Abstract
Objective
We report performance indicators and costs of the first round of a cervical cancer screening programme based on the human papillomavirus (HPV)-DNA test.
Methods
We implemented a demonstration study using HPV as the primary test in Guidonia, Italy (90,000 inhabitants). All women aged 25-64 were invited to undergo a Hybrid Capture II high-risk HPV test. Two cervical samplings, smear and liquid, were taken. The smear was dyed and interpreted only for HPV-positive (HPV+) women. Women with a non-negative Pap smear were referred for colposcopy, women HPV+/cytology negative were referred to one-year follow-up with HPV. A cost-analysis indicated the price at which the HPV-based and cytological screening would cost the same per screened woman and per lesion found.
Results
Of 24,000 women invited, 7639 accepted and 427 (5.6%) were HPV+; 141 (34%) of these had a non-negative Pap test, and 20 cervical intraepithelial neoplasia (CIN) 2 or higher were found (positive predictive value 15%). Compliance to one-year follow-up was 58% (166/286); 90 (54%) were HPV-positive and five additional lesions were found (positive predictive value 9%; overall detection rate 3.4/1000). The cost analysis showed that at a price of 8.3 euros per HPV DNA test, the strategy using HPV as primary test followed by cytological triage would cost the same per screened woman, while at a price of 12.7 euros it would have the same cost per CIN2+ found.
Conclusion
The workload for management of positive women was similar to cytological screening. Low compliance to one-year follow-up was the main barrier to effectiveness. The price of HPV test should be about 9 euros to maintain the same screening budget, and can go as high as 13 euros per lesion found.
Introduction
Evidence from randomized controlled trials showed that HPV testing is more sensitive than cytology in identifying high-grade precancerous lesions, even though it is less specific. 2 4
At present, triage of borderline4,5 or low-grade cytology 6 with HPV testing is universally accepted and only cost-effectiveness limitations represent a matter of concern.6,7 There is also agreement that women who are HPV-negative, independent of their cytology, are at negligible risk for high-grade lesions, according to both our knowledge of the natural progression of the disease and the results of the National Tuberculosis Curriculum Consortium (NTCC), the Swedish and the Dutch trials.3,8-11
Theoretically, the logical consequence of all of this evidence should be to invert the testing order: first the most sensitive and then the most specific.12,13 The main advantage of this model is that we can distinguish three groups of women with different risk levels: (1) the HPV-negative with very low risk; (2) the HPV-positive and cytology-negative, with intermediate risk and a referral at shorter intervals; and finally (3) the HPV-positive and cytology-positive with the highest risk and immediate referral for colposcopy.
This protocol has almost no risk of increasing over-treatment with respect to cytology, since no cytology-negative women would be referred directly to colposcopy. The only source of over-treatment could be the early rescreening (1 year) for women who are HPV-positive and cytology-negative, but recent results from the follow-up of the Swedish and Dutch trials demonstrated that if this overdiagnosis exists at all, it is very small.10,14
The second edition of the European Guidelines on Quality Assurance in Cervical Cancer Screening 15 clearly states that Pap tests continue to be the standard for cervical cancer screening in Europe, but also is open to HPV-based screening if randomized controlled trials were to show lower incidence of cervical intraepithelial neoplasia (CIN)3+ in subsequent screening rounds among women who were HPV-negative compared with those who were cytologically negative in the first screening round. This evidence is now available.10,14,16 Pilot studies, based on validated HPV-DNA tests, such as Hybrid Capture n, can be implemented in an organized screening programme with careful monitoring of the quality and systematic evaluation of the outcomes, adverse effects and costs.
The introduction of new technology into a mass screening programme also has to take into account different issues regarding the accuracy and efficacy of the test; 17 in particular, we should address the availability of human and financial resources as well as the applicability of new technologies and new protocols. Some pilot studies were implemented several years ago, but mostly in research settings. 18 21
Aim of the study
A feasibility study involving one out of four districts in one of 12 local health authorities (Aziende Sanitarie Locali-ASL) was planned in Lazio region to answer the following questions:
acceptability of the HPV as the primary test in the target population;
feasibility of the cytology triage protocol, particularly focusing on compliance at one-year follow-up;
cost-effectiveness of the protocol evaluated at different price levels for HPV testing.
