Abstract
A comparison of cervical cancer incidence and mortality in the Czech Republic with that from other countries shows that the burden of cervical cancer here is considerably higher than in Western Europe, where screening is widespread. In May 2008, the International Conference on Human Papillomavirus in Human Pathology was convened to review the latest evidence and to formulate consensus recommendations for the reduction of cervical cancer rates. The Czech Republic is spending considerable resources on cervical cancer prevention, but these resources are being used inefficiently. The current system is characterized by a lack of coordination and monitoring that leads to the over-screening of a minority of women while the majority of the target population are under-screened or not screened at all. It was recommended that a comprehensive, organized programme be implemented, coordinated by an independent administrative body with legal and budgetary responsibility. As the laboratory infrastructure and professional technical skills required for a quality-assured organized screening programme are already in place, implementation of this programme would not require much in the way of additional resources to produce substantial cost-effective reductions in cervical cancer rates.
INTRODUCTION
Cervical cancer remains a serious public health problem in the Czech Republic. As the sixth most common cancer in Czech women, it represents a substantial, but unnecessary burden on the Czech population. Comparison of the Czech incidence and mortality data with databases recently compiled at the European level reveals that the burden of cervical cancer in the Czech Republic is considerably higher than in Western European countries where screening is widespread (Table 1, Figure 1). 1,2
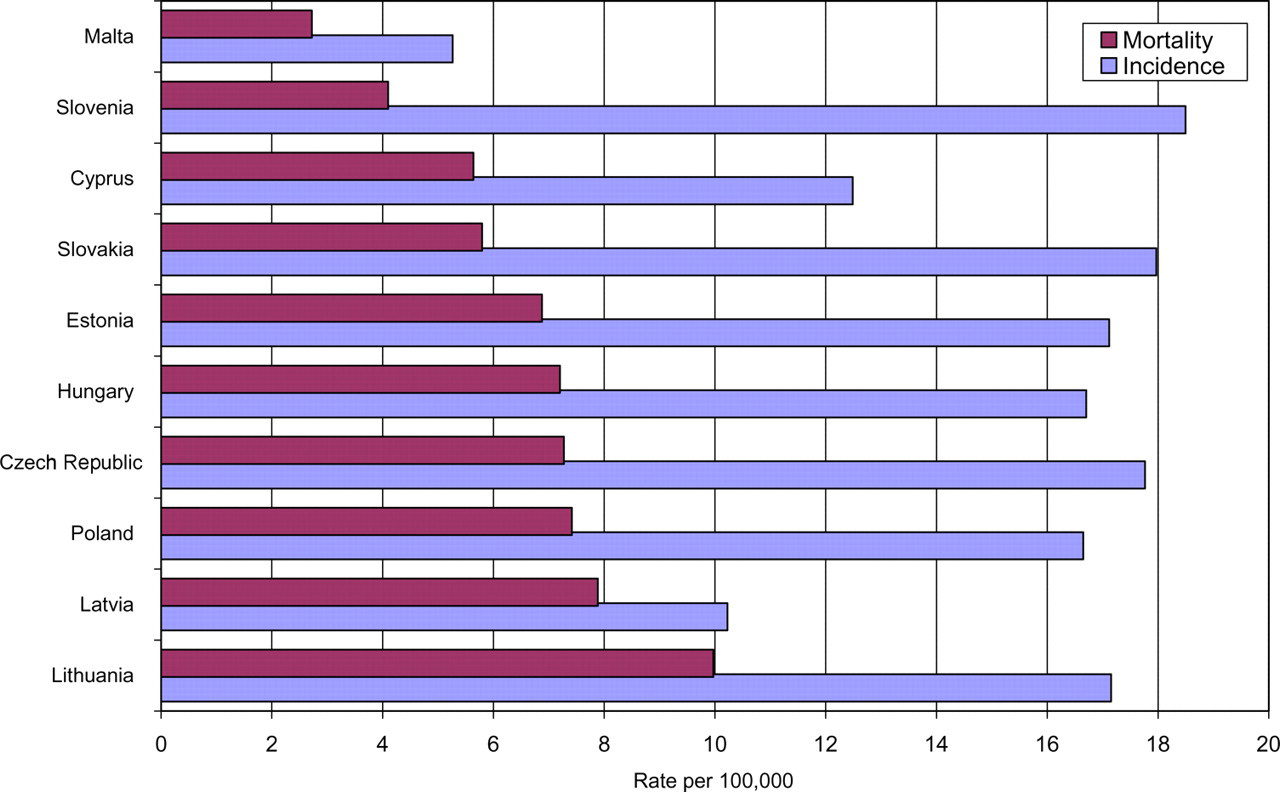
Age-standardized rates of incidence and mortality from cervical cancer (/100,000 women-years) in the 10 member states that joined the European Union in 2004, ranked by increasing mortality, estimates for 2004 (direct standardization using the world reference population). Source: ÚZIS CR, NOR CR 2006, 4 Slovenia (Slovenian Cancer Registry 2003), other member states 1,2
Incidence and mortality from cervical cancer (corrected for NOS): number of cases or deaths, crude and standardized rates and cumulative rates up to the age of 74 years; for 2004, in parts of the European Union and the Czech Republic
EU15, 15 old member states belonging already to the European Union before 2004 (Austria, Belgium, Denmark, Finland, France, Germany, Greece, Ireland, Italy, Luxembourg, Netherlands, Portugal, Spain, Sweden, the UK)
EU27, EU15 + 10 member states that acceded to the EU in 2004 (Cyprus, Czech Republic, Estonia, Hungary, Latvia, Lithuania, Malta, Poland, Slovakia, Slovenia) + Bulgaria and Romania, which acceded to the EU in 2007
HISTORY OF CERVICAL CANCER SCREENING
