Abstract
Objective
There is a continuing need to monitor and evaluate the impact of organized screening programmes on cancer incidence and mortality. We report results from a programme assessment conducted within the International Cancer Screening Network (ICSN) to understand the characteristics of cervical screening programmes within countries that have established population-based breast cancer screening programmes.
Methods
In 2007-2008, we asked 26 ICSN country representatives to complete a web-based survey that included questions on breast and cervical cancer screening programmes. We summarized information from 16 countries with both types of organized programmes.
Results
In 63% of these countries, the organization of the cervical cancer screening programme was similar to that of the breast cancer screening programme in the same country. There were differences in programme characteristics, including year established (1962-2003 cervical; 1986-2002 breast) and ages covered (15-70+ cervical; 40-75+ breast). Adoption of new screening technologies was evident (44% liquid-based Pap tests; 13% human papillomavirus (HPV)-triage tests cervical; 56% digital mammography breast). There was wide variation in participation rates for both programme types (<4-80% cervical; 12-88% breast), and participation rates tended to be higher for cervical (70-80%) than for breast (60-70%) cancer screening programmes. Eleven ICSN member countries had approved the HPV vaccine and five more were considering its use in their organized programmes.
Conclusion
Overall, there were similarities and differences in the organization of breast and cervical cancer screening programmes among ICSN countries. This assessment can assist established and new screening programmes in understanding the organization and structure of cancer screening programmes.
Introduction
There is a continuing need to monitor and evaluate the impact of organized screening programmes on cancer incidence and mortality. Programme effectiveness depends on coverage of at-risk populations, application of high-quality screening tests and accurate test interpretation.7–10 The ability to compare screening programme policies and organization can aid existing programmes as well as countries that are planning new programmes.10,11 Furthermore, approaches for optimally incorporating new technologies into programmes can facilitate appropriate uptake of evidence-based new technologies into clinical practice. The International Cancer Screening Network (ICSN), a voluntary consortium of countries that have active population-based cancer screening programmes or registries, has sought to identify efficient and effective approaches to cancer control by comparing data from screening programmes internationally (http://appliedresearch.cancer.gov/icsn/).
The ICSN conducts periodic assessments to obtain information on policies and activities related to the organization, delivery and evaluation of cancer screening programmes in its member countries. 1 3 In 2007-2008, we conducted an assessment with 26 countries to gain updated information from previous assessments. We present a summary of this assessment for countries that have both cervical and breast Screening programmes. Our aim was to describe the structure and characteristics of cervical screening programmes within countries that have established population-based breast cancer screening programmes. To our knowledge, this is the first description of both breast and cervical cancer screening programmes in developed countries.
Methods
The study population comprised ICSN member countries in 2006 when the programme assessment was initiated. The assessment was administered by the US National Cancer Institute and sent by email in May 2007 to individuals within these countries who were responsible for the implementation or evaluation of organized cancer screening programmes. Representatives were sent up to four emailed reminders to complete the survey. Of the 26 ICSN representatives contacted, 23 responded (response rate = 88%).
The assessment included questions on breast and cervical cancer screening programmes. As the ICSN focused only on breast cancer screening programmes from 1988 to 2006, similar questions on breast cancer screening programmes were included from past assessments. The 2007-2008 assessment was the first collection of information on cervical cancer programmes; therefore, only breast and cervical cancer screening programmes are included in this synopsis. The questionnaire was organized into separate sections to facilitate completion by more than one individual, because in some countries the person responsible for the breast cancer screening programme is not the same individual who oversees the cervical cancer screening programme. Items addressed the structure and policies of the country's breast and cervical cancer screening programmes, including the number of women screened, screening rates, detection methods used, recommended screening intervals, age groups covered, methods for recruitment and use of quality assurance guidelines. Additional items in the breast cancer screening section covered diagnostic follow-up, use of double reading, and performance and programme impact measures (information available on the ICSN website http://appliedresearch.cancer.gov/icsn/). The assessment also included a question on whether the human papillomavirus (HPV) vaccine had been approved for use (yes approved, under review by government agency, and not approved).
