Abstract
Objective
To document patient decisions after being informed of a first trimester sequential screen Down's syndrome risk between 1/51 and 1/270.
Setting
A database analysis of sequential screen results for patients seen in the Philadelphia, PA (USA) area between January 2006 and March 2008 was examined.
Methods
All patients with first trimester sequential screen Down's syndrome risks in the 1/51–1/270 range were identified. Patient decisions regarding invasive testing (prior to completing the second trimester stage of the sequential screen), completion of the second trimester blood draw or no additional testing were tabulated.
Results
A total of 10,850 patients underwent first trimester sequential screening during this interval. Five hundred and fifty-seven patients (5.1%) met the study inclusion criteria and had risks between 1/51 and 1/270. Ninety-three percent of these patients completed the sequential screening process before making any decisions regarding invasive testing. Four percent did not elect an invasive prenatal diagnosis procedure, but also did not complete the second trimester sequential screening blood draw and only 3.2% elected an invasive procedure based on their first trimester risk without completing the second trimester blood draw. Five women (0.9%) with low risks after the second stage screen chose to have an amniocentesis.
Conclusion
The vast majority (97%) of patients in the moderately increased Down's syndrome risk range (1/51–1/270) following first trimester sequential screening did not pursue an invasive procedure based on their first trimester sequential screen risk. Using a ≥ 1/50 risk cut-off in the first trimester is an effective screening policy for sequential screening.
Introduction
The Genzyme Genetics first trimester sequential screening protocol utilizes a Down's syndrome risk cut-off of ≥ 1/50 as a screen-positive result. This is calculated to detect 70% of Down's syndrome pregnancies at a 1.2% screen-positive rate in the first trimester. Following completion of the second trimester sequential screen, 90% of Down's syndrome fetuses will be detected utilizing a risk cut-off of ≥ 1/270. The second trimester false-positive rate is calculated at 2.5%, with an overall sequential screen false-positive rate of 3.7%.
We sought to determine if patients with a moderately increased first trimester risk of Down's syndrome (1/51–1/270 risk) elected to pursue an invasive prenatal diagnosis procedure instead of completing the second trimester stage of the sequential screening test. Our concern was that patients may intervene based on moderately increased risks, especially because a Down's syndrome risk cut-off of 1/270 has traditionally been utilized as indicative of an increased risk of Down's syndrome. If patients with these moderately increased risks did intervene after the first trimester stage of the sequential screen, it would result in an increased rate of invasive procedures. These patients who failed to complete the second trimester stage of the sequential screen would then be deprived of the opportunity to find out if the addition of second trimester analytes to the screening process would have placed them in a low-risk range (< 1/270 risk).
Methods
A query of the Genzyme Genetics sequential screening database was performed on patients in the Philadelphia area seen between January 2006 and March 2008. All first trimester nuchal translucency ultrasound measurements were obtained by sonographers and maternal fetal medicine specialists certified in nuchal translucency measurement by the Fetal Medicine Foundation and/or the Society for Maternal Fetal Medicine.
Once the results of the first trimester stage of the sequential screen were available, the patient was contacted directly by phone and informed of their numerical odds of carrying a fetus with Down's syndrome. A patient was informed they were screen-positive if their Down's syndrome risk was ≥ 1/50. The risk reported pertained to the risk of carrying a Down's syndrome fetus in the first trimester. These patients were also informed that genetic counselling was recommended to discuss the option of an invasive prenatal diagnostic procedure given their screen-positive result. If they were found to have a Down's syndrome risk <1/50, they were advised to return to complete the second trimester stage of the sequential screen. Our genetic counsellors who contacted patients to inform them of their results were specifically instructed to be informative, but not to provide genetic counselling over the phone. The genetic counsellors informed patients of their individual numerical odds of Down's syndrome (i.e. 1 in 300, etc.) and whether this placed them in as ere en-positive (high-risk) or screen-negative (low-risk) category according to our sequential screening algorithm. For patients who had numerous questions or unresolved issues with their screening results, an appointment was offered for them to obtain in-person genetic counselling.