Materials and Methods
Setting
Lazio is a region of central Italy with a population of 5,561,017 according to the 2008 census (ISTAT). 22 The regional territory is divided into local health authorities (ASL), geographically based healthcare organizations responsible for assessing health needs and providing comprehensive medical care for a defined population.
The study was planned for the ASL Roma G, including the eastern outskirts of Rome and a rural area of the region. The ASL had a cytological screening programme that actively invited all residents aged 25-64. Only one of four districts, Guidonia, was identified as the study population (target population about 26,000 women). The screening programme had not previously invited the population of the Guidonia district.
The pilot study started in June 2007; here we report the results for women invited before February 2008 and tested before May 2008. The follow-up for colposcopies was updated in September 2009.
The pilot study started a few months before the evidence from randomized trials was fully available, and consequently the protocol was approved by the Ethics Committee of the ASL as an experimental procedure.
Screening protocol
The whole target population was invited in the period June 2007-January 2008. Four sampling units were active with 20 dedicated midwives.
A double sampling, conventional smear and Specimen Transport Medium (Digene Corporation, Gaithersburg, MD, USA) for HPV testing were performed on all the participating women. The HPV tests were performed in the local laboratory. After the results of the HPV testing the slides of women who tested positive were dyed and interpreted by a single cytologist. Only women with cytology ASCUS (atypical squamous cells of undetermined significance) or more severe (see next paragraph) were referred for colposcopy, while women with negative cytology were referred for one-year re-check with HPV test. The entire protocol is summarized in the flow chart (Figure 1).
Flowchart of the screening process. The numbers present the results of the pilot study. * Including three unsatisfactory smears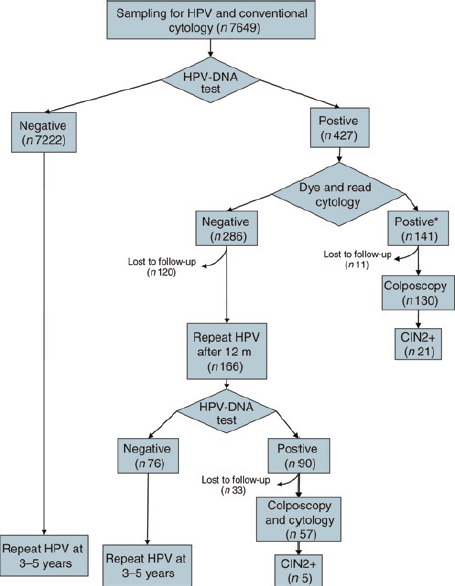
Molecular and cytological procedures and quality controls
Cytology was classified according to the Bethesda 2001 guidelines. 23 An external quality control was scheduled and was particularly intensive for the first phase of the study. It was based on a review of all cytology slides by an external supervisor.
HPV testing was done by using the Hybrid Capture II assay (Digene Corporation, Gaithersburg, MD, USA). Only the group of probes designed to detect high-risk HPV types 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 59 and 68 was used. HC2 results were expressed as the ratio of the specimen's light emission to that of three concurrently tested 1 pg/mL HPV-DNA controls (relative light units [RLU]). Therefore, RLU is a measure of the specimen's viral concentration relative to 1 pg/mL.
Outcomes
The following outcomes were assessed:
The participation rate (number of women who underwent the test/number of women invited). This outcome will be compared with the participation in the other programmes in the Lazio region and with the other districts in the same Local Health Unit (in the conventional Pap test);
HPV positivity rate (number of positive tests/number of tests performed);
Cytology positivity rate among HPV-positive women (number of ASC-US cytology or greater/total number of Pap tests performed);
Unsatisfactory cytology among HPV-positive women (number of women referred for repeat cytology/total number of Pap tests performed);
Detection rate (DR) (number of CIN2 or more severe histology/number of women screened).
