Abstract
Objectives
The objective of this study was to compare breast cancer outcomes among women subject to different policies on mammography screening frequency.
Setting
Data were obtained for women participating in the Screening Mammography Programme of British Columbia (SMPBC) for 1988–2005. The SMPBC changed its policy for women aged 50–79 years from annual to biennial mammography in 1997, but retained an annual recommendation for women aged 40–49 years.
Methods
Breast cancer outcomes were compared for women participating in the programme before and after 1997 for two groups: ages 40–49 and 50–79 years.
Results
There were data on 658,151 women. Comparing pre-1997 and post-1997, the median interscreen interval increased by 11.1 months in women 50–79 but by only 0.3 months in women aged 40–49. Excluding those detected at initial screen, 6291 breast cancers were identified. Comparing pre-1997 and post-1997: the relative rates (RR) of screen detected cancer increased in women aged 40–49 (RR = 1.32) and the rate of invasive cancers ≥20 mm at diagnosis decreased (RR = 0.83); the rate of cancers with axillary node involvement increased in women aged 50–79 (RR = 1.23). Cancer survival improved after 1997 for women diagnosed at ages 40–49 (hazard ratio = 0.62), but was unchanged for women aged 50–79. Breast cancer mortality rates did not change between the periods in either age group.
Conclusion
The proximal cancer outcomes considered (staging and survival) improved in women aged 40–49 but this was offset in women aged 50–79 associated with the change in screen frequency. These changes did not result in alterations in breast cancer mortality rates in either age group.
INTRODUCTION
Combined analyses of clinical trials have shown reductions in breast cancer and overall mortality in women randomized to invitation for screening mammography. 1,2 Consequently, mammography has become a recommended method for breast cancer screening in many countries. 3 The relative contribution of mammographic screening to observed declines in the population of breast cancer mortality rates in different countries is an area of some uncertainty 4–7 and is influenced by the age groups and time for which screening has been offered. Results from service screening conducted in different countries over the last two decades has provided added information on the positive impact of screening mammography on breast cancer mortality 6,8–11 including in Canada. 12
Most publicly funded breast screening programmes in Europe and Canada include biennial screening for women aged 50–69. 13 Annual screening is commonly used by USA health-care providers 13 and is recommended by some authorities there. 14 Randomized trials of sufficient size to measure differences in breast cancer mortality with screening frequency have not been conducted. Studies of the effects of screen frequency have primarily involved indirect measures 1 or have used surrogate outcomes 15–22 and only one was randomized.
The Screening Mammography Programme of British Columbia (SMPBC) was established in 1988 to provide breast screening to women in British Columbia (BC). 23 Women aged 40–79 were eligible to self-refer and received screening mammograms free of charge through community clinics or mobile vans with interpretation by radiologists affiliated to the programme. 24 From programme inception, women aged 40–49 have been recommended for annual re-screening and have received reminder letters to encourage return. In July 1997, the SMPBC changed its screening recommendation for women aged 50–79, from annual to biennial. At that time, the SMPBC initiated enhanced recruitment efforts targeted on women aged 50–74, including personal invitation letters at their 50th birthday. Women aged 50–79 were still permitted to return annually but programme information was altered to support the new policy and the timing of reminder letters was changed from 12 to 22 months in women over the age of 50.
Here, we report an analysis of breast cancer cases and deaths in women aged 40–79 at the time of their first SMPBC screen and compare the experience before and after the policy change.
PATIENTS AND METHODS
Data were obtained from the SMPBC data-base, the British Columbia Cancer Registry (BCCR) and the Vital Statistics Agency death file (VSA) comprising death notifications in residents of BC.
