Abstract
Objectives
Multicentre randomized trials frequently encounter difficulties in meeting their recruitment targets, resulting in extension of the trial and delays in implementation of the findings. We report on recruitment strategies implemented in a randomized evaluation of computer-aided detection in women attending routine screening in the UK Breast Screening Programme.
Setting
The target population for the trial was identified from an existing NHS database of women aged 50–70 invited for routine mammography in Coventry, Manchester and Nottingham, UK. Women were asked to consent to their mammograms being randomly allocated (in a ratio of 28:1:1) to one of three film-reading protocols. Trial information was mailed to women, along with the invitation to attend screening, and informed consent was obtained at the mammography appointment. Several strategies were implemented to increase recruitment rates.
Results
Recruitment rate increased significantly over time in the study (P < 0.0010 in all centres) with an overall acceptance rate of 46% of those attending screening. Mailing of the trial information sheet separate from the screening invitation in Coventry and Nottingham increased the recruitment rate, even after adjustment for the trend over time and for socioeconomic status of the attendees (P < 0.001). Extension of recruitment to mobile screening units in Nottingham, and the presence of an additional member of staff also improved recruitment (P < 0.001). Simplification of the trial information sheet had little effect. Increases in recruitment rate were not attributable to socioeconomic status of the attendees.
Conclusions
In multicentre trials, monitoring of local recruitment protocols is required to ensure that each centre can maximize accrual targets.
Introduction
Randomized trials in screening populations pose an additional challenge where the low prevalence of disease requires large numbers of people to participate to provide adequate statistical power. 6 In the UK Breast Screening Programme (NHSBSP), much of the work is carried out in mobile screening units with short appointment times and there is considerable pressure on radiography staff to ensure that screening sessions are kept on schedule. Staff may be reluctant to accept the extra time commitment to discuss the trial and consent with participants,7–10 and there is limited space to accommodate any additional staff solely for participant recruitment.
The CADET II trial was a prospective evaluation of computer-aided detection (CAD) in the NHSBSP, 11 with a target to recruit 30,000 participants. It was estimated that this could be achieved within an eight-month period if 40–50% of women attending screening consented to participate. Extension of recruitment to other centres was precluded since this would have involved the lease and installation of additional CAD systems and reader training before new centres could commence participant recruitment. In this paper, we report on the monitoring of recruitment rates and proactive strategies that were explored to maximize recruitment at the three sites.
Methods
Setting
CADET II was a multicentre randomized trial involving women aged 50–70 years attending routine mammography at NHSBSP Services in Coventry, Manchester and Nottingham between September 2006 and August 2007. Women were asked to consent to their mammograms being randomly allocated (in a ratio of 28:1:1) to one of three reading protocols: double reading (current recommended NHSBSP practice) and single reading using CAD, double reading only or single reading only. No additional attendances would be required unless an abnormality was detected. 11
Invitation to participate in the trial
Advance mailing of the participant information sheet for the trial along with the invitation to attend screening was judged to be an appropriate and cost-effective method of informing women about the trial. Consent was obtained from the NHSBSP to liaise with screening centres to facilitate the mass mailing of trial information simultaneously with screening invitations. Informed consent was sought at the time of the mammography appointment on mobile screening units in Coventry and Manchester and in two static centres in Nottingham.
Separate mailing of information sheet
During the first few weeks of recruitment, participant accrual was significantly lower in Coventry and Nottingham than in Manchester where, for technical reasons, trial information was mailed separately from the invitation to screening. Separate mailing of trial information was implemented in Coventry in week 9. Staffing resource issues delayed separate mailing in Nottingham until week 16.
Revised information sheet
As feedback from screening centre staff indicated that women were having difficulty understanding the trial information sheet, an amended trial information sheet was submitted for research ethics committee approval. This was distributed from all centres from week 18.
Additional recruitment protocol in Nottingham
In Nottingham, recruitment was extended to mobile screening units from week 22 and a local National Cancer Research Network (NCRN) nurse or member of staff from the screening centre was in attendance one half day per week (from week 16).
Index of Multiple Deprivation
The Index of Multiple Deprivation (IMD) provides a proxy measure of socioeconomic status. Weighted scores in seven domains (income, employment, health deprivation and disability, education skills and training, housing and geographical access to services, living environment, crime) are combined into one summary score. 12 Using residential postcode data, IMD scores were derived for all women in the study data-set.
Statistical analysis
Rates of recruitment were analysed by Poisson (log-linear) regression 13 estimating the effects of changes in practice at the Coventry and Nottingham centres on recruitment. These effects were adjusted for the increasing time trend in recruitment over the period of the study and for the recruitment over time in Manchester where the only change in recruitment protocol was the use of an amended participant information sheet.
