Abstract
Anal squamous intraepithelial lesions (ASILs) are the precursors to anal cancer. Human papillomavirus infection has a direct link to ASIL formation and is responsible for up to 80% of anal cancers. But while much importance has been focused on targeting cancer precursors in the cervix, relatively little concern has been afforded to the anal canal. With the advent of cervical Pap smear screening in various regions, the incidence of cervical cancer has declined. However, marked similarities in the biological and pathological profiles of cervical cancer and anal cancer mean that anal cancer should be preventable in the same way – by curbing the progression of ASIL to cancer. This article explores the literature on ASILs and the growing problem of anal cancer in the community, along with the literature surrounding the current progress towards implementing a screening programme for ASIL in the future.
Introduction
Methods
We searched the National Library of Medicine PubMed for English-language literature publications and their relevant journal article references on anal cancer screening. The search terms we used were ‘anal cancer’, ‘anal intraepithelial neoplasm’, ‘anal squamous intraepithelial lesions’, ‘screening’, ‘human papillomavirus’ and ‘HIV’. The abstracts of the related articles were analysed and only those publications with relevance to HPV, HIV, anal cancer and screening were selected. A total of 32 articles were appraised. The information for this review was also obtained from other systematic reviews. In addition, other recent articles, editorials and discussions of relevance were also appraised and included.
Results and Discussion
HPV in cervical and anal cancer
HPV is the most common sexually transmitted infection in the USA. 3 Some individuals with HPV infection may go on to develop clinically obvious condyloma, or warts. However, latent and subclinical forms of HPV also exist, and the virus may only be clinically detectable on examination of abnormal epithelium with high-resolution anoscopy. 1 Regardless of condyloma formation, HPV infection is a direct link in the development of squamous dysplasia in the anal canal. 4 Up to 92% of anal cancers are associated with HPV.5–7 Following a multinational approach to screen for CIN with the Pap cytological smear test, the incidence of cervical cancer has plummeted in well-adopted screening areas. 8
Cervical cancer and anal cancer are similar in many ways. From a biological standpoint, both areas have a higher tendency to dysplasia as a result of HPV infection. Embryologically, both structures are also derived from the cloacal membrane, where ectodermal and endodermal tissues meet to form a transformation zone. This is the area that undergoes dysplastic change as a direct result of HPV infection. The same HPV subtypes infecting the cervical epithelium also infect the anal transformation zone. Of 100 recognized HPV subtypes, over 80 have a completed genome at this date; 35 are specific for the cervical and anal epithelium, each subtype carrying a variable capacity to cause dysplastic change. The high-grade oncogenic subtypes 16 and 18 are more likely to cause dysplasia and are identified in about two-thirds of all invasive cervical cancer cases. 4 While data on the relative frequencies of individual HPV subtypes causing ASIL and anal cancer are unavailable, a recent systematic review showed that 72% of invasive anal cancer was associated with the oncogenic subtypes 16 and 18. The crude HPV prevalence in anal cancer was 71% in that study, with the prevalence of high-grade ASILs (HSILs) and low-grade ASILs (LSILs) at 91% and 88%, respectively. 9 A further list of HPV subtypes and their associated effects on the anogenital epithelium is summarized in Tables 1 and 2. It can be seen from these tables that certain subtypes are considered ‘high risk’ for oncogenic activity.
HPV subtypes and their associated effects on the anogenital epithelium

HPV, human papillomavirus
Recognized HPV subtypes classified according to malignant potential

HPV, human papillomavirus
The anal canal, like the cervix, also undergoes stages of dysplasia before becoming cancer proper. 10 Initially, these stages were classed as either low-grade, intermediate or high-grade AINs, but in 2002 Solomon et al. 11 revised the Bethesda system of cytological classification in the anal canal. Anal dysplastic lesions are now reclassified into LSILs or HSILs. Two further categories are recognized in this system – atypical squamous epithelial cell histologies of undetermined significance (ASCUS) and those types of ASIL in which HSIL could not be excluded (see Table 3).
