Abstract
Background
It is difficult to non-invasively visualize changes in regional cerebral blood flow caused by manual compression of the carotid artery.
Purpose
To visualize dynamic changes in regional cerebral blood flow during and after manual compression of the carotid artery.
Material and Methods
Two healthy volunteers were recruited. Anatomic features and flow directions in the circle of Willis were evaluated with time-of-flight magnetic resonance angiography (MRA) and two-dimensional phase-contrast (2DPC) MRA, respectively. Regional cerebral blood flow was visualized with territorial arterial spin-labeling magnetic resonance imaging (TASL-MRI). TASL-MRI and 2DPC-MRA were performed in three states: at rest, during manual compression of the right carotid artery, and after decompression. In one volunteer, time-space labeling inversion pulse (Time-SLIP) MRA was performed to confirm collateral flow.
Results
During manual carotid compression, in one volunteer, the right thalamus changed to be fed only by the vertebrobasilar system, and the right basal ganglia changed to be fed by the left internal carotid artery. In the other volunteer, the right basal ganglia changed to be fed by the vertebrobasilar system. 2DPC-MRA showed that the flow direction changed in the right A1 segment of the anterior cerebral artery and the right posterior communicating artery. Perfusion patterns and flow directions recovered after decompression. Time-SLIP MRA showed pial vessels and dural collateral circulation when the right carotid artery was manually compressed.
Conclusion
Use of TASL-MRI and 2DPC-MRA was successful for non-invasive visualization of the dynamic changes in regional cerebral blood flow during and after manual carotid compression.
Keywords
Territorial arterial spin-labeling magnetic resonance imaging (TASL-MRI) is a non-invasive technique for visualizing the perfusion territories of individual brain-feeding arteries (1–3). In this imaging technique, water protons in the blood flowing through the brain-feeding arteries are magnetically labeled. A short time delay allows the magnetically labeled blood to travel to the brain tissue before image acquisition. Subsequent subtraction of this image from a control image in which there is no magnetic labeling shows the perfusion territory associated with a particular artery. Studies using TASL-MRI have described variations in the blood supply to brain structures from the left internal carotid artery, the right internal carotid artery, and the vertebrobasilar system in healthy subjects (1,4,5) and in patients with cerebrovascular disease (6–10).
Evaluating dynamic changes in the perfusion territories of intracranial arteries is very difficult. Few studies have performed these evaluations in either healthy or diseased subjects. Intracranial digital subtraction angiography (DSA) (11) and transcranial Doppler ultrasound (12) have been used to study dynamic changes in intracranial blood flow, but these techniques are incapable of evaluating the perfusion territories of the brain-feeding arteries. They do, however, allow the evaluation of blood flow within the large brain-feeding arteries. Nuclear medicine techniques such as brain single photon emission computerized tomography and positron-emission tomography are good methods for the evaluation of perfusion territories, but it is difficult to evaluate dynamic changes in intracranial blood perfusion without invasive temporary balloon occlusion.
TASL-MRI studies (1, 4–10) have so far been used to assess only intracranial perfusion territories at rest. However, we anticipate that TASL-MRI will be beneficial for evaluating dynamic changes in intracranial perfusion territories because of its use of magnetically labeled blood as a real-time, non-invasive tracer. Reversible changes in intracranial blood flow can be assessed by manual compression of the carotid artery; this is known as the Matas test. The Matas test is a safe clinical method of estimating tolerance to cervical carotid occlusion (13). To minimize side-effects, volunteers use their non-dominant hand to press the contralateral carotid artery.
The aim of our study was to visualize dynamic changes in the perfusion territories of individual brain-feeding arteries and the direction of blood flow in the circle of Willis during manual compression of the carotid artery, and after decompression, in healthy volunteers. To determine the direction of blood flow in the circle of Willis we used two consecutive two-dimensional phase-contrast magnetic resonance angiography (2DPC-MRA) assessments. To confirm the presence of collateral arteries we used time-spatial labeling inversion pulse MRA (Time-SLIP MRA). Time-SLIP MRA utilizes arterial spin-labeling (ASL) to provide a very detailed view of intracranial arteries and hemodynamic information. The data provided are similar to those from DSA, but the technique does not require a contrast agent and distal branches of the intracranial arteries can be clearly visualized (14). Time-SLIP MRA is a suitable alternative to DSA for proving the presence of collateral circulation during internal carotid artery occlusion (15). Because this was the first attempt to non-invasively visualize changes in regional cerebral blood flow caused by manual compression of the carotid artery and was therefore a pilot study, only two volunteers were recruited.