Six of the 23 countries participating in the assessment (26%) indicated that they did not have an organized cervical cancer screening programme (Germany, Luxemburg, Czech Republic, Turkey, Switzerland) or did not have data available from their organized cervical cancer screening programme for this assessment (Netherlands) and were excluded from this comparison. US data represented in prior assessments have come from the Breast Cancer Surveillance Consortium (BCSC), a geographically diverse network of registries with data on population-based mammography.1–3,12 Because the BCSC does not capture information on cervical cancer screening, we also excluded the USA from this description. We examined characteristics of the remaining 16 countries with organized breast and cervical cancer screening programmes, and also made selected comparisons of breast cancer screening programmes with information collected in the 1995 and 1998 assessments.2,3
Results
Programme implementation
Organization of breast and cervical cancer screening programmes in 16 ICSN countries, 2007-2008
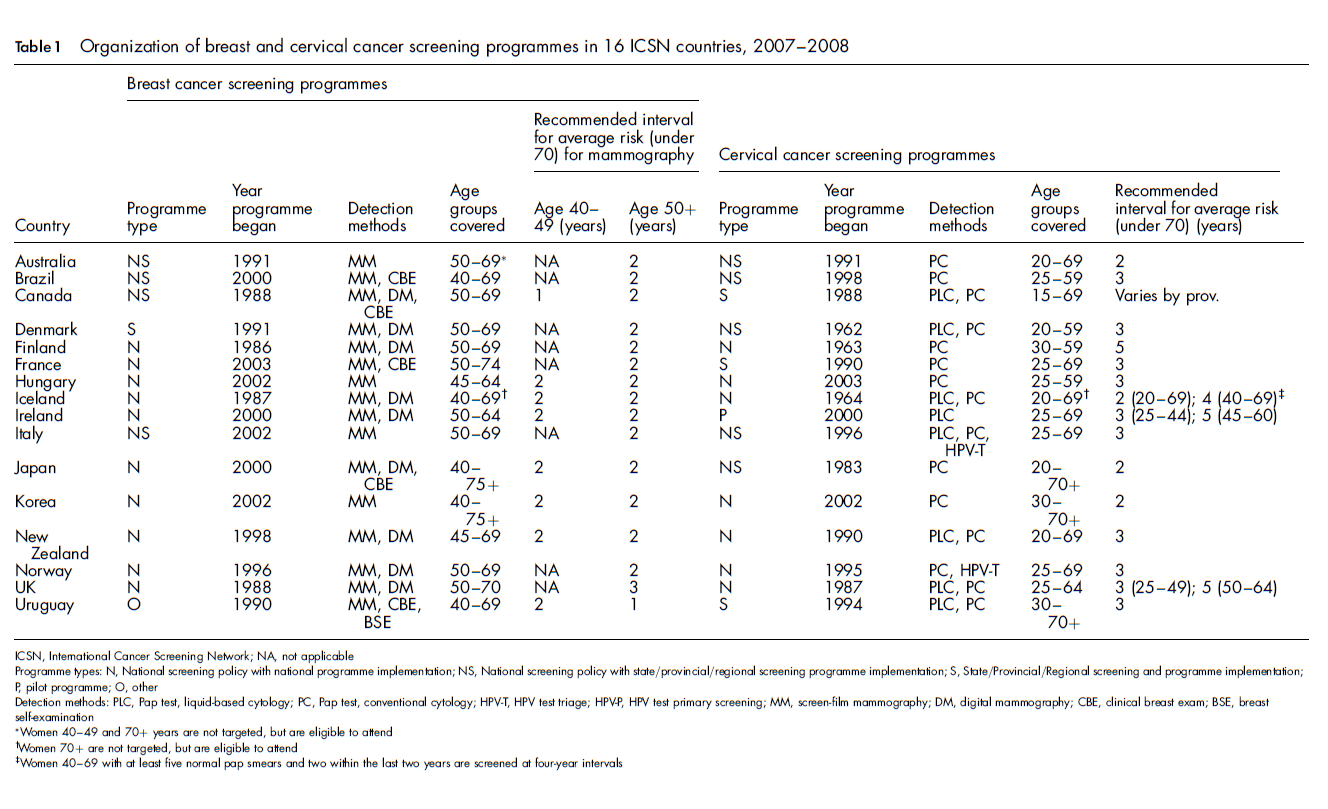
Women 40-49 and 70+ years are not targeted, but are eligible to attend
Women 70+ are not targeted, but are eligible to attend
Women 40-69 with at least five normal pap smears and two within the last two years are screened at four-year intervals
Methods of detection and adoption of new technologies
Adoption of new screening technologies was evident in cervical cancer screening programmes (44% use liquid-based Pap tests + conventional cytology; 13% use HPV-triage tests). All countries use screen-film mammography, and over half (56%) use both digital and screen film. This reflects a substantial adoption of digital mammography since the previous survey in 1995, when all ICSN countries were using only screen film mammography. 1 Five (31%) countries use clinical breast exam (CBE) despite limited evidence that CBE provides benefits on its own or combined with mammography. 13 15 This is a marked decline in the use of CBE from the 1995 assessment (58% reported). 1 We also noted a decline in reported use of breast self-examination from the 1995 assessment (23%) to only one country in 2007-2008 (6%). 1
Age ranges