Bethesda classification of anal canal cytology (from Solomon et al. 11 )
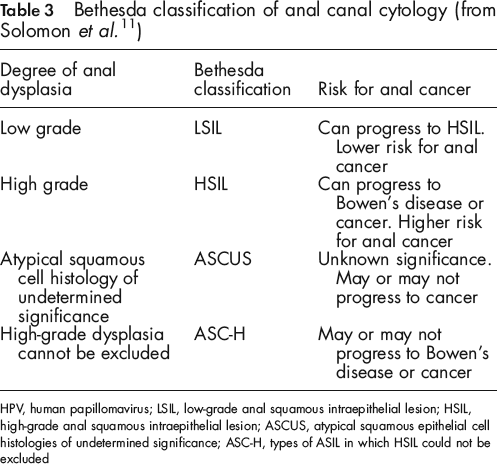
HPV, human papillomavirus; LSIL, low-grade anal squamous intraepithelial lesion; HSIL, high-grade anal squamous intraepithelial lesion; ASCUS, atypical squamous epithelial cell histologies of undetermined significance; ASC-H, types of ASIL in which HSIL could not be excluded
Prior to cervical cancer screening, an approximate 40/100,000 women in the US were found to have cervical cancer. After screening, this number fell to 8/100,000 – underlining the benefit of Pap screening. 2 For anal cancer, no large-scale screening programmes have been adopted and the Scandinavian Registries Base estimate a rise of about 2% in incidence per year in the general population. In fact, over the last two to three decades, anal cancer has already increased by 96% in men and 39% in women.12–14
ASIL, anal cancer and HIV
HIV-positive people and men who have sex with men (MSM) are two particular groups of people who are at particular risk of developing ASIL. An MSM who is also HIV positive has a very high risk of developing ASIL, and therefore eventual anal cancer. 15 This is likely to be caused by a combination of the immune-depleting function of HIV and the increased risk of ASIL and anal cancer associated with anoreceptive intercourse. 16 Previous studies with HIV-negative populations had identified anoreceptive intercourse as an important risk factor.2,17 Anoreceptive intercourse allows the anal mucosa to come in direct contact with an HPV-infecting source, in particular the transition zone, where dysplasia is more likely to occur. In the US, the incidence of anal cancer in HIV-negative MSM was 35/100,000 (data taken prior to the AIDs era). 2 After the AIDs era, the incidence of anal cancer escalated to about 70/100,000 – a number at least double that of the pre-AIDs era. 18 This can be compared with the general population where only 0.8/100,000 people have anal cancer. 12
One of the largest studies performed on HIV and ASIL was the EXPLORE study. 19 Of 1262 HIV-negative MSM investigated, ASIL was detected in about 20% of cases. Another study of HIV-positive MSM by Goedert et al. 18 also found a two-fold increase in the relative risk of anal cancer compared with HIV-negative MSM. In a further study of progression rates of ASIL, 346 HIV-positive MSM and 262 HIV-negative MSM were followed up after two years: 62% of HIV-positive MSM with LSIL progressed to develop HSIL (versus 36% of HIV-negative MSM). 20
Other studies have shown a significant risk of ASIL in MSM. Palefsky et al. 15 investigated 357 HIV-positive MSM and observed that as many as 80% had anal dysplasia while a vast 95% of cases had a concurrent HPV infection (compared with 61% of HIV-negative MSM). The oncogenic subtype 16 was found to be the commonest subtype and multiple subtype infection was also seen in 73% of cases (versus 23% in HIV-negative MSM). One study observed the risk of ASIL in HIV-positive men who did not have sex with men. Of 50 HIV-positive heterosexual men not partaking in anoreceptive intercourse, 46% were found to have anal HPV infection, 16% had LSIL and 18% had HSIL. This was compared with 67 HIV-positive MSM in the same study, where 85% had anal HPV infections, 49% had LSIL and 18% had HSIL. 21
Although fewer studies exist describing ASLL in women, the incidence of HPV-related anal cancer is known to be higher in women than in men.22,23 A study by Williams et al. 24 showed that out of 52 HIV-positive women investigated, 77% turned out to have a concurrent HPV infection. Holly et al. 25 studied 251 women and found that 26% of HIV-positive women had abnormal anal cytology compared with 8% of HIV-negative women; furthermore, 6% of HIV-positive women in this study had HSIL on histology compared with only 2% of HIV-negative women. From these studies, HIV infection appears to confer an approximate three-fold increased risk of ASLL in women (see Table 4).