Economic analysis
Parameters for the economic analysis
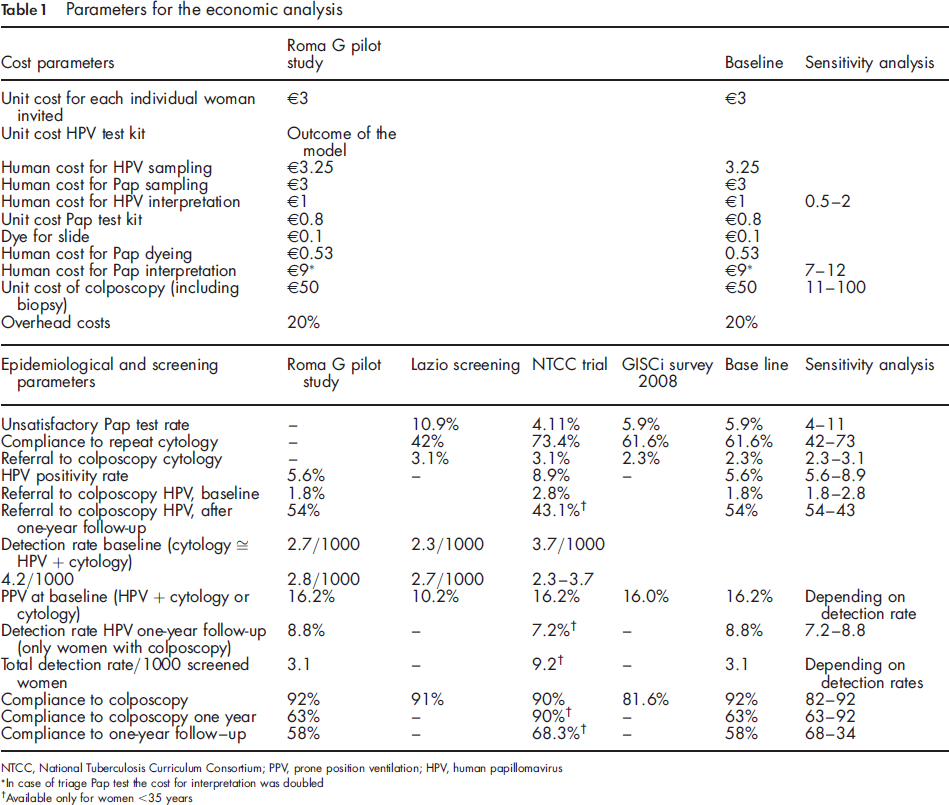
ln case of triage Pap test the cost for interpretation was doubled
Available only for women <35 years
Given the short time period, no discount rate on costs and outcomes was applied. The perspective taken was that of the Regional Health Authority.
Cost assessment
We included only direct costs associated with personnel, material and equipment.
Tests were divided into three parts: (a) cervical sampling, (b) laboratory analyses and (c) further examinations in the case of positive test results (colposcopies, biopsies and 1-year rescreening). For each phase, unit costs for personnel and material were computed. Costs are expressed in euros (€).
We assumed that the laboratory was operating fully: we ran the analysis considering the cost per test dividing the cost of the laboratory by the total number of tests potentially performed in one year.
Through an equivalence-point analysis, we identified the ratio between the total unit cost of an HPV-DNA test and that of one Pap test, which resulted in an equal overall cost per high-grade lesion detected.
Outcomes assessment
The DR for the comparator (conventional cytology) was taken from the National Centre for Screening Monitoring survey conducted for the Ministry of Health. 24 The relative DR of HPV was derived from the NTCC study results.3,9
The main outcome of the analysis was the cost (i.e. resource consumption) per lesion detected by different screening strategies.
Sensitivity analysis
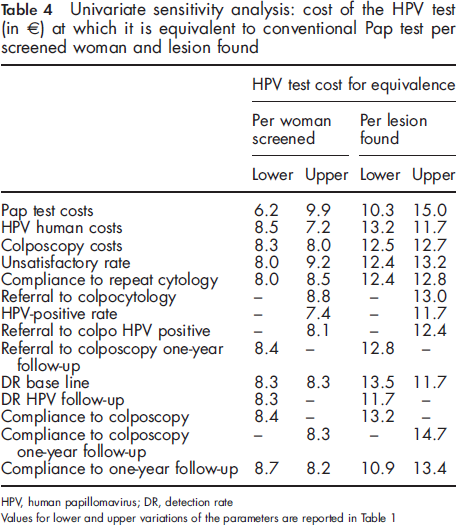
We performed a univariate sensitivity analysis for all parameters that were strongly uncertain. Table 1 shows the parameters and their range of variation. We assumed a constant DR for parameters regarding the referral for colposcopy and the positivity rate.