The study sample consisted of women aged 40–79 when they were first screened by the SMPBC between July 1988 and December 2005. Women entered the sample at the date of their first SMPBC mammogram. Breast cancer cases and deaths in the sample were ascertained through linkage with the BCCR and the VSA. Linkage was performed in two steps. First the VSA and BCCR were linked to identify the cause and dates of death in all breast cancer cases diagnosed in the study period. A second linkage to the VSA identified dates of death of individuals in study subjects not diagnosed with breast cancer. The results of the two linkages were combined. Linkage was performed using Automatch, 25 with personal health number (a unique identifier of the BC health system), address, name (first, middle, last and birth) and dates of birth and diagnosis as linking variables as necessary. The resulting data consisted of dates of birth, death, entry to the sample and first breast cancer, postal code of residence at last screen, pathology details of breast cancer and cause of death. Information extracted on breast cancer cases included mode of detection, initial presentation of disease and date, and cause of death. Data on all subjects were considered complete on 31 December 2005. A breast cancer was considered to be screen-detected if it was diagnosed within 12 months of an abnormal screening mammogram. Breast cancers not detected by screening were classified as interval (postscreen) cancers. In cases where death from breast cancer was recorded without any preceding diagnosis, the date of death was used as the date of diagnosis. Women with a prior diagnosis of breast cancer were not eligible to attend the SMPBC.
The sample was divided into four separate cohorts based upon date and age at first screening as follows: Pre97u50 (first screened prior to July 1996 between ages 40 and 49), Pre97o50 (first screened prior to July 1996 between ages 50 and 79), Post97u50 (first screened in or after July 1996 between ages 40 and 49) and Post97o50 (first screened in or after July 1996 between ages 50 and 79). July 1996 was selected as a cut-point as women aged 50–79 screened after that date would have received a reminder letter at 22 months as determined by the biennial policy.
Analyses of mammography sensitivity, breast cancer disease extent, survival and mortality rates omitted the contribution of cancers detected at first screening, as first screens would not be impacted by the policy change. Analysis of trends in screen sensitivity was undertaken by examining the 12-month postscreen invasive cancer detection rates. Analyses of screen sensitivity and the rates of cancer by disease extent at diagnosis were performed using Poisson regression with age adjustment and years-at-risk as offsets. Event rates (e.g. mortality, survival, time to next screen) were calculated using the Kaplan–Meier method. 26 Survival analyses excluded women recorded as dying from breast cancer but who had no earlier recorded diagnosis. For survival analysis death from breast cancer was used as the endpoint and cases were assumed to be at risk until the first of 31 December 2005 or death. Tests of significance for survival were made using Cox regression. 26
For mortality calculations, time at risk of breast cancer death was assumed to start from the first screen and terminate at a cohort-specific follow-up date which depended upon age, the date of screening policy change and the end of follow-up. Specifically these were: Pre97o50, first of 30 June 1997 or attaining age 80; Pre97u50, first of 30 June 1997 or attaining age 50; Post97o50, first of 31 December 2005 or attaining age 80; Post 97u50, first of 31 December 2005 or attaining age 50. Observed deaths consisted of all deaths attributed to breast cancer. Mortality rates between cohorts were compared using Poisson regression where death from breast cancer was the outcome, years-at-risk was included as an offset and age and cohort were included as covariates.
RESULTS
There were 658,151 women in the study sample. Forty percent of women were first screened prior to July 1996 and 49% of women were first screened before age 50. Table 1 provides the distribution of factors for the four study cohorts. Women in the earlier calendar period cohorts had a higher median age than the later cohorts. Time between screens, measured by the median interval between successive mammograms, was 13–14 months for those cohorts with an annual screening recommendation and 24 months for the biennial cohort. However, there was substantial individual variation in screening interval length. The first screening interval length exceeded 30 months in 35% (31%) and was between 18 and 29 months in 54% (17%) of women with a biennial (annual) recommendation. Overall, 34% of the invasive cancers, 44% of the DCIS and 28% of breast cancer deaths were associated with findings on the first screen (Table 1). The distribution of cancer cases and deaths (prior to the cohort-specific follow-up endpoints) by time, since preceding screen and mode of detection (screening and interval) for the four cohorts is provided in Table 2. The proportion of cancers detected by screening differed between the two time periods: Pre97u50-57% versus Post97u50-64% (P = 0.001) and Pre97o50-74% versus Post97o50-72% (P = 0.02) (Table 2). The pattern of occurrence reflected screening activity with the majority of screen-detected cancers occurring around the respective screening anniversary and the majority of interval cancers occurring prior to that time. Nevertheless, between 30% and 35% of cancers occurred more than 18 months after the last preceding screen in annual screening cohorts while 21% of cancers occurred after 30 months in the biennial cohort (Table 2).