Countries are not uniform in starting ages for cervical cancer screening: these range widely, from a low of 15 years (Canada) to a high of 30 years (Finland, Korea, Uruguay). For breast cancer screening, approximately half of the countries begin screening women at ages 40 or 45 years and half begin at age 50. There is variation in upper age limits for cervical cancer screening as some countries (Brazil, Hungary) stop screening at age 59 while others (Japan, Korea) screen women over the age of 70. However, most countries stop cervical cancer screening at age 69. In breast cancer screening, a few countries stop screening at 65 (Hungary, Ireland), most at 70, although some screen women over 75 years of age (Japan, Korea). There was little change in the age ranges covered for breast cancer screening programmes between the current and the 1995 assessments. 1
Screening intervals
For most countries, programmes recommend a 2-3-year interval for cervical cancer screening. Some countries recommend five-year intervals for women 45 years of age or older (Ireland and UK) or recommend four-year intervals for women over 40 years of age (Iceland) depending on their previous history of Pap smears. The recommended interval for breast cancer screening ranges from one to three years, with most countries applying a two-year interval for screening, also similar to the 1995 assessment. 1
Participation rates
Participation rates and target populations for breast and cervical cancer screening programmes in 16 ICSN countries, 2007-2008
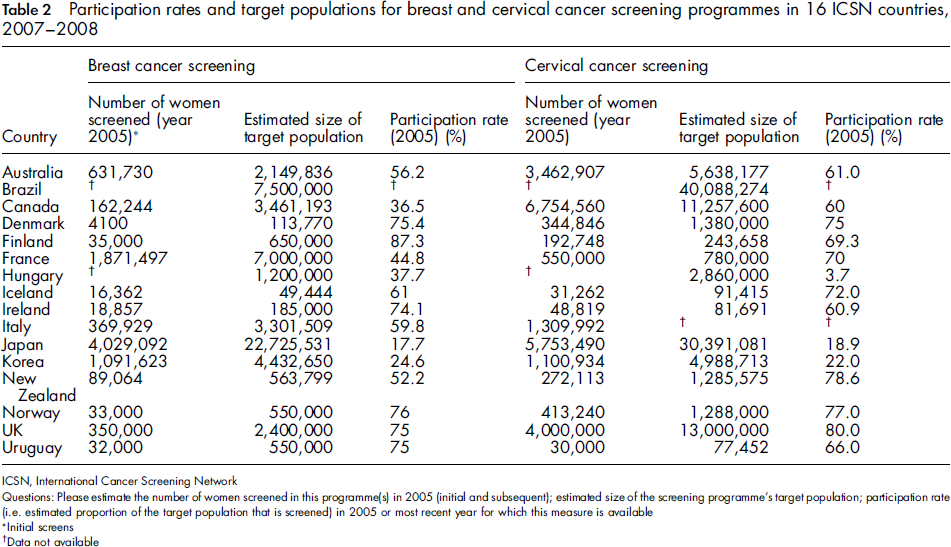
Initial screens
Data not available
Methods of Recruitment
Other characteristics of breast and cervical cancer screening programmes in 16 ICSN countries, 2007-2008
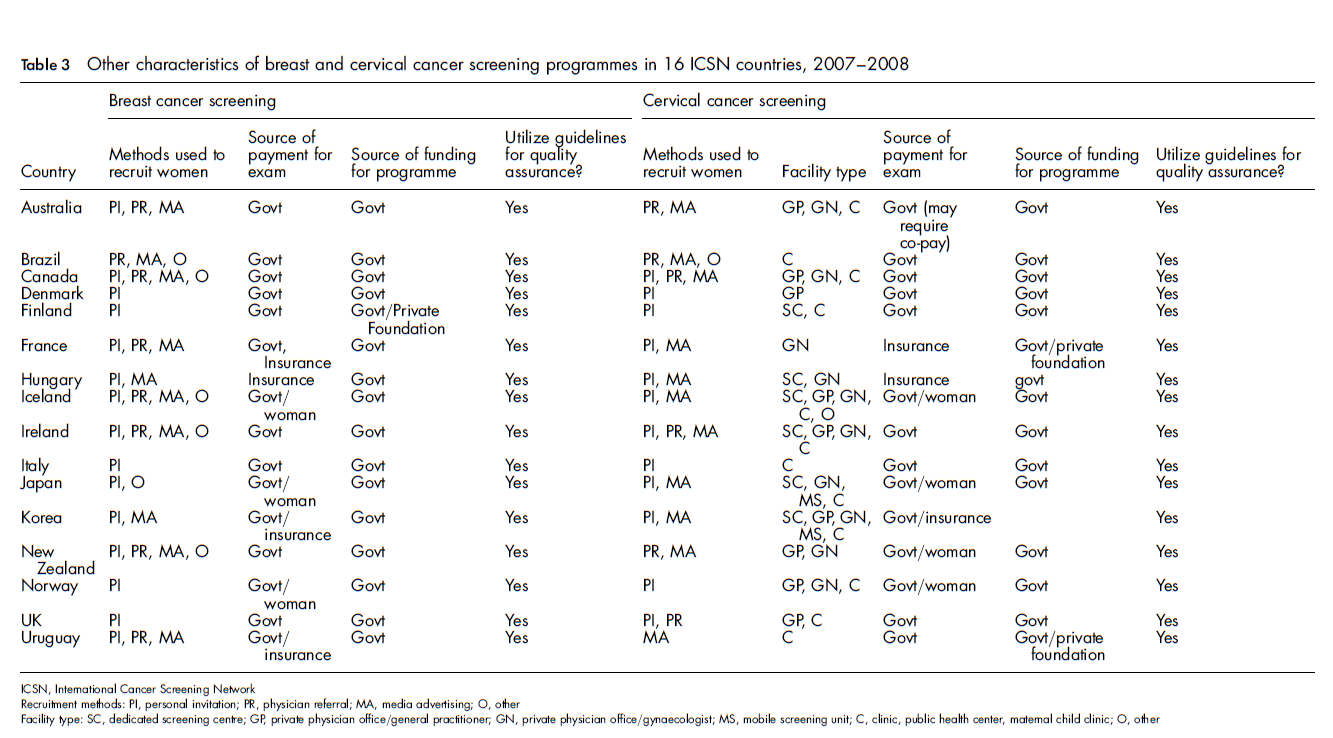
Quality assurance guidelines
All countries reported using guidelines for quality assurance in their cervical and breast cancer screening programmes, similar to the 1995 assessment 1 and a 1998 ICSN assessment on breast cancer quality assurance. 2
HPV vaccination utilization
We were particularly interested to learn more about the use of the HPV vaccine by ICSN
countries. Figure 1 shows that 11
countries had approved it for use and five more were reviewing evidence to consider its
