Abstract
Objectives
To investigate the effect of general practice (GP) and general practitioner (GPR) endorsement for faecal occult blood test (FOBT)-based screening on maintenance of participation in screening over four successive screening rounds.
Setting
South Australian residents aged ≥50 years.
Methods
Random selection of four groups (n = 600 per group): one from the Commonwealth electoral roll (ER) and three from the combined patient lists of two collaborating GPs (GP1, GP2, GP3). Subjects were mailed offers to screen using a faecal immunochemical test over four successive rounds, spaced approximately 18 months apart. The GP1 and ER groups were invited to screen without any endorsement from a GPR or medical practice; GP2 invitees received an invitation indicating support for screening from their medical practice; and GP3 invitations were printed on practice letterhead and were signed by a GPR.
Results
Multivariate analyses indicated that initial participation as well as re-participation over four successive rounds was significantly enhanced in the GP2 (39%, 42%, 45% and 44%) and GP3 groups (42%, 47%, 48% and 49%) relative to the ER group (33%, 37%, 40% and 36%). The analyses also indicated that 60–69 year olds were most likely to participate in all rounds (relative risk [RR] 1.49, 1.39, 1.43 and 1.25), and men were generally less likely to participate than women in all screening rounds (RR 0.86, 0.84, 0.80 and 0.83).
Conclusions
Associating a GPR or medical practice of recent contact with an invitation to screen achieves better participation and re-participation than does an invitation from a centralized screening unit. Furthermore, enhanced participation can be achieved by practice endorsement alone without requiring actual GPR involvement.
Introduction
Current colorectal cancer prevention strategies focus on early detection through internal examination of the bowel during sigmoidoscopy or colonoscopy, or via faecal occult blood test (FOBT) which detects minute amounts of blood in stool and facilitates the detection of neoplasia at curable stages.5–7 Randomized controlled trials5,6 show that population screening by FOBT significantly reduces mortality by 15–35% on an intention-to-screen basis. 8 These reductions at the population level were achieved with screening participation rates of a little over 50%.5,9 In those who actually participate – that is, perform FOBT biennially – mortality can be reduced by 40%. 5 Further decreases in mortality could yet be achieved through improvements in the population participation rate with likely added benefits in cost-effectiveness. 10
Several factors have been shown to impact screening behaviour, including sociodemographics, knowledge of cancer, attitudes toward screening and perceived self-efficacy. 4 Studies also show that patients are more likely to act on an offer to screen if the invitation originates from a credible and trusted source such as a general practitioner (GPR) or primary care practice (general practice [GP]).4,11,12 In Australia, the practicality of actual GPR involvement in colorectal cancer screening depends on many factors including traditionally a lack of mechanism for commonwealth reimbursement for colorectal cancer screening. While reimbursement has now become possible, although within a narrow context, other barriers include a lack oi time which may prevent GPRs advising their patients about the benefits of screening. Also, in countries such as Australia where there is choice of GP it is difficult to link people to, for example, the GPR of most recent contact for overall management of the screening pathway, as there is no feasible way of identifying them. 11
Previously, we have examined the impact of endorsement by the primary care practitioner on initial uptake of screening in a colorectal cancer prevention programme. 11 We sought to address the difficulties associated with actual GPR involvement by coordinating a screening programme through a central agency, which had identified eligible persons by obtaining medical practice patient lists. We then manipulated the extent to which the invitees’ GPR might be considered to be involved in the screening offer, although they never personally were. More specifically, participants in the previous study 11 received one of the following: (1) a standard postal invitation from a central screening service; (2) an invitation from a screening service, which was endorsed impersonally by the participant's medical practice; or (3) an invitation printed on medical practice letterhead indicating the offer was endorsed by the participant's GPR of most recent contact. Results indicated that invitations with either practice or practitioner endorsements (Invitation types 2 and 3) resulted in significantly better participation rates than did the invitation without practice or practitioner endorsement (Invitation 1). Importantly, endorsement modes required minimal involvement of the GP or GPR as they were coordinated by the screening agency, thus eliminating the time-barrier issues already noted. 11
The relative value of different methods of invitation needs to be thoroughly explored in order to understand how best to implement strategies to maximize reductions in mortality from colorectal cancer without overwhelming health service resources. Having already established that GPR endorsement does significantly improve participation without direct involvement of the GPRs themselves, 11 we have continued our earlier study in order to explore whether these effects are stable across multiple screening rounds, since re-participation in screening is crucial to achieving actual benefit. Participants in our earlier study 11 received a further three invitations to screen for colorectal cancer, making for a total of four offers over the course of approximately five years. If the effects of GPR endorsement are reliable, it would suggest that invitation types endorsed by medical practices could be introduced at the population level as a means of increasing participation. Our specific aims were: (1) to assess the effect of GPR endorsement on participation rates over three subsequent rounds of colorectal cancer screening; and (2) to assess whether the demographic variables sex or age were associated with re-participation in subsequent screening rounds.
