Abstract
Objectives
To see whether revised screening standards and health-professional training are associated with changes in the performance of a neonatal screening programme for congenital hypothyroidism (CHT).
Methods
Screening data from the regional screening service in Durham and Newcastle, which covers north-east England and North Cumbria.
Setting
We assessed the timing of the different stages of the screening process leading up to the introduction of the revised guidelines between April 2004 and March 2005 (year 1) and afterwards between April 2005 and March 2006 (year 2) in all babies notified as having CHT. We also assessed the interval between sampling and specimen arrival in the laboratory at the beginning and end of year 2 in all babies screened.
Results
Twenty-three babies tested positive or borderline in year 1 and 18 babies in year 2. There was reduced variability in the overall time from birth to notification in year 2 versus year 1 (P = 0.001). This reduction was a consequence of a reduced interval between sample collection and arrival in the laboratory (P = 0.047) and for the laboratory to notify the positive test result (P = 0.003). There was a reduction in the mean time from sampling to receipt by the laboratory in the 2997 babies screened in the final month compared with the 2498 babies screened in the first month of year 2 (P = 0.01).
Conclusion
There was an improvement in neonatal screening programme performance around the time that revised neonatal screening guidelines were introduced. This highlights the importance of ongoing education and training for those involved in screening programmes.
Introduction
The UK Newborn Screening Programme Centre published revised standards in April 2005. 5 The earlier guidelines suggested that 100% of positive cases should start treatment by 35 days of life (95% by 21 days) 1 and the revised version was intended to lead to improved timing of blood-spot collection, sample arrival in the laboratory and notification of raised TSH values to the nominated paediatrician so that all babies with CHT are treated by 21 days of age. It is of note that the Working Group for Neonatal Screening in Paediatric Endocrinology of the European Society for Paediatric Endocrinology has suggested that treatment should commence before 15 days of life. 6
Temporal changes in screening programme performance have been reported in the UK 7 and our aim was to see whether there was any evidence of altered screening performance around the time that the revised guidelines were published.
Subjects and methods
The study was discussed with and approved by the Local Research Ethics Committee.
Around 31,000 infants in the north-east of England and North Cumbria are screened every year in a single centre by measuring blood spot TSH levels. The newborn screening centre was based in Durham from 1996 but moved to Newcastle in April 2005.
Health professional training
Although the revised UK Newborn Screening standards were published in April 2005, the groundwork for implementing the new standards in north-east England started at least six months earlier. The Regional coordinator for Antenatal and Child Health in the northern region of England and the Regional Educator met local coordinators on a regular basis to discuss the steps needed to implement the new standards. From October 2004, the local coordinators of the screening programme (including antenatal, postnatal, midwifery, community and neonatal coordinators) conducted training sessions with the midwives and nurses involved with blood spot sample collection. The emphasis was on improving the timing of the blood spot test and ensuring that samples were dispatched as soon as possible. Materials used to educate staff include compact discs, newsletters, formal training sessions and emails.
TSH blood spot assay
Blood spot TSH was measured throughout the study period by solid phase two-site fluoroimmunometric immunoassay (DELFIA, Perkin-Elmer, Waltham, USA). The inter-assay coefficient of variations (CVs) for the TSH assay in Durham were 9.5 and 10.1% at TSH values of 15.6 and 59.8 mU/L, respectively, and in the Newcastle laboratory were 11 and 12% at TSH values of 16 and 60 mU/L, respectively. Positive and borderline cases were defined as a blood spot TSH > 6 mU/L throughout the study period. The cut-off of 6 mU/L has not changed during the entire period of the study.
