Abstract
We carried out an audit of antenatal screening for Down's syndrome using the Integrated test (which provides a single screening result from information collected in the late first and early second trimesters of pregnancy) which was introduced into routine antenatal care at two London hospitals, University College Hospital (UCH) and St Mary's Hospital, in 2003–4. The audit was based on 15,888 women who accepted screening and booked in the first trimester. The Down's syndrome detection rate was 87% (95% confidence interval [CI], 74–95) consistent with an expected detection rate of 89% based on applying the estimates of screening performance of the Serum, Urine and Ultrasound Screening Study (SURUSS) to the maternal age distribution of women who were screened at UCH and St Mary's. The observed false-positive rate was 2.1% (95% CI, 1.9–2.3), compared with an expected of 2.5% for women of the same age. An audit trail (conducted at UCH) indicated that 98% (10,746/10,961) of women accepted integrated screening (2% having a first trimester test) and of these, 94% (10, 116) completed both stages of the test. The audit demonstrated that it is feasible to conduct integrated screening within the NHS with a high acceptance rate and a screening performance consistent with that determined from previous research studies.
Introduction
Methods
The Integrated test for Down's syndrome is performed in two stages. At both hospitals the risk of a Down's syndrome pregnancy was estimated using maternal age, together with a measurement of the blood serum concentration of pregnancy associated plasma protein-A (PAPP-A) and an ultrasound measurement of nuchal translucency (NT), both performed between 10 and 13 weeks of pregnancy, and measurements of the blood serum concentrations of alphafetoprotein (APP), unconjugated oestriol (uE3), human chorionic gonadotrophin (hCG), either total hCG as used at St Mary's Hospital, or the free β subunit as used at UCH, and inhibin-A (at UCH only), all performed between 14 and 22 weeks of pregnancy, usually 15 or 16 weeks. At St Mary's, only the Integrated test was offered. At UCH, women were offered the Integrated test, but if they asked for a test with an earlier result, they had the option of the first trimester combined test (maternal age with NT, free β-hCG and PAPP-A).
The period covered by this audit of screening using the Integrated test was between 2003 and 2007 for UCH (including additional data to those previously reported 2 ), and between 2004 and 2007 for St Mary's Hospital. Measurements on all the Integrated test screening markers described above, together with maternal age, were recorded for 14,296 singleton pregnancies (9369 UCH and 4927 St Mary's). Biochemical tests on blood serum samples were performed at the Wolfson Institute of Preventive Medicine for UCH, and at Birmingham Women's Hospital for St Mary's Hospital and NT measurements were performed locally. In all women, gestational age was established by an ultrasound scan. The risk of a Down's syndrome pregnancy was estimated using the software αLPHA (version 6). In women with positive results (risk estimates > 1:150 at term) who chose to undergo amniocentesis, a PCR-based diagnostic test on the amniotic fluid gave the results in 48 hours. Down's syndrome pregnancies, including those missed by screening, were ascertained from hospital records, cytogenetic laboratories, and by linking data with the National Down Syndrome Cytogenetics Register (NDSCR) so that all cases should have been ascertained. Observed screening performance was compared with the expected screening performance based on the maternal age distribution of the women screened and the distribution of marker values in Down's syndrome and unaffected pregnancies estimated in the Serum, Urine and Ultrasound Screening Study (SURUSS). 1
Results
The median age of women screened was 33 (range 15–51). The proportion of women aged 37 or over at estimated date of delivery (EDD) was 20% (n = 2887). The observed prevalence of Down's syndrome was 3.3 per 1000 (95% CI 2.4–4.4). The estimate is lower than that expected based on the maternal age distribution of the women screened and the maternal age specific risk of an affected live birth3,4,5 (4.2 per 1000) because some women with high NT values (>4 mm at UCH and >3.5 mm at St Mary's) were offered antenatal diagnosis without serum testing (there were 14 affected pregnancies at UCH in this category). Table 1 shows the observed and expected performance of the Integrated test at the two hospitals (restricted to women who had singleton pregnancies and had all Integrated test markers measured). Forty-one of the 47 Down's syndrome pregnancies were detected, an observed detection rate of 87% or 84% allowing for the spontaneous fetal loss of affected pregnancies between about 16 weeks and term. The expected detection rate for women of the same age was 89%. The observed false-positive rate was 2.1% and the odds of being affected given a positive test result was 1:7.
