Abstract
Objective
Previous studies have not assessed whether evidence-based information about the outcomes of colorectal cancer screening increases informed choice among people from a range of socioeconomic backgrounds nor have they assessed whether this can be administered away from a health-care provider.
Methods
Randomized controlled trial in six primary care locations. Three hundred and fourteen people aged 50–74 years received a self-administered decision aid (DA) booklet about outcomes of biennial faecal occult blood testing (FOBT) screening or government consumer guidelines (G).
Results
Significantly more DA recipients (20.9%) were ‘informed’ compared with G recipients (5.8%) (P = 0.0001, OR 4.32; 95% CI 2.49 to 7.52); the DA did not affect values clarity (61.9% clear after DA versus 59.1% after G) nor screening decisions overall (87.3% would screen after DA versus 90.5% after G). Test uptake at one month was uniformly low (5.2% DA versus 6.6% G); mostly due to being ‘too busy’. DA recipients were more likely to make decisions ‘integrating’ knowledge with values (10.4% DA versus 1.5% G). Decisions not to screen were equally uncommon in both groups but more likely to be uninformed in G (P = 0.03). More DA recipients from all education levels were ‘informed’ (P = 0.02), particularly in lower education (50.0% DA versus 17.8% G) and university-educated groups (79.4% DA versus 32.1% G).
Conclusion
Detailed absolute risk and benefit information about FOBT screening can be effectively used at home by people to increase informed choice. The DA was effective in people with lower education levels.
Trial Registration
Unique Protocol ID 211705 ClinicalTrials.gov ID NCT 00148226.
INTRODUCTION
Colorectal cancer is the second most common cause of cancer death in the UK and Canada and the most common in Australia. 1–3 Biennial screening using faecal occult blood testing (FOBT) reduces colorectal cancer mortality 4 with the UK, Canadian and Australian Governments commencing population-based screening programmes in 2006–2007. Although colorectal cancer is common, there is a low level of knowledge about screening in these communities. 5 In the UK and Australia, kits are mailed directly to people at home while in Canada they can be obtained from a range of health-care providers. Thus, unlike existing breast and cervical cancer screening programmes, colorectal cancer screening is usually done at home, referral by a doctor is not needed, and screening may be done without even any discussion with a health-care provider. These programmes are being implemented at a time of increasing patient involvement in health-care decisions, 6–8 and there has been speculation about the potential effect of individual-level informed choice on participation rates in population screening programmes. 6–9
Decision aids (DA) are effective for increasing patient involvement in decisions 10 and their potential role in screening decisions well described. 11,12 Three randomized controlled trials (RCTs) of colorectal cancer screening DAs have shown a variable effect on ‘intention to screen’ but these DAs have not been completely self-administered away from health-care providers. 13–15 Furthermore, it is important to ensure, where possible, that health inequalities are not increased. Previous screening DA trials have not reported effects in a representative sample from the general population, particularly across different levels of education. If they are effective among groups from a range of educational backgrounds they have the potential to improve health literacy.
This study was set-up to test the effect of a self-administered DA (compatible with ‘at-home’ testing) on informed choice in participants from a range of educational backgrounds, and to assesses whether their decisions are consistent with values about screening (i.e. ‘integrated’ decision-making). The study is highly relevant to national colorectal cancer screening programmes as it is the first to explore the effect of individual-level informed choice on screening uptake in a representative sample of eligible ‘screenees’ in the community.