Summary of the increased risk of abnormal anal cytology and HPV infection in association with HIV infection
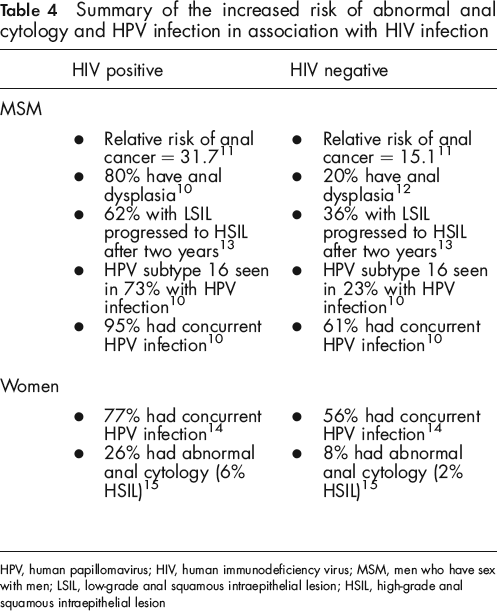
HPV, human papillomavirus; HIV, human immunodeficiency virus; MSM, men who have sex with men; LSIL, low-grade anal squamous intraepithelial lesion; HSIL, high-grade anal squamous intraepithelial lesion
Does highly active antiretroviral therapy have an effect on ASIL in HIV-positive individuals?
Rising numbers of people with anal cancer may only have come to light with the development of antiretroviral drugs. Highly active antiretroviral therapy (HAART) now enables HIV-positive individuals to live a near-normal quantity of life, but this increased survival may have allowed the anal transformation zone sufficient time to develop from ASIL into cancer. 26
While HAART therapy is promising against HIV, it appears to have a smaller effect in treating HPV infections. High rates of ASIL were found to be maintained regardless of CD4+ counts.27–29 other cohort studies found no statistical differences between anal cancer in the pre-HAART and HAART eras.30–33 Palefsky et al. 34 investigated the outcomes of ASIL after HAART therapy and found no difference in incidence or rate of progression compared with no HAART therapy, but HIV-infected individuals appeared to be less able to clear their HPV infections than HLV-negative people. A later observation by the same authors, however, showed that after six months of therapy, 27% of ASILs had regressed and 4% of cases did not show any dysplasia; 57% of cases, however, showed no significant change.
Screening for ASIL
The prognosis of anal cancer is strongly related to stage of disease at diagnosis. 35 The report by Johnson et al. 12 on anal cancer incidence and survival from 1973 to 2000 revealed that five-year survival rates were highest for localized disease (78%) compared with regional (56%) or distant disease (18%). Therefore, many practitioners have adopted screening practices for particular at-risk groups, such as HIV-positive MSM. There is reason to suggest that anal Pap screening would be a feasible and cost-effective approach in preventing anal cancer as well as cancer of the cervix. The target population for anal screening is definable from the above studies: these would include HIV-positive MSM, any HIV-positive individual, all MSM or any individual engaging in high-risk sexual activities 36 (see Table 5).
Population that would benefit from screening for anal dysplasia

HIV, human immunodeficiency virus; MSM, men who have sex with men; SIL, squamous intraepithelial lesion
Screening techniques include the following clinical investigations: thorough clinical review of historical risk factors, digital rectal examination, anal Pap smear test and/or high-resolution anoscopy.1,37 Like cervical Pap screening, the purpose of the anal Pap smear would not be to determine staging, but to simply establish a present-absent response. The current recommendation is for Pap screening to be carried out every three years in HLV-negative individuals, and yearly in HIV-positive individuals. 37 A recommended technique involves positioning the patient in the lateral or dorsal lithotomy position with both buttocks retracted to expose the anal canal. A moist Dacron swab is inserted as far as possible into the anal canal to about 5–6 cm (wooden cotton-tipped sticks should not be used due to the risks of splintering). As the swab enters the anal canal, the swab may be felt to progress past the internal anal sphincter as it moves to the transition zone. Slow and gentle unidirectional turns of the swab against the wall of the anal canal allow capture of cells in the anal canal and the swab is returned to the vial. In the vial, the swab should be rotated vigorously for about 10 turns with pressure against the vial wall, followed by a swirl of the swab in the solution to ensure an optimal release of cells. 36 If the anal Pap smear is subsequently positive, then referral for proper visualization procedures and biopsy would be indicated (see Figure 1).
Screening algorithm for anal squamous intraepithelial lesion (ASIL) as recommended by Matthews
37
. LSIL = low-grade ASILs; HSIL = high-grade ASILs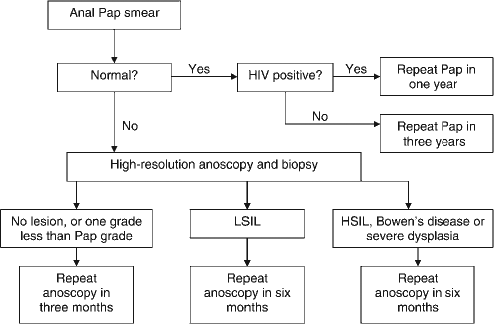
Is screening cost-effective?