Material and Methods
Subjects
The ethics committee of the study institution approved the study protocol. Two healthy volunteers were recruited (a 30-year-old man and a 28-year-old woman). Changes in cerebral blood flow were caused by manual compression of the right carotid artery with the left hand. Both volunteers were right-handed. First, the anatomic features of the circle of Willis were evaluated with time-of-flight magnetic resonance angiography (TOF-MRA). Then the contributions to the perfusion territory were visualized with TASL-MRI. The direction of blood flow in the circle of Willis was determined by using 2DPC-MRA. TASL-MRI and 2DPC-MRA were performed at rest, during manual compression of the right carotid artery, and after decompression. In one volunteer, Time-SLIP MRA was performed to confirm collateral flow at rest, during manual compression of the right carotid artery, and after decompression.
Magnetic resonance imaging
All MRI except Time-SLIP MRA was performed by using a clinical 3 T MRI system (Achieva; Philips Medical Systems, Best, The Netherlands) with an eight-element phased array head coil. To show the perfusion territories of the internal carotid arteries and the basilar artery, territory-specific perfusion images were acquired with a pseudo-continuous selective ASL perfusion sequence, according to a previously published protocol (16). Time-SLIP MRA was performed with a clinical 1.5 T MRI system (EXCELART Vantage XGV; Toshiba Medical Systems, Tokyo, Japan) with a five-channel head coil.
The whole imaging protocol consisted of a scout image, a SENSE reference scan, TOF-MRA, TASL-MRI, and 2DPC-MRA. Images acquired from the TOF-MRA (TR/ TE = 22/4.6 ms; flip angle = 19°; FOV = 220 mm; recon matrix = 512 × 512; number of slices = 220; slice thickness = 1.5 mm; acquisition time = 2.6 min) were used for accurate planning of the labeling slabs for the TASL-MRI. TASL-MRI data were obtained by using a QUASAR sequence (17) with the following parameters (18): FOV = 240 mm; matrix = 64 × 64; 7 slices (8 mm, 1 mm gap); TR/TE = 4000/23 ms; flip angle = 35°; Til/ΔTI = 50/ 390 ms (10 time points); SENSE = 3, 96 averages (32 for each territory); acquisition time = 6.8 min. The labeling volume for the vascular territories was planned according to Hendrikse et al.’s protocol (2) on the basis of the maximum-intensity projections and native data from the TOF-MRA; the area covered spanned from the carotid bifurcation to the circle of Willis. Two consecutive 2DPC-MRA measurements, one phase-encoded in the anterior-posterior direction and one phase-encoded in the right-left direction, were conducted according to a previously published imaging protocol (19) with the following parameters: TR/TE = 9.4/ 5.9 ms; flip angle = 7.5°; FOV =250 mm; single slice, 8 averages; slice thickness = 13 mm; velocity sensitivity = 40 cm/s; acquisition time = 20 s.
Time-SLIP MRA (14) was conducted using the following parameters: TR/TE = 5.2/2.6 ms; flip angle = 120°; FOV = 280 mm; matrix = 448 × 448; delay time after IR pulse = 400-1400 ms (by 200 ms); partition thickness/gap = 2.5/ 0 mm; labeling slab = approximately 90 mm thick; acquisition time = 12 min.
TASL-MRI postprocessing
All TASL-MRI images were exported to a Windows PC running IDL 6.1 software (Research Systems Inc., Boulder, CO, USA). Labeled ASL images of the perfusion territories were first subtracted from unlabeled ASL images to produce ASL subtraction images of the different perfusion signals (one for each of the individual territories). These images were then combined into a red-green-blue frame to demonstrate the spatial distribution of the three perfusion territories (2). The left internal carotid artery (ICA) territory was visualized in green, the right ICA territory in red, and the vertebrobasilar system in blue. Areas demonstrating a mixing of perfusion from more than one vessel were visualized as combined colors. For example, perfusion contributed to by the right ICA and the vertebrobasilar system would be visualized as purple (red + blue). The color images from the multiple inversion time points were presented to two readers (KA and HM, with 4 and 14 years’ experience in neurovascular MR imaging, respectively). Dynamic variables included time of arrival and bolus duration, both of which can differ between regions because of collateral perfusion. The visual evaluations given by the two readers for the perfusion distribution were in agreement. Territorial ASL images from the study are shown at a single inversion time of 1610 ms because of the high signal-to-noise ratio of this relatively short inversion time compared with those of longer inversion times. The possibility of non-uniform compression was excluded by comparing the upper and lower slices and the other inversion time ASL images.