Methods
Study population
Participants were South Australian residents selected from two sources. The first source was GP patients. The lists of patients aged 50 years or over who had no known medical conditions that precluded offers of colorectal cancer screening were obtained from two primary care practices in Adelaide's southern suburbs. The lists were combined and three groups (GP1, GP2 and GP3) of 600 people were randomly selected. Eligibility criteria required that all participants must have consulted with a GPR from either of the two practices within the 12 months preceding the first round screening invitations.
The second population (electoral roll [ER]) was a comparison group drawn from specific place of residence postcodes of people aged over 50 years present on the ER of the Australian Electoral Commission. The purpose of this population was to determine whether there were differences in the behaviour of people recruited from the ER compared with those recruited from GPs. Postcodes were chosen that were adjacent to the most common place of residence postcodes of the primary practice patients, and were comparable for socioeconomic status as measured by the Socio-Economic Index for Areas (Australian Bureau of Statistics). Six hundred invitees were randomly selected from this group. The process for random selection for both GP and ER groups is described in our previous report. 11 Before first round invitations were posted, names and addresses were cross-checked between groups to ensure that no person was selected for more than one group. A check was also performed to ensure that if two persons had been selected who resided at the same address, both were assigned to the same experimental condition (i.e. received the same invitation).
Offer of screening
Similar invitations to screen were used for each treatment group. Invitations differed in terms of invitation letterhead and type and level of endorsement. Formats for invitations were as follows:
Invitation 1 to ER and GP1 groups (no GP or GPR endorsement): An invitation sent on central screening service letterhead (Bowel Health Service, Repatriation General Hospital), signed by the screening coordinator, without any indication that the invitee's GP or GPR was involved.
Invitation 2 to GP2 group (GP endorsement): An invitation sent on central screening service letterhead, signed by the screening coordinator and endorsed impersonally by the participant's medical practice by simply stating that the relevant (named) practice supported the screening offer.
Invitation 3 to GP3 group (GPR endorsement): An invitation sent on the invitee's medical practice letterhead, indicating that the screening offer was endorsed by the practice and signed by the person's GPR of most recent contact.
The first round of invitations was distributed in June of 1999 and has been reported elsewhere. 11 Subsequent rounds commenced in November 2000, February 2002 and September 2003, respectively. Participants received identical invitations for all rounds. The invitational kit included: (a) a bowel cancer information sheet; (b) a brief questionnaire confirming personal details and preferred doctor for follow-up; and (c) a faecal immunochemical test (FIT). In rounds one and two, the FlexSure OBT (Beckman Coulter, Fullerton, CA, USA) three-sample stool kit was used. In round three approximately half of the participants in each group received Flexsure OBT (Beckman Coulter, USA), while the other half received InSure OBT (Enterix Inc, North Ryde, Australia) two-sample stool kit. In round four, all participants received the InSure kit. At each round, participants were asked to complete the test according to the manufacturer's instructions and return the samples by post to the central screening service. Reminders were sent by post at six weeks after the initial letter, if a completed collection card had not been received.
