It allows parents to make choices concerning TOP, careful planning for care following delivery for some anomalies, and identification of the small proportion of anomalies that may be amenable to antenatal treatment. With the development of the national FASP, the importance of a uniform feasible method of monitoring outcomes is essential. This paper describes an initial attempt to provide data on prevalence and prenatal detection rates from BINOCAR registries which allows the development of prenatal diagnostic standards for performance management and enables women to exercise informed consent when making a decision on whether to have an anomaly scan.
Regional congenital anomaly registers were set up, in part, to audit the newly emerging specialty of prenatal diagnosis and because the NCAS, set up in 1963 following the thalidomide epidemic 19 to monitor congenital anomalies, does not collect information on prenatal detection, affected late fetal losses or on cases resulting in TOP for fetal anomaly. The NCAS relied on a non-mandatory notification system that in recent years has been shown to under-report in some areas by more than 50%. 20 Without the availability of regional congenital anomaly registers, reliable estimates of prevalence and diagnostic rates would not be possible. The 2004 CMO's report 21 and the 2008 NICE antenatal care guidelines 6 highlight the importance of monitoring screening programmes. The latter states that ‘participation in regional congenital anomaly registers and/or UK National Screening Committee approved audit systems is strongly recommended to facilitate the audit of detection rates’. The way forward is to establish total coverage of England by regional registers that also have the remit to supply FASP with information required to audit prenatal screening programmes. The 2004 CMO's report 21 states that the ‘geographical coverage and reporting rigour of regional congenital anomaly registers should be increased and central core funding should be made available to secure the long-term future of these precious national resources’. If this recommendation is followed, it will be feasible to monitor the ever-changing discipline of prenatal diagnosis at a national level, to set national standards, to provide units with detection rates against which to compare their own performance and to enable women to make a more informed choice about whether or not to enter the FASP.
Conclusion
Ultrasound screening for fetal anomaly is recommended by a number of organizations including the NICE and the RCOG. Antenatal detection provides parents with information regarding severity and treatment allowing them to make informed choices concerning their affected pregnancy. It is vital that birth prevalence and prenatal detection rates are monitored nationally via the ultrasound component of FASP so that comparisons between populations can be made, planning of resources facilitated and parents given information to assist them in their decision on whether to enter the screening programme.
Contribution to Authorship
The BINOCAR Management Committee (ED, JM, PB and AT) initiated the study, CR and AT collated the data and carried out the primary analysis. PB wrote the first draft of the paper. All authors were involved in analysis and interpretation of the data, writing and reviewing the paper.
Ethical approval
BINOCAR registers have MREC approval (Trent MREC, date 11/09/2009 09/H0405/48) for studies involving the use of their data.
Funding
This paper reports on an independent study which is funded/part-funded by the Policy Research Programme in the Department of Health and the Healthcare Quality Improvement Partnership (HQIP). The views expressed are not necessarily those of the Department. Part funding is also from local primary care trusts and the Welsh Assembly Government.
Footnotes
Acknowledgements
We thank the staff of the contributing registers (Congenital Anomaly Register for Oxfordshire, Berkshire and Buckinghamshire [CAROBB], Wessex Antenatally Detected Anomalies Register [WANDA], East Midlands and South Yorkshire Congenital Anomaly Register [EMSYCAR], South West Congenital Anomaly Register [SWCAR], Northern Congenital Abnormality Survey [NorCAS], West Midlands Congenital Anomaly Register [WMCAR], Congenital Anomaly Register and Information Service [CARIS]) for providing and checking the data. We are grateful to all data collectors across all the geographical areas covered by the registers. The Fetal Anomaly Screening Programme team discussed and chose the selected anomalies analysed.