Data collection
Data were collected from records kept in the biochemistry departments at Durham (year 1 between April 2004 and March 2005) and Newcastle (year 2 between April 2005 and March 2006). We compared data on all positive and borderline cases (blood spot TSH >6 mU/L) in year 1 leading up to the introduction of the new guidelines and in year 2 following their publication. We looked at (1) the interval between birth and age at sampling; (2) the interval between sampling and sample receipt by the laboratory; (3) the interval between sample receipt by the laboratory and notification of positive and borderline results; (4) the overall time from sampling to notification and (5) the time from birth to notification. In borderline cases, the date that a second sample was requested (by fax or phone-call and letter) was taken as the notification date. We intended to exclude all infants born preterm (<37 weeks) because of the reported association between pre-term delivery and hypothyroxinaemia and either normal or raised TSH levels. 8
The interval between sampling and sample receipt by the laboratory was also determined in all babies screened (those with a blood spot TSH <6 and >6 mU/L) in the first and twelfth months following the introduction of the new guidelines. We compared these months because they were at the beginning and end of the period following guideline introduction, because they were at a similar time of year and because neither month included the Easter Bank Holiday period. Unfortunately, similar data was not available for the 12-month period leading up to the introduction of the screening guidelines.
Birth weight, gestational age and number of CHT cases in year 1 (April 2004 to March 2005) and year 2 (April 2005 to March 2006)
Twenty-four babies tested positive or borderline in year 1 and 20 babies in year 2. There were 31,259 births in the region in year 1 and 31,818 births in the region in year 2. This suggests an incidence of CHT of 71 and then 64 per 100,000 live births for the first and second years, respectively. One baby in year 1 was excluded because no data were available for the first blood-spot card submitted, which had yielded a borderline result. One baby who tested positive in year 2 was excluded on the basis of prematurity and a second because data had been entered incorrectly. Data were therefore analysed for 23 babies in year 1 and 18 in year 2. The mean gestational age at birth in years 1 and 2 was 39.4 ± 1.6 weeks (range 37-41) and 39.6 ± 1.8 weeks (range 37-42), respectively. The mean birth weight in years 1 and 2 was 3273 ± 485 g (range 2620-4240) and 3035 ± 405 g (range 2090-3690), respectively.
Statistics
Data were entered into a statistical package (MINITAB, Coventry, UK) and expressed as mean time (days) ±1 SD. Means were compared using a two-sample t-test and variance using the F-test, which is used to determine whether the variances of two groups are equal.
Results
Screening performance in year 1 (April 2004 to March 2005) and year 2 (April 2005 to 2006)
Time interval (mean ± SD) between different sections of the screening process in years 1 and 2
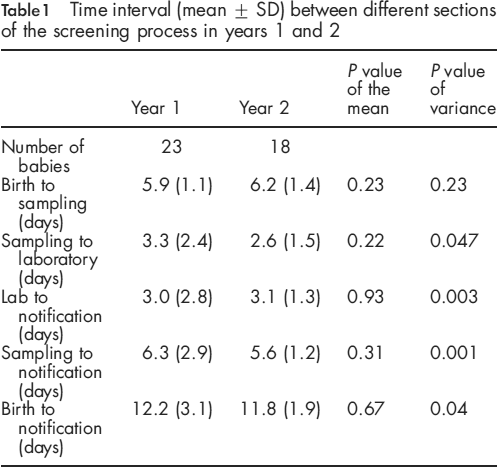
Significantly reduced variability was noted in the time taken from birth to notification (P = 0.04) and from sampling to notification (P = 0.001) in year 2 versus year 1. This reflected a reduced variance both for the time taken between sampling and receipt by the laboratory (P = 0.05) and for the laboratory to notify a positive or borderline result (P = 0.003) (Table 1).
In year 1, the screening programme failed to meet the revised standard with one out of 23 notified after 21 days. In year 2, all were notified before 21 days. In year 1 and year 2, the screening programme failed to meet the European Working Group criterion, which suggested that treatment should commence by day 15. Five out of 23 were notified after 15 days in year 1 and one out of 18 babies in year 2.
In light of these results, we also examined the difference in mean time to notification and variability in time from birth and sampling to notification in 2003-2004 (the 12-month period leading up to year 1) to see if there was evidence of a similar change in programme performance prior to 2004-2005. There was no difference in mean time from birth to notification (P = 0.9) and from sampling to notification (P = 0.7) between these two years. There was no difference in variability from birth to notification (P = 0.143) or sampling to notification (P = 0.2) either.