use in organized programmes at the time of the assessment. Status of HPV vaccine approval in ICSN countries, 2007-8. Dark shading, HPV vaccine
approved; light shading, HPV vaccine under review; ICSN countries where HPV vaccine
not approved, Ireland and Finland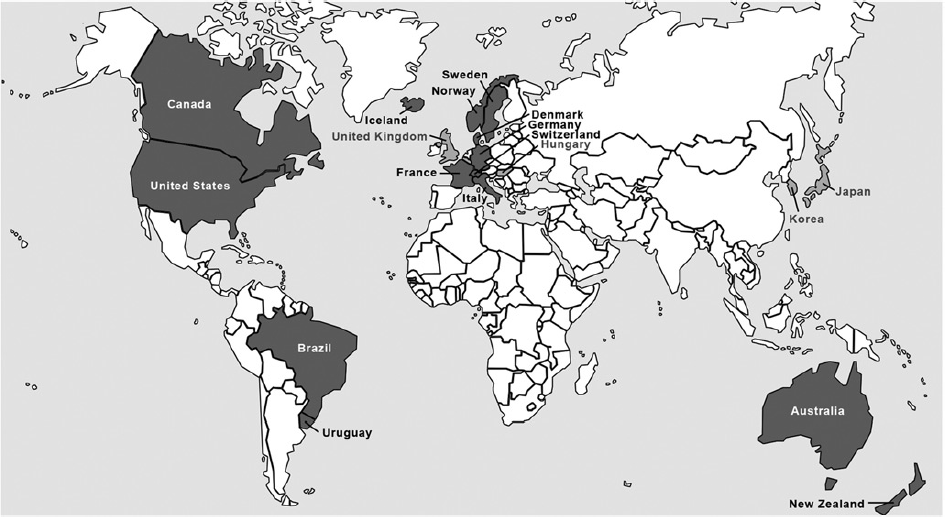
Discussion
We describe an assessment of countries participating in the ICSN that have both breast and cervical cancer screening programmes. Generally, the organization of the cervical cancer screening programme was similar to the organization of the breast cancer screening programme in the same country. We found substantial adoption of new screening technologies for both types of screening programmes. Countries are not uniform in the age ranges covered by their screening programmes, particularly in starting ages for cervical cancer screening. They are more uniform, though, in the screening intervals used. Participation rates varied widely across countries. All countries reported applying quality assurance guidelines in their screening programmes. Many have already approved the HPV vaccine for use in cervical cancer prevention and several more are considering its use.
While our assessment revealed that the organization of most cervical cancer screening programmes was similar to that of breast cancer screening programmes, overall there are fewer systematic, organized cervical than breast cancer screening programmes in ICSN countries. There are several possible explanations for this. Unlike breast cancer screening with mammography, there was never a randomized controlled trial to evaluate the efficacy of the Pap test for cervical cancer screening. 6 8 Rather, evidence from longitudinal studies suggested a decrease in cervical cancer incidence and mortality with the use of Pap smears. 7 Therefore, cervical cancer screening began in many countries as opportunistic screening or through the establishment of local organized screening programmes that later matured into larger, sometimes national, programmes. 16 Additionally, some countries had high coverage without an organized programme and it was not efficient for them to establish an organized screening programme. However, other countries had been using the Pap test since the 1960s, and it became increasingly recognized in the 1980s that organized screening programmes could optimize the use of resources. 