Abstract
We report an audit of our Prenatal Screening Programme for aneuploidies between March 2003 and August 2007. Overall detection and false positive rates were 86% and 3.6%, respectively. These results are similar to those predicted by the Serum, Urine and Ultrasound Screening Study (SURUSS).
We here report an audit of our integrated and serum integrated programme covering the period from March 2003 to August 2007. All biochemical measurements were performed with TRACE technology (BRAHMS Kryptor Systems). Risk calculation was performed with αLPHA software (Logical Medical Systems Ltd). Gestational age was estimated by ultrasound. The cut-off used to determine a screen-positive result was 1:300. We used obstetric and fetopathology records to identify Down's syndrome cases.
Table 1 shows a summary of our results. A total of 2290 women had the integrated test and 1009 the serum integrated test. The median age of women screened for the integrated and serum integrated tests was 30.6 years and 30.9 years, respectively. There were 14 cases of Down's syndrome observed during the audit period; eight of these pregnancies were screened using the integrated test, and six using the serum integrated test. The overall detection rate was 86% (95% confidence interval [CI] 67–100). The detection rates for the integrated and serum integrated tests were 87.5 (64–100) and 83% (53–100), respectively. The overall false-positive rate was 3.6% (95% CI 2.9–4.2); for the integrated test and serum integrated test, it was 2.5% (95% CI 1.9–3.1) and 6.0% (95% CI 4.5–7.5), respectively.
Results of Hospital S. Francisco Xavier Prenatal Screening Programme for aneuploidies between March 2003 and August 2007
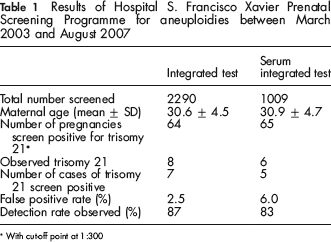
With cut-off point at 1:300
The false-positive rate for the integrated test (2.5%) was similar to that reported in the Serum, Urine and Ultrasound Screening Study (SURUSS) 1 for the same detection rate (about 85%). The serum integrated test false positive rate was 6.0%, also as expected. The number of cases observed was small but also consistent with the expected results from SURUSS. The addition of second trimester markers unconjugated oestriol (uE3) and inhibin-A would enhance screening performance. It is possible that a lower cut-off (for example, 1 in 250 instead of 1 in 300) would improve the false-positive rate without much loss in the detection rate.
This audit demonstrates that the integrated test and serum integrated test achieve false-positive rates in practice similar to those predicted by SURUSS.