All patients with first trimester sequential screen results in the 1/51–1/270 range were identified. Patient decisions regarding invasive testing (prior to completing the second stage of the sequential screen), completion of the second blood draw or no additional testing were tabulated. Patients who had a spontaneous loss between the first and second blood draws, a diagnostic procedure because of a family history indication, an abnormal ultrasound or a positive first trimester trisomy 18 screen were excluded from the analysis.
Results
A total of 10,850 patients underwent first trimester sequential screen testing during this interval, and 573 patients were identified as having Down's syndrome risks between 1/51 and 1/270. However, 16 patients with Down's syndrome risks between 1/51 and 1/270 were excluded due to either a family history indication for an invasive procedure, a spontaneous loss between the first and second blood draws, an abnormal first trimester ultrasound or a positive trisomy 18 screening test. Therefore, 557 patients (5.1%) met our criteria for inclusion into our study. Five-hundred and fifteen patients (93%) completed the second trimester blood draw, five patients pursued a chorionic villus sampling (CVS) procedure without completing the second trimester stage of the sequential screen, 13 patients elected an amniocentesis without having the second trimester blood draw and 24 patients had no further testing (Figure 1). Five women (0.9%) with low risks after completing both stages of the sequential screen chose to have an amniocentesis.
Decisions made by patients with first trimester sequential screen risks of Down's syndrome between 1/51 and 1/270. CVS, chorionic villus sampling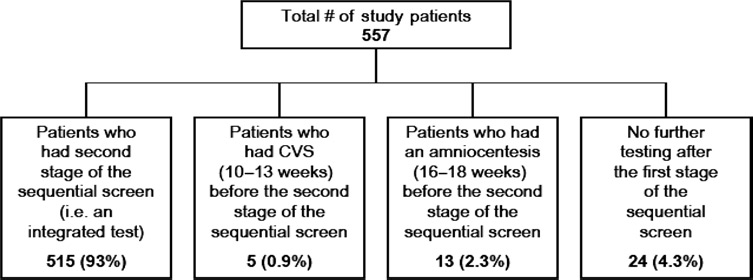
Among our study sample of 557 patients with moderately increased Down's syndrome risks, the range of Down's syndrome risks were between 1/51 and 1/270 and the mean risk was 1/181. Thirty-nine of 515 women (7.6%) who had moderately increased risks of Down's syndrome after the first stage of the sequential screen, who underwent the second blood draw to complete the sequential screen algorithm, elected an amniocentesis. Of these 39 women, 28 had an increased risk of Down's syndrome after completion of both stages of the sequential screen, four had ultrasound-detected fetal abnormalities in the second trimester (Dandy-Walker malformation, echogenic bowel, cleft lip and palate, echogenic intracardiac focus), one had an amniocentesis because of an increased risk of an open neural tube defect and another underwent invasive testing because of an increased risk of trisomy 18. Five women elected an amniocentesis despite a negative completed sequential screen. The first and second trimester Down's syndrome risks for these five women went from 1/220 to 1/10,000; 1/245 to 1/10,000; 1/225 to 1/1000; 1/215 to 1/580; and 1/85 to 1/800, respectively. Eighteen women with moderately increased Down's syndrome risks elected to have a diagnostic procedure prior to a second trimester blood draw. The range of risks for these women was between 1/55 and 1/270, with a mean risk of 1/126. Twenty-four women (4% of our sample of 557 patients) neither elected to have a diagnostic test after their first trimester results were reported nor completed the second stage of the sequential screen. Their Down's syndrome risks ranged from 1/105 to 1/270, with a mean risk of 1/196.