Results
Weekly recruitment at each centre
A total of 31,057 women were recruited into the study from 68,060 women attending routine screening mammography (Table 1). In the first week, 377 women were recruited, compared with 580 in week 45. On average, the numbers recruited increased by between four and five subjects per week. Accrual in Manchester remained fairly constant at an average of 215 per week, until an increase to 306 per week from week 30 onwards enabling this centre to achieve their accrual target in 43 weeks compared with 49 weeks in the other two centres.
Demographics of the study population
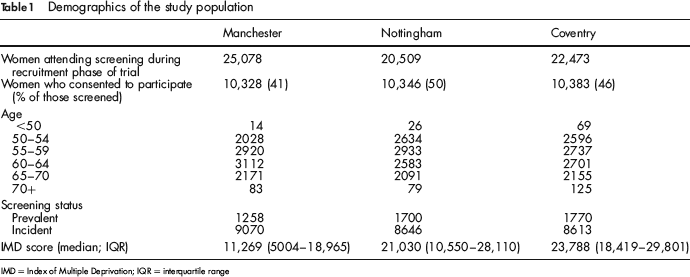
IMD = Index of Multiple Deprivation; IQR = interquartile range
Recruitment rates and IMD score
As shown in Figure 1, recruitment increased significantly over time in all centres (P < 0.001 in all cases) except around the time of Christmas and Easter.
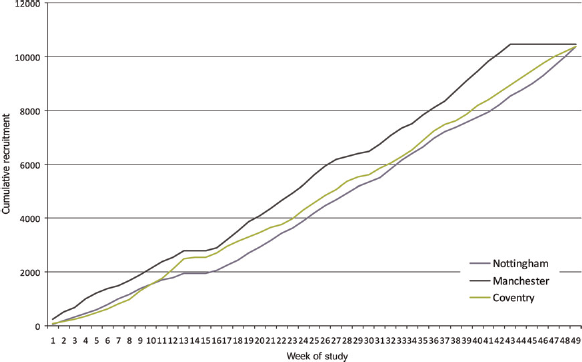
Cumulative recruitment into the CADET II study at each centre over time
To illustrate the Poisson regression analysis, we consider the results from Coventry. The mutually adjusted Poisson regression coefficients were 0.49 (95% CI 0.41–0.57, P < 0.001) after the separate mailing policy compared with before, 0.003 (95% CI 0.001–0.005, P = 0.001) per week of the study and 0.0001 (95% CI 0.000002–0.000019, P = 0.02) per unit IMD ranking. These translate to a 64% increase in recruitment after the separate mailing, a 0.3% average increase in recruitment per week of the study and a 0.001% increase per unit IMD ranking (corresponding to an approximate 7% increase per IMD quintile). The 64% increase after the separate mailing was significant after adjustment for the trend over time and the increased recruitment with greater affluence (higher IMD ranking).
A higher rate of recruitment was observed in association with higher IMD of the screenees in all centres (P < 0.001). Approximately contemporaneously with the separate trial mailing in Coventry and Nottingham an increase in recruitment rate was observed, significant in both cases, after adjustment for the trend over time and for IMD (P < 0.001). In Nottingham, extension of recruitment to mobile screening units and assistance with the consent process conferred another increase in recruitment rate, which was also significant after adjustment for the time trend and IMD (P < 0.001) (Figure 2).
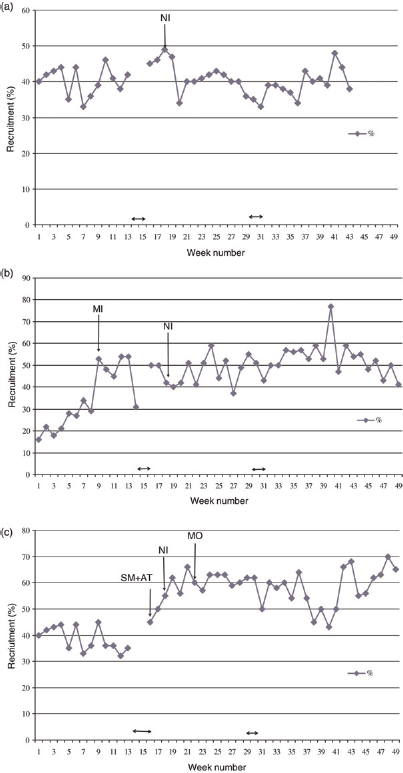
Weekly recruitment rate at each centre: (a) Manchester. NI = New trial information sheet; ↔ = Christmas/New Year and Easter holiday periods, (b) Coventry. SM = Separate mailshot of trial information sheet; NI = new trial information sheet; ↔ Christmas/New Year and Easter holiday periods, (c) Nottingham. SM + AT ↔ Separate mailshot of new trial information sheet and attendance of NCRN research nurse or member of staff from Nottingham Breast Institute 0.5 days per week from this time point; NI = new trial information sheet; MO = recruitment commenced from two mobile screening units; ↔ = Christmas/New Year and Easter holiday periods
Revised information sheet
From week 19, all women attending screening received the revised and simplified trial information sheet. Weekly recruitment rates in Coventry and Manchester remained steady, and in Nottingham the recruitment rate was significantly (P < 0.001) increased, after adjustment for the increasing trend with time and for IMD (Figure 1).