Results
Compliance
Compliance to screening invitation was 32.6% (7649/23,488). The average compliance in the same period in Lazio was 21.8%, while compliance in the other districts of the same ASL in the previous year was 17.7%. In 2006 average compliance to screening invitation in all of Italy was 38.5%.
Compliance to one-year follow-up was 58% (166/286). It was strongly influenced by mode of contact: in the group invited directly by phone, compliance was 66%; after a complaint about privacy from one woman, the ASL decided to invite the remaining women by letter alone, and for this second group compliance was 34%.
HPV positivity rate
Out of 7649 screened women, 427 (5.6%) tested positive (1 pg/μL cut-off), and all samples were satisfactory for testing. The age-group-specific prevalence is reported in Figure 2.
Age-group specific HPV-DNA prevalence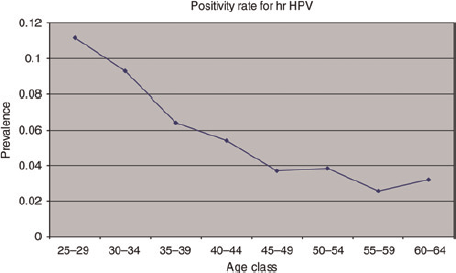
Among women referred for one-year follow-up, the positivity rate was 54.2% (90/166).
Cytology results
Distribution of cytological diagnoses among 427 HPV-DNA-positive women
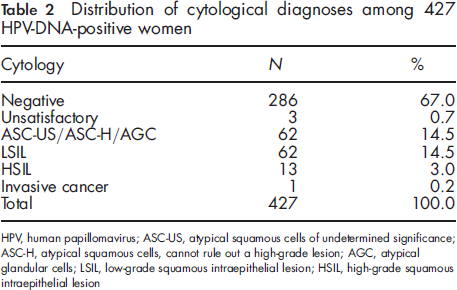
Overall, taking into account HPV and cytology results, 5.6% of the results were non-negative, 1.8% referred to colposcopy and 3.8% referred to one-year follow-up. More than 8% of results in nationwide screening were non-negative (referred to colposcopy, immediate repeat and 6-month or 1 year follow-up), while non-negative results in the Lazio region screening programmes were 14%.
Colposcopy and histology results
Colposcopy compliance was 92% (130/141) at baseline. The biopsy rate was 47% (61/130). Among the 61 women for whom the biopsy was considered necessary, five refused, four were negative, 31 had a CIN1 and 21 had a CIN2 or more severe.
The total DR at baseline was 2.7/1000 (21 CIN2+ out of 7649 screened women); the average DR in Italy is 2.7/1000, in the Lazio region it is slightly lower (2.3/1000). Compliance to colposcopy at follow-up was 63.3% (57/90).
Additionally, five CLN2+ were found in the 90 women who underwent one-year follow-up (DR) out of the women referred to one-year follow-up = 30/1000). Consequently, the overall DR was 3.4/1000. Two of these cases were invasive cancers, one squamous and one adenocarcinoma.
Economic analysis
Economic evaluation of the Pap test and HPV test strategies with equal costs per woman screened and per lesion found. Applied to a 100,000 women cohort
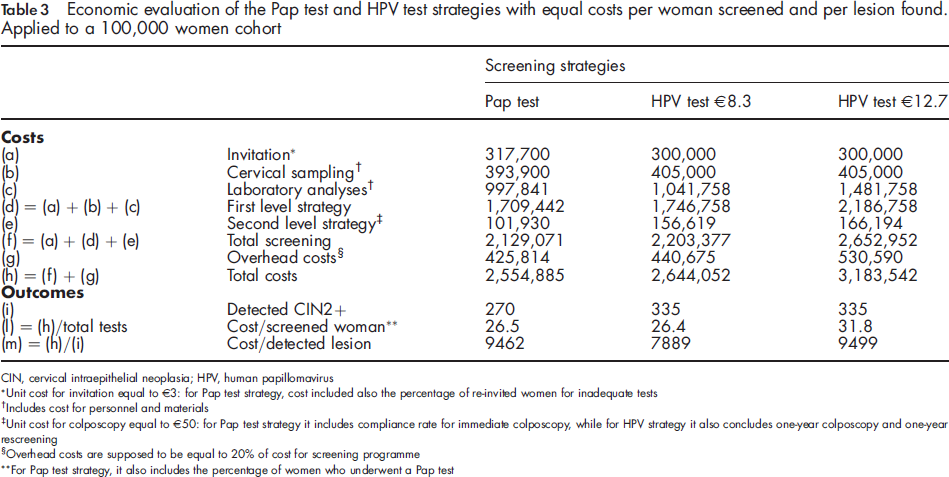
Unit cost for invitation equal to €3: for Pap test strategy, cost included also the percentage of re-invited women for inadequate tests
Includes cost for personnel and materials
Unit cost for colposcopy equal to €50: for Pap test strategy it includes compliance rate for immediate colposcopy, while for HPV strategy it also concludes one-year colposcopy and one-year rescreening
Overhead costs are supposed to be equal to 20% of cost for screening programme
For Pap test strategy, it also includes the percentage of women who underwent a Pap test
Unit costs of the Pap test were estimated in a previous study conducted by the Agency of Public Health conducted in three ASL. We identified a unit value of €15 (personnel and materials included) for cervical sampling and laboratory analyses.