In the US, a cost-effective screening programme is determined to be one that has a treatment cost of under $30,000–50,000 per year of life saved (PYLS). 37 Taking cervical cancer as the example, Pap smears every three years for HIV-negative women incur a treatment cost of approximately $180,000 PYLS (assuming the natural history of neoplasia in women with CD4 cell counts >500 cell/μL to have the same risk for progression and regression of cervical disease as non-FHV-infected women with similar risk factors, and assuming that the health-related quality of life would then be greater in HIV infection or local cancer). In HIV-positive women with yearly cervical Pap smears, the treatment cost would be about $13,000 PYLS. 38 Estimates for Pap screening in anal cancer showed that the costs of screening for ASIL in HIV-positive men with annual testing amounted to $11,000 PYLS. A three-yearly testing regimen in HIV-negative men was estimated to be about $7,800 PYLS, a cost lower than that of cervical cancer screening.39,40
One systematic review observed that Pap screening in CIN II or CIN III conferred reliability coefficients of around 0.67–0.76. 37 When applied to the anal canal, this sensitivity is reduced, owing to the fact that anal transition zones are not as clearly identifiable as the cervix. Anal Pap smears would thus necessitate blind swabs of the anal canal and anal transition zone. 41 Matthews and colleagues 42 determined a kappa statistic agreement of 0.3 between single repeat tests for HSIL/LSIL against ASCUS/normal cytology. Six further studies then established sensitivities ranging between 69% and 93% and specificities between 32% and 59%. Palefsky et al. 43 demonstrated that positive predictive values could be increased from 38% to 78% after two years of screening, with a negative predictive value increased from 46% to 79%.
The agreement between concurrent cytology examination with biopsy was also calculated at 74.7% with a weighed statistic of 0.35, a figure that is similar to the absolute and weighted agreement of cervical cytological examination and biopsy (64–91% and 0.18–0.65%, respectively).44–46 Goldie et al. 39 also developed a state transition Markov model incorporating several parameters – lifetime costs, life-expectancy, quality of life-expectancy – for several screening strategies in HIV-positive MSM. They obtained an incremental cost-effectiveness ratio of $16,000 per quality-of-life year saved in Pap screening versus no screening, a figure comparing well with other already well-adopted screening procedures.
Obstacles to screening
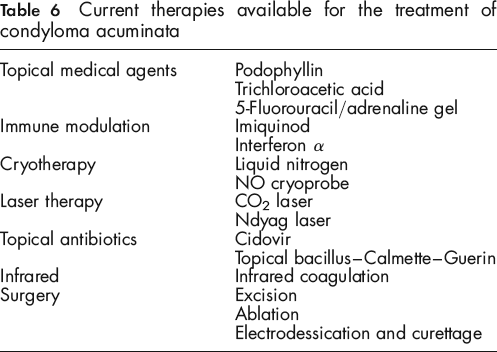
The Achilles’ heel in developing a screening protocol for anal cancer currently lies in the treatment modalities used. Most techniques are fraught with adverse effects, high recurrence rates with little or no published evidence of success. Other medical treatment strategies including the use of imiquinod or podophyllin are often poorly tolerated due to local side-effects (see Table 6). Screening for anal dysplasia would have little clinical value if evidence-based efficacious therapeutic options were unavailable. There is also a lack of clinicians trained in anal cytology sampling and high-resolution anoscopy. Many shy from the uncomfortable discussion with their patients about anoreceptive intercourse. For patients, the fear of anoscopy and biopsy may also further hinder screening. Current recurrence rates are estimated to be as high as 50–85% depending on treatment modality. 37 At present, the treatment standard for anal cancer is chemoradiotherapy, which yields a five-year survival rate of 70–80% in the general population.47,48
Current therapies available for the treatment of condyloma acuminata
Unlike cervical dysplasia, where a large loop excision of the transformation zone has 95% one-year cure rates in HIV-positive women, no evidence for such efficacy with simple procedural intervention is provided for the anal canal. 49 The anal canal is also structurally more difficult to negotiate than the cervix and excision of the entire transformation zone would not be possible. There are no documented benefits in favour of anal Pap smear screening to date and large population-based randomized controlled trials are needed to examine the survival benefit in anal Pap smear versus no screening test. Until these points are addressed, there will likely be continued ramifications affecting the initiation of a screening programme.
Role of vaccines?