Number of women, age at first screen, number of screens, time between screens and follow-up time for the four study cohorts*
*Prior to cohort-specific follow-up time
Breast cancers and breast cancer deaths by mode of detection and time since preceding screen for the four study cohorts*
*Prior to cohort-specific follow-up time
The rate of postscreen invasive cancers in the first 12 months (per 100,000 person-years) following a negative screening mammogram was as follows: Pre97u50 = 80.4, Post97u50 = 54.4, Pre97o50= 67.4 and Post97o50 = 64.3. After adjustment for age, the relative rate (Post- to Pre-97), RR and 95% CI, of such cancers was RR = 0.83 (0.73, 0.95) with no interaction with age (P = 0.24). The distributions of subjects' pathology characteristics, excluding those diagnosed at the first screen, are provided by cohort in Table 3. Age-adjusted Poisson regressions were performed comparing cases before and after 1997 in the two age groups for screen detection (yes/no), invasive cancer within 12 months of a negative screen, degree of invasion (invasive, ductal carcinoma in situ [DCIS]), tumour size (<2, ≥2 cm), grade (poor, other) and axillary nodes (positive, other). The results of these analyses are presented in Table 4. There were differences between the periods in both age groups.
Prognostic characteristics percent (counts) of cancers, excluding those detected at the first screen, in the four study cohorts
*Invasive cancers only
†Missing values: tumour size – 209; grade – 436; axillary nodes – 121
Age-adjusted relative rates of cancer types excluding those detected at first screen comparing post-1997 with pre-1997 for women aged 40–49 and 50–79
*Invasive cases only