7 Furthermore, while implementation of cervical cancer screening with the Pap test does not require complex equipment or dedicated facilities, this is not true for mammography, and many countries have chosen to implement breast cancer screening programmes on a larger scale. Additionally, because of the emergence of randomized controlled trial evidence, there was a relatively uniform time period over which many breast cancer screening programmes were started which was not the case for cervical cancer screening.
In describing breast and cervical cancer screening programmes, we also noted that cervical cancer screening programmes tended to have higher participation rates than breast cancer screening programmes. This could be because breast cancer screening programmes more often rely on physician referrals to recruit women into the programme than do cervical cancer screening programmes. Another possibility is that breast cancer screening usually requires attendance at a mammography facility while cervical cancer screening can occur at the time of a single visit to a variety of facilities, including general practitioner or gynaecologist offices, public health centres and maternal/child clinics.
However, it is challenging to assess and compare participation rates across different screening programmes. A participation rate is a performance indicator and each programme is distinct and covers populations with different characteristics. We assessed the participation rates for a given year, but participation rates can be greatly influenced by both the screening interval for a specific programme and the reference period over which the rate is calculated for each country. In addition to the participation rate, we included the size of the target population and the number of women screened because they are useful indicators of the scope of the screening programme. Therefore, while caution needs to be exercised when comparing and interpreting participation rates, these estimates can provide a useful snapshot of the performance of organized screening programmes. The ICSN currently is supporting a working group to further understand how countries define screening participation rates.