Integrated test: observed (and expected) screening performance
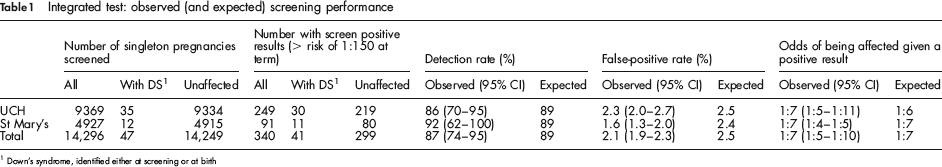
Down's syndrome, identified either at screening or at birth
Figure 1 shows the reported Integrated test risk estimates for the 47 affected pregnancies and the median, 95th and 99th centiles of the estimates for the 14,249 unaffected pregnancies. The median risk was 1:5 for affected and 1:23,500 for unaffected pregnancies, more than a 4000-fold difference. Among women with screen positive results (risk > 1:150 at term), the median risk was 1:3 for affected and 1:70 for unaffected pregnancies, which is reflected in the uptake of amniocentesis being 95% for Down's syndrome pregnancies and 68% for unaffected screen positive pregnancies (71% overall).
Reported Integrated test risk estimates in Down's syndrome pregnancies and selected centiles of risk in unaffected pregnancies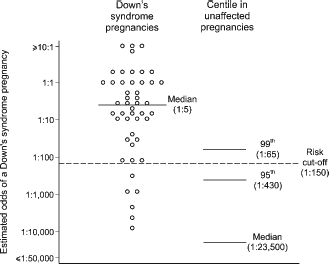
An audit trail of women's choices was available for UCH, but not for St Mary's Hospital. This is set out in a flowchart (Figure 2) of women who chose to be screened. Ninety-eight percent of women accepted the offer of an Integrated test (2% chose the first trimester Combined test) and 94% of these completed both stages of that test (10,116 pregnancies). Of these, 9898 women had an integrated test including an NT measurement and 218 had a serum integrated test because an NT measurement was not obtained.
Screening flowchart relating to women who attended in the first trimester of pregnancy and accepted Down's syndrome screening at UCH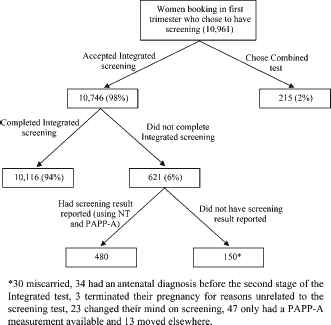
Table 2 shows the median gestational age at which the first and second trimester samples were taken, the proportion of samples taken late (after 20 weeks) for UCH and St Mary's, and the interval between samples. The median gestational age at the time when first trimester samples were taken was 12 weeks 4 days, and 16 weeks 1 day at the time second trimester samples were taken. Of women attending UCH, 4.6% provided a second sample after 20.0 weeks’ gestation, whilst at St Mary's Hospital this figure was only 0.2%, where a visit to the clinic at 16 weeks was part of routine antenatal care. The median interval between samples was 3 weeks 5 days (10th centile 2 weeks 2 days, 90th centile 5 weeks 5 days).
Gestational age of blood sampling

Discussion
The results of our medical audit of antenatal screening for Down's syndrome using the Integrated test demonstrate the feasibility and acceptability of this test in routine obstetric practice. This was also recently shown in Ontario, Canada. 6 Our results show that the observed screening performance was close to that expected from the results of SURUSS (the detection rate would have been a little higher had CVS or amniocentesis not been offered directly to some women with high NT measurements).
The audit specifically demonstrates the high acceptance of integrated screening, with only 2% of women included in our audit trail electing to have a first trimester screening test. The median interval between the first and second stages of the Integrated test was, as planned, about four weeks. The completion rate of the Integrated test was 94%, indicating that women re-attending to provide a second blood sample was not a major problem. Among the women who did not attend for a second stage of the test, about one-fifth miscarried, another fifth had an antenatal diagnosis, and the remaining three-fifths comprised women who either changed their mind on screening, had no NT measurement (so no screening report could be issued) or moved to another hospital without any further details being available.
Women with high risks had a higher uptake of amniocentesis, and because women with Down's syndrome pregnancies had higher risk estimates, they had a higher uptake of amniocentesis than women with unaffected pregnancies (95% vs 68%).
The initial UK Government recommendation supported the use of the Integrated test (NICE guidelines 2003 7 ) but in March 2008 these were revised in favour of first trimester only screening (NICE guidelines 2008 8 ) on the grounds of uncertainty over acceptability and feasibility of integrated screening within the NHS. This concern seems to have outweighed both the increased efficacy and safety associated with screening using the Integrated test and the evidence that women prefer a later screening test if it is more effective and results in a safer screening programme. 9 The expected detection rate in our screening program was 89%; if the women had had the first trimester Combined test (NT, hCG and PAPP-A) instead of the Integrated test, the expected false-positive rate would have been 9.4% instead of 2.5% for the same detection rate.
This audit shows that it is acceptable and feasible to conduct integrated screening within a public health-care system such as the UK National Health Service.
Declaration
NJ Wald holds a patent for Integrated screening.
Footnotes
Acknowledgements
The authors thank Vicky Edwards at Birmingham Women's Hospital for providing the screening data for St Mary's Hospital.