Breast cancer-specific survival rates were calculated for invasive cancers, excluding those diagnosed at first screen, in each of the cohorts. There was a significant difference in hazard rates of breast cancer survival between pre- and post-97 in cases diagnosed before age 50, HR = 0.62 (0.4, 0.97) but not in those diagnosed after age 50 (HR = 1.10 [0.86,1.41]). Figure 1 provides the survival curves by cohort. The age-adjusted hazard ratio for the difference in breast cancer survival for women pre-versus post-97 for those over 50 relative to the trend in those under 50 was HR = 1.78 (1.05, 2.97), primarily reflecting the improvement seen in those diagnosed before age 50.
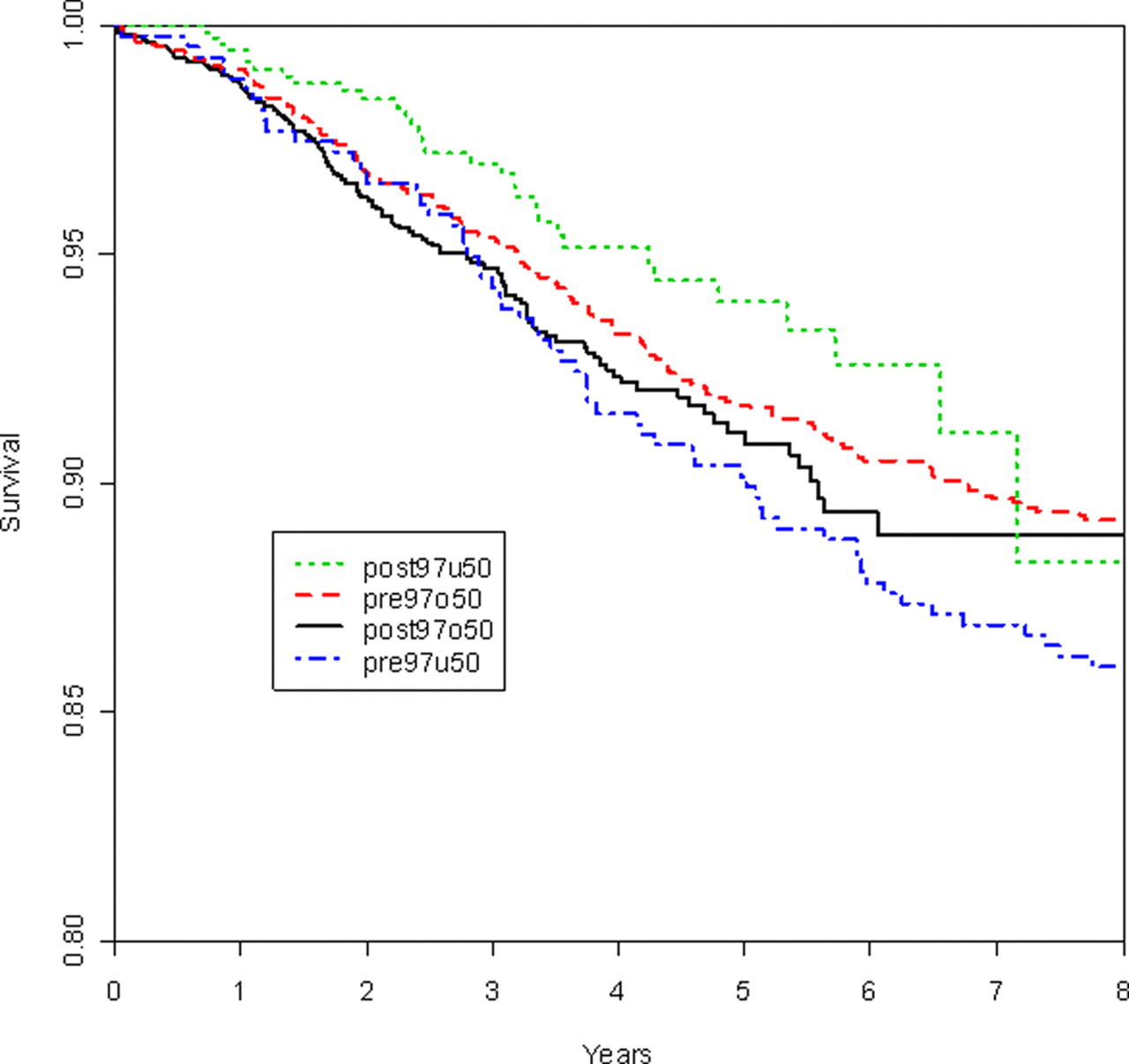
Breast cancer-specific survival rates for invasive cancers for the four cohorts excluding those detected at first screen and those and reported breast cancer deaths without a preceding diagnosis
The distribution of breast cancer deaths occurring prior to the cohort-specific follow-up times are given by cohort and time since preceding screen in Table 2. Excluding deaths associated with cancers detected at the first screen, interval cancers contributed the majority of the deaths in each cohort (Table 2). Cumulative breast cancer mortality rates for the four cohorts excluding women diagnosed at first screen are shown in Figure 2. As anticipated, breast cancer mortality rates were lower in the younger cohorts. Cumulative mortality rates in each of the two calendar periods were similar within each age group. Poisson regression analyses were conducted with years at risk as an offset, age in quintiles and period in each pair of cohorts to compare breast cancer mortality between the two time periods after adjustment for age. The resulting mortality ratio (MR) estimates for breast cancer death were: under 50 years (post-versus pre-97) MR = 0.67 (0.33, 1.37) and over 50 years MR = 1.06 (0.76, 1.46). There was no statistically significant difference in mortality from breast cancer between the two periods in either age group. The age-adjusted hazard ratio for breast cancer death for women pre-versus post-97 for those over 50 years relative to the trend in those under 50 years was HR = 1.57 (0.71, 3.44).