The Czech Republic was one of the first countries in the world to introduce cervical cancer screening based on the Pap smear in 1947 and it was also one of the first to make it widely available to women when in 1966 the Czech Republic parliament enacted legislation allowing all women to have a free annual gynaecological examination that included a Pap smear. 3 As a result of these measures, the incidence of cervical cancer decreased steadily from 32 cases/100,000 women in 1960 to 21/100,000 in 1990. 4 Since then, the incidence rate has remained stable at this level in spite of increased availability of opportunistic screening. Coverage of women in the screening age (30–59 years) is estimated at 35%. 5 In contrast, organized screening programmes with direct invitation of women and full quality assurance at all levels have been shown to provide the maximum reductions in cervical cancer rates, to provide the most equitable coverage of the population and to make the most cost-effective use of health-care resources. 6–10
To date, the accepted standard for cervical screening has been the Pap test. However, new technologies such as testing for human papillomaviruses (HPV) and HPV vaccination have been demonstrated to be highly effective in reducing cervical cancer precursors. 11–17 Therefore, modern and sustainable programmes for cervical cancer prevention must now consider the integration of these technologies based on these data, together with a full assessment of national cost-effectiveness analyses, health-care priorities and budgets.
In recognition of these issues, the International Conference on Human Papillomavirus in Human Pathology 18 was convened to bring relevant Czech experts, including members of the Working Group for the Cervical Cancer Screening of the Czech Ministry of Health, together with their international colleagues to review the latest evidence on cervical cancer prevention and to incorporate these data into consensus recommendations for the implementation of a modern cervical cancer prevention programme.
EXISTING INFRASTRUCTURE IN THE CZECH REPUBLIC
Recommendations for the implementation of an organized cervical cancer prevention programme in the Czech Republic must take into account the existing infrastructure that will form the foundation for that programme. This infrastructure includes:
Existence of a national unique personal identification number that can be used as the basis for an invitational screening programme; A national cancer registry established in 1976, is recognized by the International Association of Cancer Registries and can participate in quality assurance and programme evaluation; Sufficient Pap test processing capacity for a national organized programme through a network of some 50 cytology laboratories that all use the Bethesda cytology classification system as recommended in European guidelines;
19,20
Sufficient clinical capacity for the follow-up of women with abnormal cytology through a nationwide network of some 1500 gynaecologists trained and equipped for cervical screening, as well as diagnosis and treatment of screen-detected cervical intraepithelial neoplasia; A national colposcopy training programme with expert certification; Recommendations for management of women with precancerous lesions or cervical cancer, which is in accordance with European guidelines;
21
A network of 30 HPV testing laboratories with an established national quality assurance programme;
22
An internationally recognized National Reference Laboratory for Papillomaviruses which is a member of the International Network of Quality Assurance (QA) in HPV testing and which administers the Czech national HPV QA programme;
22
The Institute of Biostatistics and Analysis which already administers the screening registries for breast and colorectal cancer and which could easily be expanded to include a cervical cancer screening registry.