Screening performance in first and twelfth months of year 2
To further understand the influence of the new guidelines, we determined the interval between blood spot collection and sample receipt by the laboratory for all babies screened in the year following the introduction of the new guidelines. A total of 2498 babies were screened in the first month (April 2005) and 2997 in the final month (March 2006). The mean time from sampling to receipt by the laboratory was 3.0 ± 1.8 days (range 1-17) in the first month and 2.9 ± 1.7 days (range 1-18) in the twelfth month (P = 0.01).
Discussion
The neuro-developmental outcome of babies born with CHT will be influenced by the timing of treatment with thyroxine 9 although whether an improved outcome will follow from even earlier treatment initiation than that associated with well-established treatment programmes is unclear.10–13 A particular concern for many health professionals is the avoidance of late treatment and one of the key objectives of the revised UK screening standards was to ensure timely sample collection. 5 The revised guidelines were, nevertheless, introduced a number of years after a European Working Group had suggested that treatment should commence by day 15. 6
We have found two patterns to suggest that this process may have had a beneficial impact on the performance of the screening programme. First, there was significantly reduced variability in the time taken to notify health professionals about a positive or borderline result (or request a second sample in borderline cases) following the publication of the new guidelines. This indicates that there were a reduced number of outliers and hence fewer babies being treated at a relatively late stage. A reduction in the number of outliers was not seen when we looked back beyond the 12 months leading up to the introduction of the new guidelines. Secondly, while the number of positive and borderline cases studied is small (CHT is not common) we found that the reduction in late notifications was matched by a small but significant (3-4%) reduction in the mean time from sampling to specimen receipt by the laboratory in the large number of screening ‘negatives’. The reason that the number of positive notifications was greater than 1 in every 3-3500 babies (the reported incidence of hypothyroidism) may reflect our own experience in the north-east and from elsewhere suggesting that CH is becoming more common. 14
We suspect that the trends that we have identified reflect a real change in screening programme performance. If so, then there are at least two potential explanations for them. First, there appears to have been a reduction in the number of late submissions to the laboratory following sample collection. This in turn could be due to changes in practice such as that of blood-spot card ‘batching’ by midwives. The reduction in the number of children treated relatively late could also reflect altered practice in terms of sample handling and analysis in the laboratory. At a local level this could also be linked to the fact that the screening laboratory moved sites. Some of the improvements identified relate to events prior to sample receipt by the laboratory and so this is unlikely to be the only factor.
A significant criticism of our study is that it was conducted over a relatively short period of time and involved a relatively small number of affected infants although changes in screening programme performance may well be transient. Our attempts to examine population data and compare the number of infants screened with the number of births were complicated by limitations in the way that data had been collected and stored at a time when computerized systems were developing. As an example of the problems encountered we found that the number of infants screened in 2004-2005 was 32,383 compared with a birth rate of 31,259 over the same time period. It will be easier to examine issues such as coverage and short- and longterm patterns as the data collection systems evolve. This is already apparent for the year 2005-2006, where feedback from the Child Health Record Department has suggested that coverage is close to 100%. We intend to conduct a longer term assessment of the programme in our region to establish whether the changes identified in screening programme performance persist.
In summary, our data suggest that the performance of the screening programme for CHT in the UK improved around the time that the new national newborn screening guidelines were introduced. We suspect that education programmes and an increased awareness of the new guidelines among the midwives and nurses responsible for the blood spot tests is primarily responsible for the changes described. Our study highlights the need to train staff and appraise the performance of a screening programme on a regular basis.
Footnotes
Acknowledgements
We would like to acknowledge the significant contributions to this work that have been made by Enid Avis, Research Associate, Department of Paediatric Endocrinology, Royal Victoria Infirmary, Newcastle upon Tyne, Mark Pearce, School of Clinical Medical Sciences (Child Health), Newcastle University, UK, Dr Sri Panditi Department of Paediatrics, Royal Victoria Infirmary, Newcastle Upon Tyne, Dr Hilary Wastell, Consultant Clinical Biochemist, Royal Victoria Infirmary and Dr David Allison, Department of Microbiology, University Hospital of North Durham, Durham, DH1 5TW. We would also like to thank Kim Moonlight, North-East Regional Antenatal and Child Health Screening Co-ordinator.