METHODS
Recruitment of primary care practices
Sixty-nine family practitioners had expressed interest in this study during an earlier survey of 107 randomly selected New South Wales clinicians. From these, one rural and five urban practices were recruited across the range of Australian Bureau of Statistical area-based indices of socioeconomic disadvantage scores 16 and Australian Government Index of Rurality and Remoteness scores. 17
Participant eligibility and recruitment
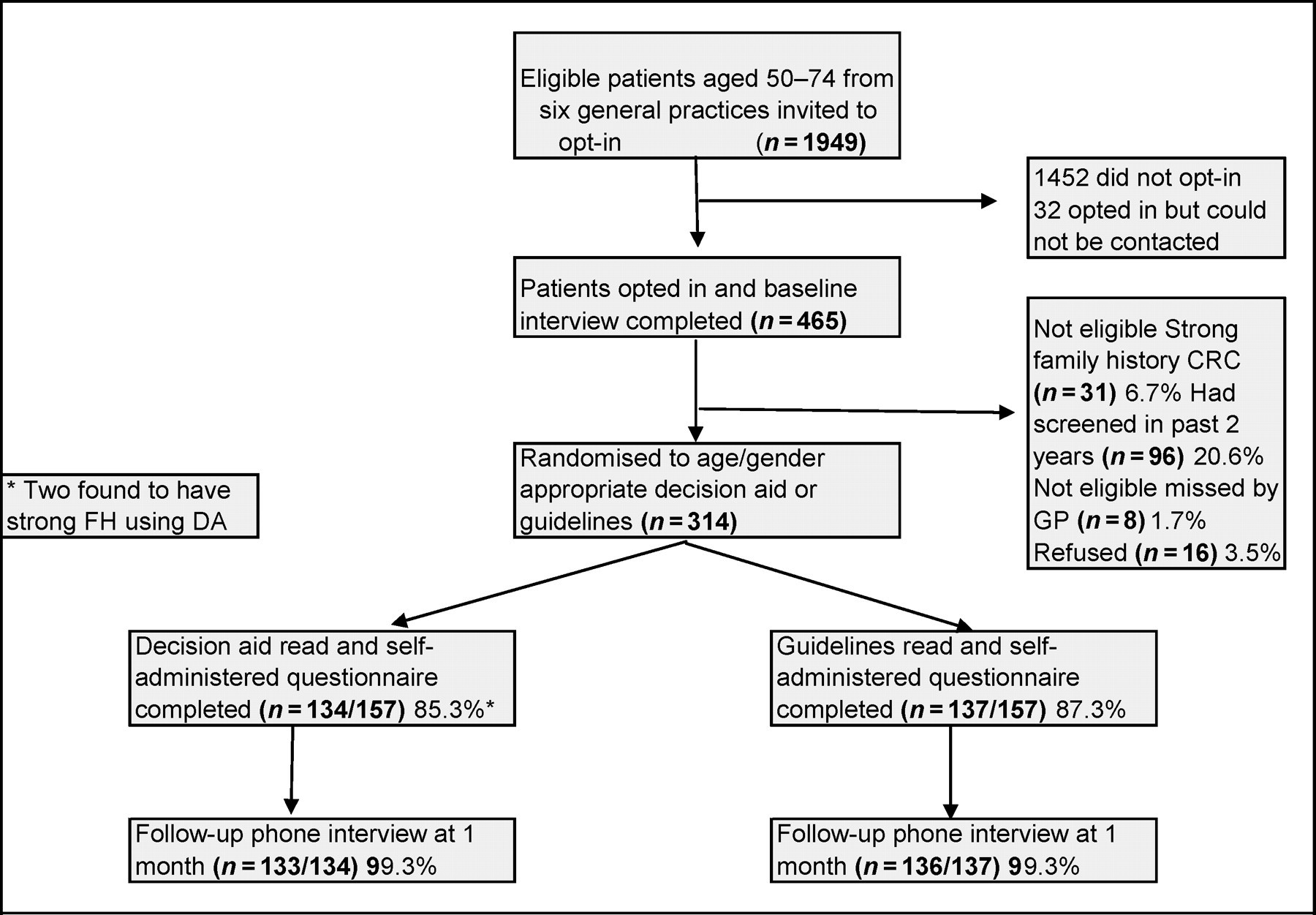
Practices generated a list of patients between the ages of 50 and 74 years from their databases, excluding people known to have poor English, significant cognitive impairment (e.g. dementia), serious physical or mental illness, residents of nursing homes and people with a personal history of colorectal cancer. A letter of invitation was sent on practice letterhead to eligible patients advising them of the study and they were contacted and interviewed by telephone if they returned a reply paid card, contacted a free telephone number or emailed the researchers. 18 This opt-in design was required by the University of Sydney Human Research Ethics Committee. Some people were further excluded from the study at the point of baseline interview because they had completed an FOBT, sigmoidoscopy or colonoscopy in the previous two years or because they had a strong family history of colorectal cancer and should be considered for colonoscopy screening under Australian guidelines. 19 After completion of the baseline telephone interview, participants were randomized to receive by post, the age-gender-family history specific DA or the Australian Government's consumer guidelines on FOBT screening along with a self-administered questionnaire.