Inclusion and exclusion criteria for screening rounds two to four
All round one invitees were theoretically eligible for re-screening in rounds two to four. In each of these subsequent rounds however, invitations resulted in additional written and telephone contact with invitees or their close relatives and at times, with their GPs. Sometimes, this information influenced subsequent eligibility. Specifically, people were excluded from further screening offers if they were deceased, had requested no further contact with the screening programme (whatever the reason), were in an alternative bowel cancer screening programme, had tested FOB positive in a previous round and/or had had an adequate colonoscopy within the last four years, were not contactable at the address provided by the practices or shown on the ER, or could not understand the instructions. Furthermore, in rounds three and four only participants aged <80 years were invited to participate.
FOBT, development and follow-up
Test development was undertaken by or on behalf of the Bowel Health Service according to the manufacturer's instructions. The overall result for a participant was considered positive if any of the returned samples tested positive, and they were advised of the results (positive or negative) within five days of receipt of samples. Both the participant and the nominated GPR were notified in the case of a positive result for follow-up procedures.
Outcome measures and analyses
Participation was defined as the return of a completed FIT within 12 weeks of posting the initial kit, and was the key outcome variable. We used multinomial logistic regression to determine the significance of invitational type, age and sex on rescreening behaviour. Each of the potential predictors was investigated in combination – i.e. multivariate models – for each round. We included test type in the round three regression model to control for differences in participation attributable to these different technologies.
Results
Characteristics of treatment groups
A comparison of the demographic characteristics of treatment groups as previously 11 reported is shown in Table 1. As can be seen, the groups were well matched for age distribution and sex and they did not differ significantly on these variables. Similarly, there was good representation of each medical practice within the different GP invitation groups. Table 1 also presents data concerning the number of screening offers per round. As can be seen, the rate of attrition did not differ across the groups with a comparable amount of offers being distributed at each of the screening rounds. Overall, there was on average 25% attrition from round one to four for each group.
Comparison of demographic characteristics (n [%]) of groups according to screening invitation method and the number of invitations per screening round
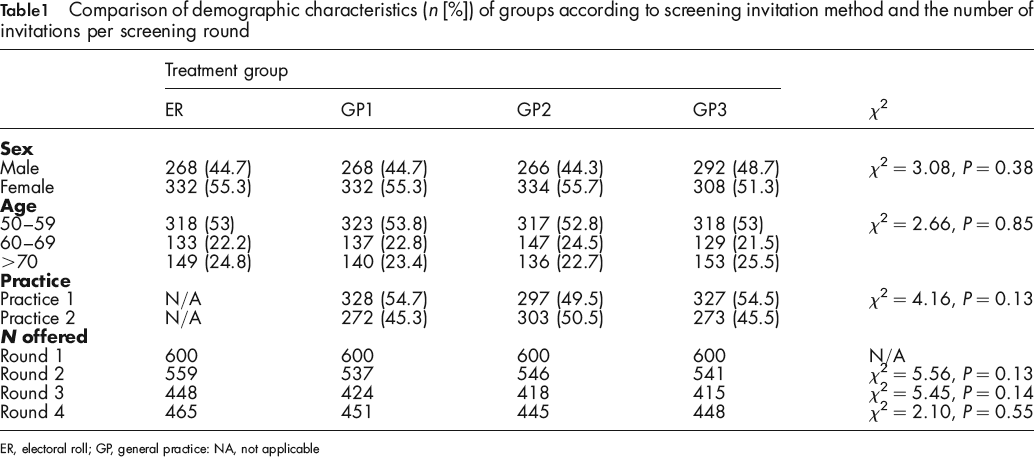
ER, electoral roll; GP, general practice: NA, not applicable
Participation rates, mode of offer, sex and age
Figure 1 displays participation rates over the course of screening rounds by invitational group. There is a consistent difference between the groups associated with the level of GP involvement in the offer. The ER and GP1 modes of offer resulted in comparable participation rates. However, participation rates in GP3 and GP4 groups exceeded those of the former two.