To calculate the unit value for the HPV test, we had to run the analyses with different scenarios for the price kit (prices range from €9 to €13.5 including VAT). The reason behind this choice was due to the fact that the kit price is the main item cost, with the greatest effect on total costs.
Data from the pilot study highlighted that the referral rate for colposcopy was 39.6%; we considered the unit cost for second-level strategies as the average of a negative colposcopy without punch biopsy and a positive colposcopy with punch biopsy and consequent histological test. This parameter ranged from a minimum of €11 to a maximum of €100, with a baseline of €50. Finally, the compliance rate for repeat cytology was 62%, 24 for colposcopy was 92% and for a one-year re-screening was 63%. The cytology results and cytology DRs were derived from the nationwide screening average. 24 The relative DR was derived from the NTCC trial,3,9 but was adjusted for the observed compliance to follow-up and colposcopy.
The equivalent point in terms of cost per lesion detected (€9499) was found with a unit cost per kit equal to €12.7 for HPV. This equivalent point showed that at this price level, costs per lesion detected were basically identical in screening strategies even though HPV tests presented a 20% higher per person cost; in fact, the equivalent point for screening was €8.3.
Under the hypothesis of delivering 100,000 tests and at the same cost per lesion found, the total cost of a conventional screening programme was €2,544,856, while the total cost with the new protocol amounted to €3,183,542. Those features included both levels of analyses (invitation, cervical sampling, laboratory analyses, referrals to colposcopy and possible biopsy) and the overhead costs. The second-level analysis costs were higher in HPV screening (56-66% more) because of its nature: given its higher sensitivity and lower specificity, HPV testing detected a greater number of cancerous lesions but required more triage and colposcopy tests (335 lesions detected with HPV versus 270 with Pap tests).
In the sensitivity analysis the most relevant parameters for the cost per lesion found are the compliance to follow-up and colposcopy. On the other hand, the cost per woman screened showed small variations with all parameters, except Pap test cost and unsatisfactory rate.
Discussion
In 2007, the results of two trials showed a substantial earlier diagnosis with the HPV test, than the Pap test, and no or minimal over-diagnosis.10,14,16,25 This evidence suggests starting with pilot studies to verify if the new screening protocol is feasible in non-research settings, is acceptable by women and health carers and is cost-effective.
Technical feasibility
The implementation of our pilot screening with HPV as the primary screening test did not identify any technical or organizational problems.
The increase in workload was consistent only for the molecular biology laboratory, while the pathology unit managed the Pap tests without any substantial organizational impact. The burden for colposcopy was actually the same as in the Pap test screening.
Screening and health outcomes
We cannot measure the effect HPV testing had on the compliance to screening invitation, but we can say that compliance was higher in our pilot study than in the surrounding areas using conventional Pap testing.
The DR of CIN2+ at baseline was similar to the rate in the surrounding areas, while the total DR, including the cases found at one-year follow-up, was slightly higher. Nevertheless, the DR in Italy is one of the lowest in Europe 26 due to some of the following factors: relatively low HPV prevalence and strong diagnostic pressure by opportunistic screening also on women who participate in organized screening programmes.