Intervention: DA booklet
Six versions of the DA booklet were produced, each containing age, gender and family-history-specific probabilities of the outcomes of biennial FOBT screening over five screening rounds. The DA had a theoretical framework based on the Theory of Planned Behaviour and incorporated research-derived expert and lay beliefs about core issues for informed choice about FOBT.
11,20–24
The five core issues addressed in the DA were:
Definition of screening (i.e. for asymptomatic, well people). Baseline probability/personal susceptibility/risk factors for colorectal cancer over next 10 years. Absolute reduction in colorectal cancer mortality with screening over next 10 years. Probability of test outcomes from biennial FOBT over 10 years (the cumulative risk of screen-detected colorectal cancer, interval cancer, false-positives and large polyp detection follow-up colonoscopy). Information about how to collect a FOBT.
Information was presented according to best evidence using words and 1000-face diagrams. 12 The booklet was 20 pages long and the Flesch-Kincaid readability score was Grade 10. Examples of decisions were included and a personal worksheet required participants to indicate what was important to them.
Participants were mailed the relevant booklet for their age and gender and a questionnaire which they were asked to complete and return. An FOBT kit was not included with the package but information was provided about how to obtain one. Participants were contacted by telephone one month later and screening uptake was recorded.
Comparison: guidelines
The consumer version of Australian guidelines (at the time of this trial), was mailed to the control participants along with the self-administered questionnaire. 25 The consumer guidelines consisted of three pages of text with information recommending biennial FOBT for people over the age of 50, to reduce mortality from colorectal cancer. It contained very minimal information about false-positives and none about the risks of follow-up colonoscopy testing. There was no quantification of outcomes apart from an unreferenced statement that up to 80–90% of colorectal cancer could be detected through FOBT screening. The Flesch-Kincaid readability score was Grade 9. There were no graphs or pictures and no personal worksheet or examples.
Primary outcomes: informed choice and integrated decisions
Informed choice and integrated decisions were the primary outcome and were defined by three measures; 1) Adequate knowledge, 2) Clear values and 3) Screening intention (decision). 26
Knowledge was measured by coding responses to four open-ended questions about key domains of FOBT knowledge which were derived from research. 22–24
(1) Having read the information kit, can you please write down what you understand is meant by ‘screening for bowel cancer’ (2) What do you now believe are the main factors that affect your risk of bowel cancer? (3) In view of what you now know about bowel cancer, what would you say are the main factors in favour of having FOBT screening? and (4) In view of what you now know about bowel cancer, what would you say are the main factors against having FOBT screening? A question scored a maximum of ‘3’ if three or more pieces of information were included in a response, ‘2’ for two included and ‘1’ if only one was mentioned. One mark was deducted for each incorrect piece of information. Participants were deemed to have ‘adequate knowledge’ if they had a positive score for understanding the potential benefits and for understanding the potential harms of screening. A priori this was deemed to be the minimum level of knowledge for making an informed decision. (Appendix 1 for marking criteria). Values were deemed to be ‘clear’ if a score of 25 or less was obtained on the ‘values subscale’ of the Decisional conflict scale. This is the accepted cut-off point for a person being clear about their personal values using this validated scale. 27,28 Intention to screen was deemed to be a proxy for participants' screening decision. Participants were considered ‘informed’ if they had adequate knowledge according to the above criteria. They made an ‘integrated decision’ if values were clear and they had adequate knowledge. 29
Secondary outcomes – test uptake, DA acceptability and psychological outcomes
Self-reported FOBT uptake was assessed via telephone one month after completion of the DA or Guidelines and self-administered questionnaire. The Ottawa acceptability scale, 30 short-form state anxiety scale, 31 full decisional conflict scale 28 and self-efficacy scale 32 were all used to measure psychological outcomes of decision-making immediately after reading the booklet. Participants were not supplied with an FOBT but given information about where they might obtain one.
Sample size
The primary outcome for this study was the proportion of people who made an ‘integrated’ decision, a new outcome measure derived for this study. The Cochrane review states that on average knowledge scores are 20% higher in DA groups and decisional conflict is 10% lower. Sample size was estimated on a 15% increase in our composite outcome which includes increased knowledge and reduced decisional conflict. The sample size estimate was therefore 364 participants although a sample of 206 should be sufficient to detect a 20% change in knowledge consistent with informed choice.