Discussion
In our programme, 97% of patients with a first trimester sequential screen risk of Down's syndrome between 1/51 and 1/270 did not elect an invasive procedure based on their first trimester Down's syndrome risk. Ninety-three percent completed the sequential screening process before making any decisions regarding invasive testing. Four percent did not complete the second trimester stage of the sequential screen, but also did not elect a CVS or amniocentesis. Only 18 patients (3.2%) with a Down's syndrome risk between 1/51 and 1/270 decided to pursue a CVS or amniocentesis without completing the second trimester blood draw. The mean risk of Down's syndrome in this group of 18 patients who intervened prior to completing the second stage of the sequential screen was 1/126. This mean risk was higher than the mean risk for the group that completed the sequential screen (1/182). It is understandable that there was a higher mean risk among these 18 patients who elected a CVS or amniocentesis prior to completing the second stage of the sequential screen compared with those who did not intervene after their first trimester results were reported. There were 13 patients who elected an amniocentesis after being informed their first trimester Down's syndrome risk was between 1/51 and 1/270. Two women were informed of their moderately increased risk before 12 weeks, six women were informed between 12 and 13 weeks and two women were informed between 13 and 13.5 weeks. Three patients did not receive their first trimester sequential screen results until past 14 weeks (too late for a CVS). Therefore, 10 of these women could have had a CVS (an earlier test), but instead elected to wait for an amniocentesis. Reasons that women may have preferred an amniocentesis instead of a CVS include the following: (1) some women prefer to avoid a CVS because of the 1% risk of confined placental mosaicism (which does not occur with an amniocentesis), (2) an amniocentesis provides AFP information to determine neural tube defect risk, while a CVS does not and (3) some patients may prefer their obstetrician to care for them exclusively. Most obstetricians can perform an amniocentesis without difficulty, whereas a CVS requires referral to a specialist. Therefore, some patients may wait for an amniocentesis because they want the comfort of being cared for by a physician they trust.
Most importantly, 539 of 557 (97%) patients did not intervene despite being informed of a moderately increased first trimester risk of Down's syndrome. Instead, they followed the recommended algorithm and completed the sequential screening process. From a screening perspective, it is important that so few patients elected an invasive prenatal diagnosis procedure based on their moderately increased first trimester Down's syndrome risk. Based on parameters from the Serum Urine and Ultrasound Study (SURUSS) and the maternal age distribution in England and Wales (2002–2004) with first trimester markers measured at 12 completed weeks, the odds of being affected in the group with risks of 1/51–1/270 is approximately 1/115. 2 These odds are inferior to most Down's syndrome screening protocols. Therefore, it is advantageous that only 3% of patients with risks in this range elected an invasive procedure instead of following the recommended protocol of completing the sequential screen test. For these women who had moderately increased risks but were designated as screen-negative, electing an invasive procedure before completing the second stage of the sequential screen prevents them from benefiting from data derived from a complete analysis utilizing both first and second trimester screening parameters. If these 18 patients had completed the full sequential screen, or if they had undergone an integrated screen, many would have been informed that their risk of Down's syndrome was much lower than their first trimester results. Many of these women could have then decided to avoid the risks associated with a CVS or an amniocentesis.
Only five patients (0.9%) with moderately increased first trimester results underwent an amniocentesis despite being informed that their Down's syndrome risk was low after completing both stages of the sequential screen. These five women must have had a residual concern that was not allayed even after completing both stages of the sequential screen. In these five patients, a definitive diagnostic test was requested instead of relying on a reassuring (low risk) screening test. It is uncertain if these five women would have avoided an invasive diagnostic procedure if they had only been provided with a single, screen-negative, second trimester risk assessment as would have occurred if they had undergone an integrated screen. Importantly, 99% of patients with a moderately increased first trimester Down's syndrome risk are reassured and do not elect an amniocentesis if their completed sequential screen reveals a low risk result.
The observation that patients with first trimester sequential screen risks between 1/51 and 1/270 generally do not intervene based on these moderately increased risks has important implications regarding the false-positive rate for sequential screening in clinical practice.
Ninety-seven percent of patients follow the recommended protocol based on their designation as being either screen-positive or -negative in the first trimester. Therefore, the false-positive rate, and hence the rate of invasive prenatal diagnosis procedures for sequential screening, is not significantly affected by patients with a moderately increased (1/51–1/270) risk of Down's syndrome. Of additional importance is the observation that providing first trimester risk information did not significantly affect overall screening efficacy.