Our assessment shows that breast and cervical cancer screening programmes internationally continue to evolve through the adoption of new screening technologies. We noted that the countries implementing new technologies in breast cancer screening programmes also tended to adopt new technologies for cervical cancer screening, possibly related to the economic status of the country, the maturity of the programmes or to a systematic process for decisionmaking related to the adoption of new technologies within programmes. It will be important to understand how breast cancer screening programmes continue to evolve and incorporate new technologies, such as digital mammography and magnetic resonance imaging, and their impact on clinical care and outcomes. 17
In cervical cancer screening, programmes have had to consider the emergence of several new technologies, including liquid-based cytology, HPV test-triage methods and the HPV vaccine. Many countries have implemented liquid-based cytology and some, such as the UK, are planning to phase out conventional cytology. While studies have suggested reduced costs and increased specificity with the HPV-triage test, 10 only a few ICSN countries were actively using it in their cervical cancer screening programmes. One advantage of implementing new screening technologies in organized programmes is the potential to use screening resources more efficiently. 10 At the time of the assessment, most ICSN countries had either approved the HPV vaccine for use or were considering it. Several ICSN countries participated in the randomized controlled trial for the HPV vaccine (Denmark, Finland, Iceland, Sweden, Norway, Spain, the USA and the UK). 18 It will be important to monitor the implementation of the HPV vaccine by ICSN countries in the future as countries establish vaccine registries and possibly revise their screening policies based on vaccine use.
While this summary of breast and cervical cancer screening programmes provides a unique look at the organization of these cancer screening programmes internationally, a few limitations should be mentioned. The assessment was conducted at one point in time; screening programmes continually evaluate existing and new technologies and evidence so that changes in programme policies in one or more countries may have occurred subsequent to our study. We also only considered countries that participate in the ICSN; countries with organized breast and cervical cancer screening programmes that are not ICSN members therefore are not represented in this work. We did not include the USA in this analysis; although much of the screening that occurs in the USA is opportunistic, there are several organized breast and cervical cancer screening programmes, including those implemented by some integrated healthcare systems and by the Centers for Disease Control and Prevention (www.cdc.gov/cancer/NBCCEDP). Finally, it is challenging to document and compare screening programmes that are conducted within widely divergent healthcare contexts.
Overall, we found both similarities and differences in the organization of breast and cervical cancer screening programmes among countries participating in the ICSN. The comparison and reporting of organized screening programmes can aid existing programmes as well as countries that are planning implementation of new screening programmes. For example, countries in Latin America, which have a significant burden of cervical cancer, are actively restructuring their cervical cancer screening programmes after several attempts to implement programmes without much impact.19,20 As a positive development, restructuring of the screening programme has been associated with a decline in cervical cancer mortality in Chile. 20 Reporting on the structure of ICSN screening programmes in both mature and more recently established programmes promotes the understanding of service delivery in cancer screening and may serve as a resource for countries that are working to optimize programme resources.
In the past several years, there has been a substantial increase in the number of countries interested in participating in ICSN meetings and responding to assessments. Additionally, there is a growing interest within the ICSN member countries in expanding beyond breast cancer screening programmes to not only address issues in cervical and colorectal cancer screening but also to anticipate the future of population-based cancer screening for other cancer sites such as prostate, ovary and lung. The ICSN will continue to document and compare the structure of screening programmes in cervical, breast and colorectal cancer as well as monitor how countries adjust programmes to take account of changes in the screening population and technologies. The ICSN also might serve as a network to stimulate research in comparative effectiveness through its ability to examine screening technologies applied in clinical practice at the population level.
Footnotes
Acknowledgment
We gratefully acknowledge the following members of the International Cancer Screening Network who provided information about their screening programmes: Julianne Quaine and Alison Lang (Australia), Ronaldo Silva Correa (Brazil), Jay Onysko (Canada), Adam Svoboknik (Czech Republic), Elsebeth Lynge (Denmark), Nea Malia and Ahti Anttila (Finland), Helene Sancho-Garnier (France), Dominik Deitz (Germany), Imre Boncz (Hungary), Kristjan Sigurdsson (Iceland), Tony O'Brien (Ireland), Gad Rennert (Israel), Eugenio Paci (Italy), Hiroshi Saito (Japan), Astrid Scharpantgen (Luxemburg), Won Chul Lee (Korea), Jacques Fracheboud (Netherlands), Brian Cox (New Zealand), Solveig Hofvind (Norway), Jean-Luc Bulliard (Switzerland), Caner Fidaner (Turkey), Sue Moss (UK), Rod Walker (USA), Gonzalo Pou (Uruguay). We also thank Michael Coffey at Information Management Systems, Inc for programming support for the development and synopsis of the web-survey and Kathy Sedgwick at NOVA Research Company for her logistical support for the ICSN.