Randomization and blinding
We aimed to recruit up to 10 people from six age-gender strata in each practice, thereby ensuring that we had adequate numbers of people assessing the age-gender and family history-specific versions of the DAs. Sequential ID numbers were randomly assigned by computer program to DA or Guidelines (G) in blocks of four. Allocation was concealed via the password-protected program. ID numbers were independent of allocation and the subjects were kept blind to the study hypothesis. Questionnaires were self-administered. Researchers were blinded to allocation for all telephone interviews. We noted that the small strata size was affecting the block randomization part-way through the first recruitment phase and changed to randomization by blocks of two for the remainder of the study.
Statistical methods
The proportion of people who made an ‘integrated decision’ was cross-tabulated for allocation to DA or Guidelines and chi-squared test of significance applied. Knowledge, values clarity and decision variables were all dichotomized using the criteria outlined above. To further explore the relationship between education level, and having adequate knowledge (i.e. being ‘informed’), logistic regression was used to test for effect modification by education. Similarly, to explore the relationship between absolute colorectal cancer risk and screening decisions (i.e. intention to screen) logistic regression modelling was undertaken.
RESULTS
Participant characteristics
From the six participating primary care practices, 465 out of 1949 invited people aged 50–74 years opted-in to be contacted by researchers and 314 were eligible and randomized. Excluded patients comprised 31 who were identified as having a strong family history and were referred for discussion about colonoscopy screening under Australian guidelines; 96 who had had FOBT or colonoscopy during the preceding two years; eight who did not meet eligibility criteria such as English language skills and 16 who refused to participate after receiving further study information.
Characteristics of sample at baseline (n = 314)
Effect of DA on screening decision
Integrated decision, adequate knowledge, clear values, intention to screen and acceptability: decision aid (DA) versus guidelines (n = 271)
Secondary outcomes – Test uptake, DA acceptability and psychological outcomes
After one month the proportion of people who had completed FOBT was uniformly low (5.2% DA versus 6.6% G; P = 0.64). ‘Being ‘too busy’ was the most common reason for not having the test (23.6% respondents). ‘Needing further information’ (9.5% in G versus 3.2% in DA) and ‘not having any bowel symptoms’ (16.7% in G versus 10.5% in DA) were more common reasons for delaying testing in the Guidelines group compared with DA.
There was no statistically significant difference in ‘satisfaction with decision (P = 0.56), Anxiety (P = 0.59), perceived behavioural control (P = 0.63) nor decision control preference (P = 0.38).
The DA was more likely to be perceived as being balanced and fair with respect to FOBT compared with the guideline document (χ 2 = 18.09 df 4, P = 0.001). In addition to being perceived as balanced, the decision was more likely to be assessed as containing about the right amount of information compared with the guideline document. (χ 2 = 17.07, df 4, P = 0.002). Participants were also more likely to consider that the length of the DA was about right compared with those who received the guidelines (χ 2 = 52.13, df 4, P = 0.0001). While the amount and length of the DA was rated more positively and it was more likely to be perceived as balanced and fair, there were not significant differences in ratings of clarity of information (P = 0.45) helpfulness of the information kits (P = 0.30) or whether recipients would recommend it to others (P = 0.78). People generally thought that the information was clear regardless of whether they received the DA or guidelines, both formats were thought to be helpful and both would be recommended by the majority of readers.
Interaction by absolute colorectal cancer risk
Although it is plausible that screening intention may be influenced by absolute risk of disease, there was no evidence of interaction between risk and the effect of the DA on intention to screen (OR = 0.22 95% CI 0.03–1.86, Hosmer Lemeshow goodness-of-fit P = 0.66).
Interaction by educational status
Decision aid (DA) effect on informed choice by education level
INTERPRETATION
Principal findings
This study has shown that both informed choice and integrated decision-making for colorectal cancer screening can be significantly improved by a self-administered DA booklet without affecting screening participation rates. This effect was observed across all education groups.
Strengths and weaknesses
One of the strengths of this study is that it was deliberately designed to reflect the context of colorectal cancer screening programmes. The intervention was self-administered at home in a similar manner to the postal recruitment and delivery of the FOBT information in some national programmes. In many countries, people can also purchase FOBT kits directly and use them ‘at home’. This design is novel in that it evaluates the use of a DA in a setting without a health-care provider – a necessary context given the frequent ‘at-home’ delivery method of FOBT screening. An additional strength of this study is the RCT design with good quality randomization, equivalent groups at baseline and high follow-up rates. Low recruitment rates were unfortunate but unavoidable given ethical requirements. We have found that opt-in recruitment is less likely to recruit people from lower education and non-screening backgrounds. 18 As such, our results may be an underestimate of the effect when generalized to the wider community.