Advocates of integrated screening on all women have argued that Down's syndrome risk information should be held until all first and second trimester data have been evaluated. 3 By reporting only a single Down's syndrome risk, it has been reported that integrated screening is a simpler test. Other investigators have expressed concern that divulging first trimester risk information may lead to unnecessary invasive procedures without the benefit of completing the screening process. 4 This concern does not appear to be justified based on our observations.
Previous studies have revealed that when results are reported as dichotomous events using terms such as Tow risk’ or ‘high risk’ based on specified cut-off levels, patients may be reluctant to ask questions. They will often base their decisions on how they have been categorized and generally follow the expected response to such a categorization, the so-called compliant behaviour. 5 Some investigators have concluded that verbal expressions of risk, while not as precise as numerical risk assignment, may help decision-makers present a problem so that a choice can be more easily made. 6 However, they also acknowledge that if statistical information is available, this is the kind of information recipients prefer. In our experience, patients’ decisions regarding invasive testing are not necessarily determined based on a patient's numerical risks or odds of Down's syndrome, but predominantly on a verbal classification of risk. This may be related to the observation that reporting results in terms of categories such as ‘high risk’ or ‘low risk’ may be viewed as facilitating directive counselling.5,6 In our counselling approach, patients were informed both of their odds of Down's syndrome (i.e. 1 in 100, 1 in 500, etc.) and whether this placed them in a high- or low-risk category based on our screening algorithm. Patients had the option to proceed to invasive testing (CVS or amniocentesis) regardless of their Down's syndrome risk based on patient autonomy as stipulated by the American College of Obstetrics and Gynecology. 7 However, only a small percentage (3%) of patients with a risk between 1/51 and 1/270 opted for invasive testing. It therefore appears that a patient's categorization of risk outweighs their numerical risk assessment in deciding upon intervention in the form of invasive prenatal diagnosis. From a screening performance standpoint, it is preferable to await a full risk assessment than basing decisions on the first trimester results only. A complete assessment can only be accomplished after second trimester analytes are added to the risk analysis. Most patients with first trimester risks of Down's syndrome between 1/51 and 1/270 will be placed in a low-risk category (< 1/270) after completion of the second stage of the sequential screen. Therefore, if these patients had decided to have a CVS after their first trimester results, many would have undergone invasive testing unnecessarily as they would have been identified as being at low risk following completion of the second trimester stage of the sequential screen. Intervention based on moderately increased first trimester risks would result in an increase in iatrogenic pregnancy loss and a decrease in cost-effectiveness of the screening programme.
It has previously been documented that the introduction of first trimester screening results in a decrease in invasive testing for patients of advanced maternal age. 8 Currently, all patients, regardless of maternal age, can be counselled that their decision to proceed to CVS or amniocentesis should be based on their risk of Down's syndrome, not on their age alone. Maternal age is incorporated into the screening analysis and should be viewed as just one component of risk along with the other parameters such as hCG, PAPP-A, AFP, uE3, inhibin-A and nuchal translucency.
The Genzyme Genetics sequential screening programme was designed to identify those patients with an extremely high risk of Down's syndrome in the first trimester (≥ 1/50 risk) as screen-positive. Using data from SURUSS, the utilization of > 1/50 as a risk cut-off would be anticipated to lead to a 70% Down's syndrome detection rate at a 1.2% screen-positive rate. 2 Following completion of the second trimester sequential screen, 90% of Down's syndrome fetuses will be detected utilizing a risk cut-off of ≥1/270.
It is apparent that patients do not intervene based primarily on their numerical odds of Down's syndrome, but instead on their categorization of being either screen-positive or screen-negative. In addition, it was rare for patients with moderately increased risks to schedule a genetic counselling appointment. Almost all waited until their second trimester results became available. This is beneficial to patient care, Down's syndrome screening performance and public health. Our data confirm that the sequential screening algorithm, using a first trimester risk cut-off of ≥ 1/50, is an effective screening paradigm.
Declaration
All authors are employees of Genzyme Genetics.