Actually one of the major uncertainties about the effectiveness of this screening algorithm is how women will respond to the protocol and, particularly, how to monitor compliance to the one-year interval for HPV-positive and cytology-negative women. These women present quite a high risk and it is crucial to follow them in order to realize the advantages of HPV strategy.3,9,10,14 On the other hand, it is also important that a conservative approach be taken, since the risk of over-treatment, particularly in younger women, could be very high if they received immediate colposcopy. 11 Therefore, it is important that they trust the screening recommendations without obtaining a colposcopy outside the programme. Our results clearly highlight how it is difficult to obtain high compliance in this group: only one-half of them returned, and we can infer that six more CIN2+ lesions were present in the women who did not attend the follow-up HPV test, or were not compliant to follow-up colposcopy.
Furthermore, our results show how the way this group of women is contacted may influence compliance: direct phone contact was much more effective than the mail. Given the relatively high risk of high-grade lesions in this group, and also the risk of over-treatment if referred immediately to colposcopy, particularly for younger women, effective communication is important that reassures the women enough to not obtain an extra colposcopy outside the screening programme and also convinces them of the need for a subsequent test one year later.
Cost analysis
Our report suggests that the HPV test could be a reasonable option as a primary screening test at a price less than €12.7 per kit if we use the cost per lesion found in the first round, and less than €8.3 if we use the cost per women screened to compare the two strategies.
Actually, the main determinant of the total cost of mass screening using HPV as the primary test is the price of the kit, while in a screening programme using the Pap test the main cost determinant is still the person/time for slide interpretation.
The situation presented is directly applicable only if we have extremely elastic labour conditions, i.e. we should be able to convert the specialists that were formerly committed to slide interpretation to other functions useful to the health service. This is an uncommon condition, given the high specialization of cytologist involved in this task. In our case study, the elasticity of these resources is absolute, given that the slide interpretation was externalized in out-sourcing.
Univariate sensitivity analysis: cost of the HPV test (in €) at which it is equivalent to conventional Pap test per screened woman and lesion found
More complete cost-effectiveness analyses have been performed to compare Pap tests with HPV testing: 27 33 all of these studies produced an incremental cost-effectiveness ratio, comparing utilities and costs of the two strategies. Our results can be compared with this kind of study only to estimate costs, but not for the utilities, since our analysis assumes the same health utilities for all the strategies. The unit cost estimates used in this study are in line with the costs estimated by Kim and colleagues 32 for Italy in 2005, except for the colposcopy for which we used a lower value and the Kim estimate is approximately the upper value used in the sensitivity analysis.
Limits
Our approach does not permit a cost-effectiveness analysis, and actually assumes that the utilities are the same for each strategy. In the short term, given the very low incidence of interval cancers identified in cervical cancer screening34,35 in Italy, this assumption is reasonable. Our unit to measure benefits is the lesion found or the woman screened. Both the cost per lesion found and per screened woman have limits in their applicability to measure real health gain. 36 38 The cost per woman screened does not consider that the HPV provides longer protection and it is valid only if we do not use a longer screening interval after a negative HPV test, and even in this case it does not consider that in the following round the positivity rate will be lower.10,14,16 On the other hand, the cost per lesion found is more linked to the health outcome, but the advantage of a higher DR at base line may be transformed into economical gain only if an optimal screening interval is adopted; in fact, if we still screen at three-year intervals, at the following round the DR will be dramatically lower and the cost per lesion found will go up.10,14,16
Our approach was to give a clear indicator of at what price HPV becomes convenient with respect to the Pap test: the threshold price of the kit.
The uncontrolled design of this pilot phase did not allow us to clarify several doubts (relative adhesion to screening inviting letter for HPV testing versus conventional Pap test and relative DR), but had some other advantages, in particular it allowed us to evaluate the costs and the feasibility of implementation in a real situation and not in the unrealistic condition of having two organizational models operating in parallel in the same clinic.
Finally, we tried to present timely data; to do that we had a short follow-up to collect all the colposcopies and histological results. It is possible that some of the data slightly underestimated HPV-positive cytology-negative women at one-year follow-up, in particular the compliance and the DRs may increase in the following few months.
Conclusions
The HPV test was accepted in the general population at least as well as the Pap test as a screening tool. The algorithm adopted did not cause any relevant problems in managing positive women, while compliance to one-year follow-up was the main barrier to the effectiveness of the adopted protocol.
Based on this pilot study, the HPV test will reach the same cost per screened woman at a price of about €8.3, while the cost per lesion found would be equal to conventional Pap smear at a price of €12.7.