On a positive note, new vaccines are yielding promising results. Klencke et al. 50 presented their ZYC101 plasmid-derived vaccine, which encodes HLA-A2-restricted cytotoxic T-lymphocytes from the E7 protein. In their small study of 12 men with HPV 16 infection and HLA-A2 restriction, 10 out of 12 evaluated cases developed increased immune response to the epitopes encoded within ZYC101, with continued elevation of immunity after six months. Palefsky et al. 51 developed another Bacilli–Calmette–Guerin-derived heat-shock protein (Hsp65) fused to the E7 protein of HPV 16. Of 82 HIV-positive patients with HSIL who were recruited and given the vaccine, half demonstrated moderate regression towards normal. The same vaccine showed a 71% regression to LSIL from HSIL within six months.
The recent suggestion of adding a quadrivalent HPV vaccine to standard vaccination schedules is in the forefront of cancer prevention. Two quadrivalent vaccines targeting the four main subtypes of HPV infection (6, 11, 16 and 18) were licensed for use in the European Union. The first vaccine (Gardasil, Mercke, Sharpe and Dohme, Whitehouse Station, NJ, USA) 52 was introduced in 2006 followed by another (Cervarix, GlaxoSmithKline Biologicals sa, Rixensart, Belgium) 53 in 2007. Again, there is a paucity of literature regarding the use of these vaccines in anal cancer, whereas a significant number of studies exist supporting their use in other female urogenital cancers. Chesson et al. 54 estimated the cost-effectiveness of implementing HPV vaccinations in 12-year-old girls in the United States. Their conclusion was that introducing HPV vaccinations in these girls was not only cost-effective, but also conferred the additional benefits of preventing anal, vulvar, vaginal and oropharyngeal cancers. Kulasingam et al. 55 analysed the cost-effectiveness of adding a quadrivalent HPV vaccine to existing cervical cancer screening programmes in the UK. They concluded that the vaccine would be cost-effective, with reductions in lifetime risks of cancer from 0.71 % to 0.29% in a cohort of 100,000 women.
While this is reason to introduce the vaccine in women, the overall incidence of anal cancer is still far below that of cervical cancer (0.8/100,000 versus 8/100,000, respectively) in the general population. Difficulty in determining the sexual preferences and future risk profiles in prepubertal men makes it less reasonable to vaccinate boys in order to deter anal cancer, especially since condylomas may spontaneously regress in healthy individuals. Kim et al. 56 estimated the cost-effectiveness of including boys in an HPV vaccination programme versus girls alone in a pre-adolescent vaccination programme against two subtypes of HPV (16 and 18) in Brazil. Assuming lifelong protection and up to 90% coverage in both genders, the vaccination of girls alone reduced cancer risk by 63%. The inclusion of boys into the vaccination programme only increased total cancer reduction by 4%. The cost of vaccinating girls resulted in <$200 PYLS, but including boys in the vaccination schedule resulted in $810–18,650 PYLS. At all coverage levels, the cost of vaccinating girls alone is more cost-effective than vaccinating both boys and girls in a vaccination programme.
However in the USA, the Food and Drug Administration Vaccines and Related Biological Products Advisory Committee agreed in September 2009 that the efficacy, immunogenicity and safety data from clinical trials in males supported the use of Gardasil in boys and men aged 9–26 years for the prevention of genital warts caused by HPV types 6 and 11. 57 Gardasil is currently approved for boys aged 9–15 years in the European union and elsewhere. Regulatory approval in other countries for young men and adolescents can be expected if ongoing efficacy trials remain positive. 58
Conclusion
Anal cancer is a growing threat to young men and women around the world, with strong links to HPV infection and anoreceptive intercourse. In just a few decades, the incidence of anal cancer has doubled and continues to rise at a rate of roughly 2% per year. Following success with Pap screening for detection of cervical cancer, a similar stratagem with anal Pap testing, clinical examination and high-resolution anoscopy is feasible in anal cancer. Studies have estimated anal Pap screening to be more cost-effective than Pap screening for cervical cancer. However, current treatment modalities are fraught with high recurrence rates and side-effects. With a growing number of HIV-infected people and wider acceptance of MSM in our community, increasing figures may only represent the tip of a much larger iceberg. The addition of a quadrivalent HPV vaccine to current vaccination schedules for girls may abolish the need for screening in females, but some studies show that including males may not be financially feasible, although such vaccination may become universal for herd immunity. As the lack of treatment success is the most significant obstacle to screening, more attention needs to be focused on the development of efficacious treatments for ASILs. Knowledge of risk cohorts for anal Pap screening is essential in caring for high-risk individuals. Until then, young HIV-positive men and women, who enjoy close-to-normal lives on HAART, are at serious risk of anal cancer.