Discussion
The overall acceptance rate for the trial was 46% of those attending screening, which is considerably higher than that reported for recent UK cancer screening trials. 14 It has been suggested that a breast screening programme with high attendance provides an ideal setting to conduct additional research studies 15 with no evidence that attendance at screening is adversely affected by invitations to participate in adjunct studies.16, 17 Socioeconomic status has been shown to have a major influence on the uptake of breast screening, with lower uptake in more deprived areas.18, 19 In this respect our study population was a self-selected group since only women attending screening would be eligible to participate. However, our results indicated that the increase in weekly recruitment rate in Coventry and Nottingham after the introduction of separate mailing of trial information remained significant after adjustment for IMD, and could not be attributed to increasing affluence of the women attending screening.
There are two possible explanations for lower accrual rates when trial information was included with the invitation to attend screening. Women who previously attended screening may be less inclined to read any additional information and would only note the date and time of their appointment. This would be consistent with the results of a study in the Dutch Breast Screening Programme in which it was estimated that standard breast screening information was read by only 54% of women. 20 For women attending their first screening appointment, or for women who had a ‘false-positive’ experience at a previous attendance, 21 the inclusion of any additional information could further increase underlying anxiety and distress about attending breast screening.22, 23 Although separate mailing of trial information necessitated an additional postage cost, the trial reached its recruitment target without incurring additional expenditure on staff time and resources. Interestingly, simplification of the information sheet, the only modification of the recruitment protocol in Manchester, did not appear to have any significant effect on recruitment rates in either Manchester or Coventry. The concept of CAD and the need for a trial in the screening programme are difficult to explain in lay terms within the confines of a participant information sheet so it is perhaps not surprising that even a simplified information sheet may have had little impact. This may indicate that women were not reading the information sheet 20 or that they had difficulty understanding the original and the simplified version. 24
In Coventry and Manchester, all women recruited into the study attended mobile screening units, an environment that is not ideal for recruitment due to logistical limitations of space, time and available staff. In Nottingham, the weekly recruitment rate was enhanced when recruitment was extended to two mobile screening units and an additional member of staff attended some of the inner city clinics. A significant increase in weekly recruitment was observed in association with these, after adjustment for the time trend and for IMD. The effects cannot be separated as the changes occurred at almost exactly the same time. Although recruitment on the mobile units is almost certainly responsible for part of the increase, the ad hoc presence of an additional member of staff could also have contributed. This would be in agreement with a US study reporting that the physical presence of research staff resulted in a fourfold increase over passive recruitment by distribution of an information pack. 7 Independent staff with more time to inform women about the trial would also minimize any conflict of interest from clinic staff under pressure to maintain screening schedules.
Our data complement the observations of Menon et al. 2008. 14 Monitoring of recruitment rates and flexibility are required to adopt the most appropriate recruitment strategies for each centre's needs. In our study, enlisting the assistance of radiography staff in the consent process was a key element in achieving our target accrual. This was achieved by good communication with radiography staff to engage their support during the set-up phase of the trial. The concept of ‘selling’ a trial to clinical professionals and participants using business and marketing strategies has been reported in qualitative studies.25, 26
Strengths and limitations
Utilizing the NHSBSP mailing lists of women invited for routine breast screening provided a platform for the distribution of trial information and enabled differences in recruitment rates between centres to be monitored. Since the three participating centres were a representative sample of breast screening centres in England, the successful recruitment strategies should be applicable to future research in this population. A limitation of the study was that we were unable to separately evaluate the impact on recruitment rate in Nottingham by the presence of a member of staff dedicated to the research since this happened contemporaneously with the addition of recruitment from mobile units.
Conclusions
Our data have two important implications for research studies involving women attending the UK breast screening programme. Separate mailing of trial information was more effective than sending information along with the screening invitation, but with an additional cost. Secondly, despite the limitations of space and time on mobile screening units, it is feasible to recruit study participants by involving radiography staff in the consent process. This is facilitated by positive marketing at the start of the trial.
Footnotes
Acknowledgements
The authors thank E Fawcett-Gough, P Shann, S Berry and S Ferguson (Manchester); K Heer, M Wheaton and S Wright (Coventry); J Matthews and L Checkley (Nottingham) and staff at the Warwickshire, Solihull and Coventry Breast Screening Service, the Nightingale Centre, Manchester and the Nottingham Breast Institute for assistance with recruitment and data collection.
Preliminary data were presented at the Society for Clinical Trials Meeting in St Louis, USA (2008). Gillan MGC, Gilbert FJ, Flight HE, Heer K, Cooper J, Wallis MG, James J, Boggis CRM, Astley SM, Agbaje OF, Duffy SW. Strategies to increase participant recruitment into large-scale screening trials. Clinical Trials