Another limitation of this study is that screening uptake was measured at one month and this may have been too early. The Australian colorectal cancer screening pilot report has subsequently shown that at four weeks, uptake was only 25% but this rose to approximately 45% by 12–14 weeks. 33 In addition, we did not provide the FOBT kit with the intervention, but only referred to other sources for obtaining this.
Strengths and weaknesses in comparison with other studies
We note that even after the DA, only 20% of people were adequately informed. Of the three previous trials of colorectal cancer screening DAs, 13,15,34 only one measured knowledge as an outcome. 34 Unfortunately, their ‘10-item scale’ was not described in the published paper. Our marking criteria were stringent yet based on educational principles of assessing competence. It is difficult therefore to compare our participants' knowledge levels with other studies. The fact that only 5% of the control group, after reading the guidelines, had adequate knowledge suggests however, that community knowledge levels about FOBT screening are exceedingly low, unlike breast and cervical cancer screening which has been in place for many years.
This study was not powered to detect interaction for absolute colorectal cancer risk on screening decisions but it appears that colorectal cancer risk is unlikely to substantially effect screening decisions, despite the presentation of personalized age-gender-family history specific screening outcomes. Baseline ‘intention to screen’ was high at around 85–90% and this remained so after the intervention which included personalized risk information. This may suggest that screening decisions are overwhelmingly driven by the belief that ‘screening is a good thing’ and that ‘prevention is better than cure’. Nevertheless, this study has shown that informed individuals, given absolute risk-specific information, in the main will opt to screen. It goes some way towards dispelling the concern that such information will be harmful to people despite, one might argue, the ethical and moral obligation to make good quality information available to all. Other studies have not explored the effect of risk-specific information on screening rates, but have tended to treat the population as one group.
Implications for policy-makers and future directions
Population-based screening programmes need to look carefully not only at minimising the practical barriers to screening but also to the quality of the information they provide in order to minimize dangerous misconceptions caused by a lack of information. Our study found that 95% of people receiving standard information made uninformed decisions to screen and of the small proportion who decided against screening, guideline recipients were more likely to have made this as an uninformed decision.
Almost half of our study sample had left school by the age of 16 years and the DA, despite its relatively high reading age and level of complexity was more likely to increase informed choice in this group than the guidelines, where the majority of people remained uninformed. It may be that the inclusion of graphics and the interactive nature of the DA were useful for people with lower education. Other authors have suggested this might be the case. 35 Thus, the assumption that complex information about screening outcomes is too difficult for many people to understand may be ill-founded. The exclusion of such information from many general population brochures such as the consumer Guidelines may be an injustice to the community.
The methods used to measure ‘informed choice’ and ‘integrated decisions’ are robust and should be further developed to assess the implementation of informed choice policies within a range of community and health settings.
This study therefore has practical implications for current colorectal cancer screening programmes and provides a challenge to these programmes to implement DAs within their programmes. This is clearly an effective tool for facilitating informed choice for colorectal cancer screening. The next challenge is to identify how this evidence can most effectively be put into practice.
Funding
National Health and Medical Research Council (NHMRC) Program Grant.
Ethics approval
Granted by University of Sydney Human Research Ethics Committee.
Clinical Trial Registration Details
Unique Protocol ID 211705, ClinicalTrials.gov ID NCT 00148226.
Excerpts from decision aid Flowchart of randomised controlled trial
Footnotes
ACKNOWLEDGEMENTS
This study was funded by a National Health and Medical Research Council Program Grant.
Thanks to Anna Isaacs and Peta Pippos who worked as research assistants on this project. Also thanks to Petra MacCaskill for her comments on the statistical analysis and study design. Dr Trevena designed the study protocol, developed the model for the DA, wrote the DA content, analysed the data and wrote the first draft of this paper. Professor Irwig provided feedback on the design of the DA and the estimates of outcomes of screening contained within. He provided input to the study design, particularly the stratification and randomization and reviewed and commented on drafts of this paper. A/Prof Barratt provided feedback during the design of the DA and the randomized trial, and reviewed and commented on drafts of this paper.