Therefore, many essential elements of an organized cervical cancer screening programme as set-out in the European Guidelines for Quality Assurance in Cervical Cancer Screening are already in place, but there remains a need to ensure an effective coordination and quality control of these elements.
CONSENSUS RECOMMENDATIONS OF THE INTERNATIONAL CONFERENCE ON HUMAN PAPILLOMAVIRUS IN HUMAN PATHOLOGY FOR CERVICAL CANCER PREVENTION IN THE CZECH REPUBLIC
Recognizing the infrastructure existing in the Czech Republic, the International Conference on Human Papillomavirus in Human Pathology recommends the following steps be undertaken to complete the implementation of a cost-effective organized cervical screening programme:
To set up a central administrative structure with overall legal and budgetary responsibility for:
Establishing the screening policy; Integrating the components involved in the implementation of the programme; Monitoring the quality and the performance of the programme; To allocate sustained financial resources that are sufficient to cover all components of the screening process through a specific item in the Ministry of Health budget; To provide effective, efficient and legally sanctioned access to a current population-based database to be used for programme recruitment; To establish a centralized registry including individual linkages to cervical cytology, colposcopy, histology and HPV test results to be used for call–recall, tracking of screen-positives, programme monitoring and QA purposes; To provide effective and efficient access to a cancer registry for programme audit including the confidential audit of screening histories of women with cervical cancer; Where they do not already exist, to establish training standards, professional guidelines and performance indicators for each stage in the screening process in accordance with scientific evidence and international benchmarks; To establish legally binding QA regulations and procedures to form an integrated QA policy covering the entire screening process from initial recruitment to the follow-up and management of women with cervical disease; To establish educational programmes and resources for the general public and health-care professionals with dedicated budgets for their execution; To establish mechanisms to identify and invite under-screened and unscreened women; To define a population-based HPV vaccination strategy that is fully integrated within the screening programme in order to be able to distinguish vaccinated cohorts from non-vaccinated cohorts and to assess the effects of vaccination a vaccination registry must be created that is linkable to the screening and cancer registry; To implement a randomized health policy evaluation to properly assess the logistics of using HPV testing as a primary screening test within the Czech screening programme.
The Czech Republic is spending considerable resources on cervical cancer prevention, but these resources are being used inefficiently. Implementing the remaining elements of an organized screening programme would inevitably produce substantial reductions in cervical cancer rates while saving money for the Czech health-care system.
Footnotes
ACKNOWLEDGEMENTS
The authors are grateful to the invited speakers (listed in alphabetical order): Ahti Anttila (Finland), Christine Bergeron (France), Gary Clifford (France), Carl Chow (UK), Joakim Dillner (Sweden), Lena Dillner (Sweden), Magdalena Grce (Croatia), Lalle Hammarstedt (Sweden), Amanda Herbert (UK), Ivan Hirsch (France), Ole Erik Iversen (Norway), Joseph Jordan (UK), Kateřina Lehovcová (Czech Republic), Ladislav Masák (Slovakia), Chris Meijer (Netherlands), Michael Pawlita (Germany), Marek Pluta (Czech Republic), Charles Redman (UK), Peter J.F. Snijders (Netherlands), Marek Spaczynski (Poland), Stina Syrjanen (Finland), Sjoerd van der Burg (Belgium), Maja Žakelj (Slovenia) as well as to the participants of the International Workshop on Human Papillomavirus and Consensus Recommendations for Cervical Cancer Prevention coming from Austria, Belgium, Bosna and Herzegovina, Brazil, Croatia, the Czech Republic, Denmark, Finland, France, Germany, Greece, Hungary, Ireland, Italy, the Republic of Korea, Mexico, the Netherlands, Norway, Poland, Portugal, Romania, the Federation of Russia, Serbia and Montenegro, Slovakia, Slovenia, Spain, Sweden, Switzerland, Thailand, Tunisia, Turkey, the UK and the USA for their active contribution in the final agreement of these recommendations.
This work was supported by a grant NR/8852-3/2006 from the Ministry of Health of the Czech Republic.