Participation rates for each group per screening round. ER, electoral roll; GP, general practice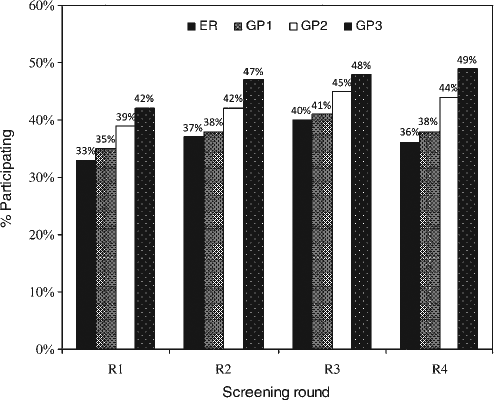
The results of the MLR modeis for each screening round are presented in Table 2. Analysis at each round included only those participants who had received an offer. We also expanded age at invitation to four categories to improve interpretation of any significant age effects observed (50–59, 60–69, 70–79 and 80+ years).
Likelihood (RR) of participating in screening for each of four rounds (R1–R4) based on sex, age at initial offer and mode of offer
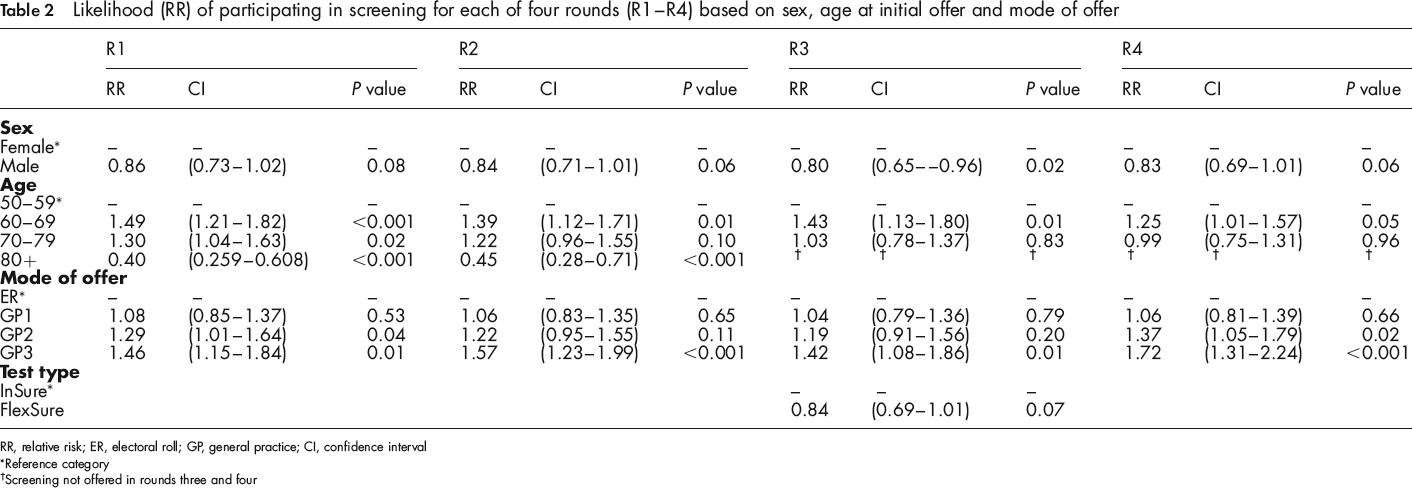
RR, relative risk; ER, electoral roll; GP, general practice; CI, confidence interval
Reference category
Screening not offered in rounds three and four
As can be seen, there was a tendency for men to screen and rescreen less than women across all four rounds, but this trend is statistically significant only in round 3. The data also show that invitees aged 60–69 are significantly more likely to re-participate than those 50–59 years, and there is a decreasing trend for increased participation in the 70–79 age group in rounds one to three. The effect of the different test technologies in round three was not statistically significant, although there is some indication those who received FlexSure OBT were less likely to participate.
GP1 invitees did not participate more than the ER group, as expected. Participants receiving the GP2 invite – endorsed impersonally by the medical practice – were more likely to participate than ER invitees in rounds 1 and 4 only. GP3 invitees were significantly more likely to participate than ER invitees in all four screening rounds. Both the GP2 and GP3 groups screened and maintained rescreening at significantly higher rates than seen in the ER and GP1 groups. Post hoc comparisons (χ2 tests) showed, however, that the GP3 participants were not significantly more likely to participate than those in the GP2 group in any of the four screening rounds (R1 [χ2(1) = 0.68, P = 0.41]; R2 [χ2(1) = 3.40, P = 0.07]; R3 [χ2(1) = 1.14, P = 0.28]; R4 [χ2(1) = 2.29, P = 0.13]).