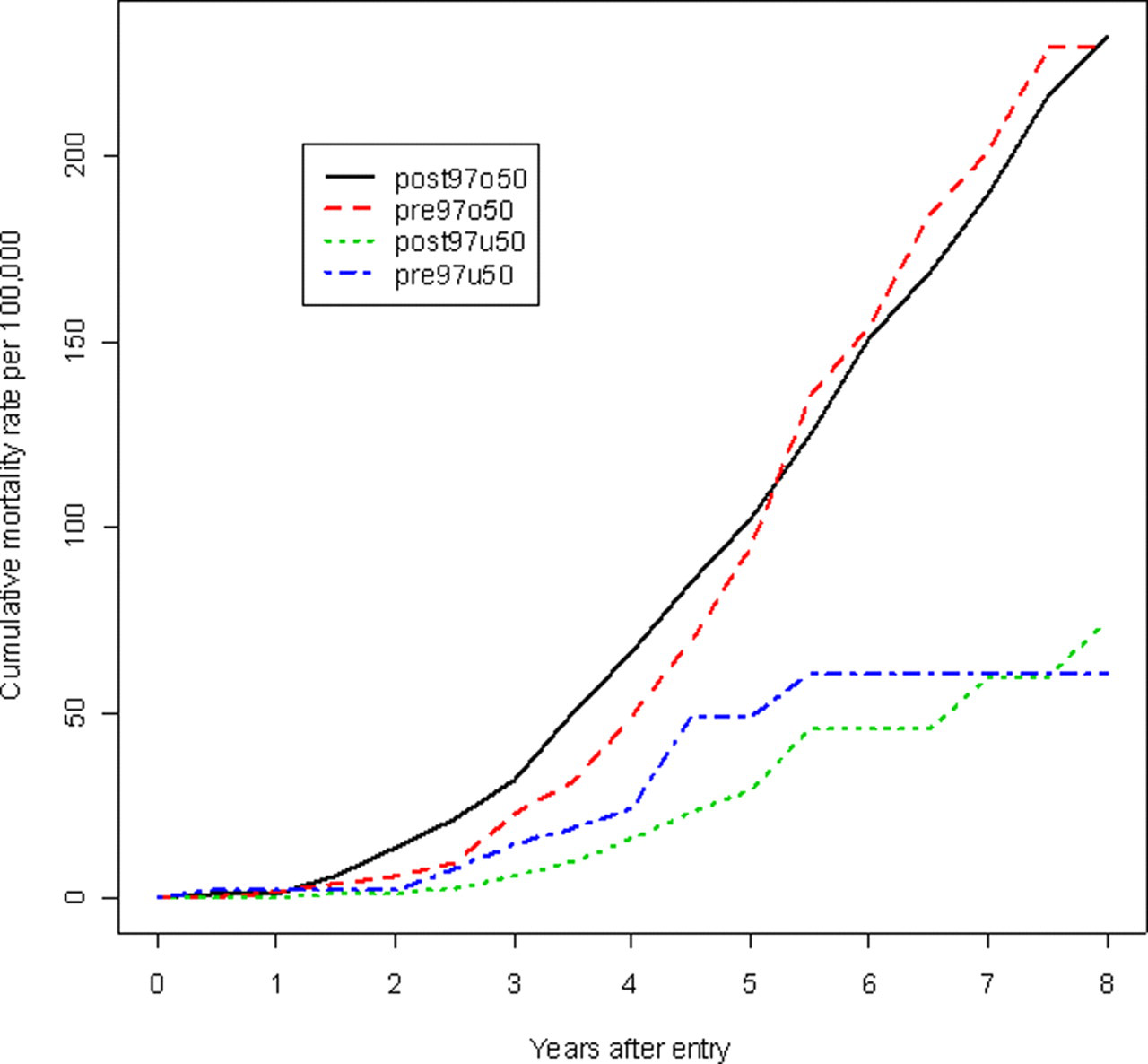
Cumulative mortality rates of breast cancer for the four cohorts excluding deaths from those detected at first screen
DISCUSSION
BC women aged between 50 and 79 are currently recommended for biennial screening. 27 However, for the first nine years of operation of the SMPBC all women were recalled annually for re-screening. The change in policy followed a review of published evidence available at that time and a comparison of the prognostic profiles of cancer detected in BC women who returned annually and biennially. Resources were shifted from screening women over age 50 more frequently to recruiting a larger number of women to be screened biennially. This policy change has provided an opportunity to review the effect of screening frequency in a large population of women.
No randomized trial has been mounted to test the effect of mammographic screening frequency on breast cancer mortality. A randomized trial examined the characteristics of breast cancers among women aged 50–62 diagnosed in a three-year period after a first screen with one group screened triennially and the other annually. 18 In that trial there were a total of 443 invasive cancers diagnosed and in the annual arm, the tumours were smaller but no significant change was noted in nodal involvement or tumour grade. Although not designed to compare changes in mortality, results based upon cancers diagnosed in the first three years of this trial among women with an initial screen, but excluding cancers diagnosed on that screen, showed a non-significant 7% reduction in breast cancer mortality. 28
A Dutch case-control study 29 used deaths from breast cancer as cases and living women from the same community as controls. The screening programme had successively increased screening interval length and mortality reductions were compared by length of the preceding screening interval. Intervals of one year or less had a similar protective effect to those of length one to two years and the protective effect subsequently decreased as the interval lengthened. However, there were relatively few deaths observed so that the precision of estimates of the relative effectiveness was low.