In a final step we calculated the total number of compliant rounds for each participant. We then performed a one-way analysis of variance (ANOVA) to test for differences between the average number of screenings per group over the course of the study. The mean number of screenings for each group was as follows: ER M = 1.3, standard deviation (SD) = 1.6; GP1 M = 1.4, SD = 1.5; GP2 M = 1.5, SD = 1.6; GP3 M = 1.7, SD = 1.6. The ANOVA revealed a significant effect [F(3,2242) = 4.71, P = 0.003]. Bonferroni post hoc analyses revealed the only significant mean differences were between GP3 and ER (P < 0.001), and between GP3 and GP1 groups (P = 0.01).
Discussion
The results of the present study indicate that invitation strategy and design can have a powerful influence on initial colorectal cancer screening behaviour and on maintenance of differentials in behaviour due to different invitation strategies. More specifically, invitations signed by a GPR resulted in the highest level of participation in each round and this was significantly better when compared with the standard invitation from the central screening agency. We can be confident this effect is not an artefact of health-seeking behaviour in the GP groups because GP1 and ER invitees showed similar levels of participation. Better participation rates when an invitation is endorsed by a GPR is consistent with screening behaviours for other cancers including breast and cervical.13,14
It is unlikely that GPs have the capacity to coordinate mass colorectal cancer screening and this is reflected in Australia's National Bowel Cancer Screening Programme, 15 which is federally coordinated and operates centrally, with little GPR involvement in the invitation and FOBT screening process. However, results of this study show that screening participation can be significantly enhanced without the GPR interacting with an invitee. The GP2 invitation strategy which mentioned the invitee's medical practice resulted in better participation than the ER and GP1 groups in rounds one and four and, overall, both GP2 and GP3 groups showed better participation rates than the ER and GP1 groups; with an average of 5% better participation in GP2 and 10% in GP3. The influence of the GPR on enhancing colorectal cancer screening participation rates might be harnessed through more general endorsements from GPRs’ professional bodies and could accompany screening invitation packs.
At the population level, a 5–10% increase in participation is substantial and would result in the detection of more curable cancers. The question becomes whether impersonal endorsement – i.e. GP2 invitations – is sufficient enough to warrant inclusion in population screening programmes, or whether a more personal endorsement by a relevant medical practice and GPR should be preferred. Associating the GPR with the personalized offer is likely to be interpreted by people in various ways and although these mechanisms were not explored, it is possible that it is interpreted as advocacy for the benefit of screening and our results show that it is relatively simple to include advocacy by the GPR and that it translates into better screening behaviour.
As already noted, sociodemographic variables have been linked to participation in screening for colorectal cancer,16–18 and it is suggested that women are more likely to participate in colorectal cancer screening by FOBT. 17 The difference is modest, however, and men were only significantly less likely to participate than women in round three, although this tendency was persistent throughout all rounds. In our initial publication of round one results, 11 we used univariate analyses and did not find this effect. The multivariate models reported here, however, show that after accounting for the effects of age and invitational type, sex is most likely a significant predictor of screening behaviour. In line with previous findings, 19 our analyses show that the 60–69 year cohort were most likely to participate. Many individuals in the older cohorts may have other health problems that inhibit or preclude their participation, magnified by the reality that over the five years of the study, many reached an age where participation decreases irrespective of invitation strategy. The poorer participation of the youngest group is concerning however, and methods of increasing participation in this group should be identified in order to achieve the maximum reduction in colorectal cancer incidence and mortality.
This study shows for the first time that advocacy of screening by GPRs significantly improves initial uptake as well as ongoing participation in screening for colorectal cancer by FOBT. As re-participation in screening is crucial to gaining benefit, such advocacy should be included wherever possible in screening programmes. It may be difficult in some healthcare systems to include personalized advocacy in large populations where there is freedom of choice in GPR but such information is obtainable at the first round and could be included subsequently. Future studies should explore the relative value of other forms of advocacy including impersonal endorsement by respected organizations such as cancer councils and public health organizations.