Several studies have reviewed prognostic factors in subjects diagnosed with breast cancer and correlated these with time since last normal screening mammogram 16,17,19–22 , with some finding a beneficial effect of annual versus biennial screening on size at detection. 16,18 Such studies are subject to the lead-time and selection biases. 30 The largest study utilized a case-control design and included 7840 cancers diagnosed in US Breast Cancer Surveillance Consortium participating centres. 17 The same study found that annual screening was associated with a reduced proportion of late-stage disease (positive lymph nodes or metastases) in those cancers diagnosed between ages 40 and 49. There was a trend towards better prognosis disease based on oestrogen receptor status in annual screenees, but no other significant trends were observed in women over age 50.
In an analysis of data for 50–74-year-old women, based on data that overlapped that used in the study presented here, a predictive model estimated an odds ratio of breast cancer death of 0.88 for annual compared with biennial mammography screening. 22 The predictive model used was similar to one used previously 31 and was calibrated to the observed impact of prognostic factors on survival in BC. The predicted reduction in breast cancer death in that analysis resulted primarily from the difference in outcomes between screen-detected and interval cancers and the reduction in the proportion of interval cancers in women screened annually.
The current study is an observational one and represents a temporal analysis of the effectiveness of a policy change in screening on breast cancer outcomes in women aged 50–79 in a population setting. Women did not rigidly adhere to the screening recommendations with more than 30% not returning within 30 months of their initial screen. Follow-up time is also limited in the study cohorts and mortality differences arising from screening take several years to become apparent. 2 Mortality rates in the cohorts are influenced by changes in treatment which have occurred during the study: a period which has seen a growing use of systemic therapy.
In this study trends in rates of cancers within prognostic categories were different in the two age groups with increased likelihood of smaller size and improved grade among 40–49-year-olds in the later period: this likely resulted from the increase in the rate of screen detection. The improvement in the rates by prognostic subtype was accompanied by improved survival but no significant change in the breast cancer mortality rate. Conversely, among women 50–79 in the later period, the rate of axillary node involved cancers increased but survival and mortality did not change significantly. As screening-related changes in survival precede changes in mortality, 23 we would not predict future differences in mortality between the two periods for women over 50 in this study.
Adjusting for potential secular trends in mortality using the 40–49 age cohorts altered the estimated MR to 1.57 between the two intervals in women over 50 years. However, the CI (0.71, 3.44) is wide and does not indicate whether the trend in women subject to the policy change (over 50 years) is different from those not (under 50 years). The estimated ratio is mainly a result of a (non-significant) decline in mortality for those under age 50 rather than an increase in those over age 50.
It would be anticipated that less frequent screening would be associated with a reduced rate of screen-detected cancers although this was not observed in women over 50 years. There was an overall decline in postscreen cancer detection prior to 12 months and an increase in the rate of screen-detected cancer in women under 50 years. These observations suggest that there existed a general trend towards increased sensitivity of mammography over the study period whose effect was offset in women over 50 years by the change in screen frequency. On average, screen-detected cancers have a better prognostic profile than interval cancers and thus one would anticipate, other things being equal, that breast cancer mortality would be reduced by more frequent screening. Stage shift alone is not sufficient to guarantee mortality reductions by screening, but is necessary. Few studies have been able to report on observed breast cancer mortality and none have reported a significant difference between annual and biennial screening. It would be anticipated that the relative effect of screening frequency would be most pronounced among women aged 40–49. However, this age group has a lower risk of breast cancer death making measurement of mortality differences more challenging.
In conclusion, this study found that the policy change in women over the age of 50 did influence the frequency of screening. The change in frequency did not affect the rate of screen-detected cancers but there did appear to be other, potentially confounding factors, which were influencing the sensitivity of screening overall. There was evidence that screen frequency and the confounders were influencing the prognostic appearance of cancers but no resulting effect on breast cancer mortality was observed.
Footnotes
ACKNOWLEDGEMENTS
The authors would like to acknowledge the assistance of Dr E Sickles who provided valuable commentary on earlier versions of this manuscript and a reviewer who made several helpful suggestions.