Perfusion territory and collateral blood flow assessment
The TOF-MRA images, TASL-MRI territory distributions, and 2DPC-MRA images were visually evaluated independently by two readers (KA and HM). By using colored TASL-MRI images, the perfusion of the thalamus, basal ganglia, cerebrum, and cerebral sphere were evaluated, and by using 2DPC-MRA images, the direction of blood flow in the circle of Willis was evaluated. One reader (KA) assessed the Time-SLIP MRA with multiple inversion times for the visualization of vessels, and in particular the visualization of contralateral flows.
Results
The two readers agreed on the anatomical features of the circle of Willis shown by the TOF-MRA, the direction of blood flow in the circle of Willis shown by the 2DPC-MRA, and the territory distributions shown by TASL-MRL
TOF-MRA showed that the first volunteer (Subject 1) had a normal circle of Willis (Al segments of the anterior cerebral artery, anterior communicating artery [A-COM], posterior communicating arteries [P-COMs], and P1 segments of the posterior cerebral artery [PCA] were visualized) (Fig. 1, upper panels). The second volunteer (Subject 2) had a well-formed left Al segment, a narrow right A1 segment, and hypoplastic P-COMs bilaterally (Fig. 1, lower panels).
Upper panels: Subject 1, TOF-MRA; normal circle of Willis. TOF-MRA showed that Subject 1 had a normal circle of Willis (Al segments of the anterior cerebral artery, anterior communicating artery [A-COM], posterior communicating arteries [P-COMs], and P1 segments of the posterior cerebral artery [PCA] were visualized); Lower panels: Subject 2, TOF-MRA. Subject 2 had a well-formed left Al segment, a narrow right A1 segment, and hypoplastic P-COMs bilaterally
TASL-MRI images showed that, in Subject 1 in the resting state, the left thalamus, cerebrum, and both occipital lobes were fed by the vertebrobasilar system, whereas the right thalamus was fed mostly by the vertebrobasilar system and partly by the left carotid artery. The basal ganglia, but not the right head of the caudate nucleus, were fed by the ipsilateral carotid artery. The right head of the caudate nucleus was fed by the left carotid artery. Each cerebral sphere, but not the occipital lobe, was also fed by the ipsilateral carotid artery (Fig. 2, upper panels). In Subject 2, on both sides, the thalamus, cerebrum, and occipital lobe were fed by the vertebrobasilar system. The basal ganglia were fed by the ipsilateral carotid artery. The cerebral spheres, but not the occipital lobes, were also fed by the ipsilateral carotid artery (Fig. 3, upper panels). In Subject 1, 2DPC-MRA showed bilateral posterior-to-anterior flow through the Al segment and bilateral anterior-to-posterior flow through the P-COMs (Fig. 4, left upper panels). In Subject 2, 2DPC-MRA showed posterior-to-anterior flow through the well-formed left Al segment. Flow was not visualized in the narrow right A1 segment or in the hypoplastic P-COMs (Fig. 4, right upper panels).
Subject 1, TASL-MRI. Upper panels: at rest - the right thalamus was fed mostly by the vertebrobasilar system and partly by the left carotid artery. The right head of the caudate nucleus was fed by the left carotid artery. The left thalamus, cerebrum, and both lateral occipital lobes were fed by the vertebrobasilar system. The basal ganglia, but not the right head of the caudate nucleus, were fed by the ipsilateral carotid artery. The cerebral spheres, but not the occipital lobe, were fed by the ipsilateral carotid artery. Middle panels: during compression - the right thalamus was fed only by the vertebrobasilar system. The right head of the caudate nucleus continued to be fed by the left carotid artery, and the right basal ganglia were fed only by the vertebrobasilar system. The area fed by the right carotid artery at rest was fed mostly by the vertebrobasilar system, and part of the frontal lobe was fed by the left carotid artery. Lower panels: after decompression - the distribution of the perfusion territory recovered to the resting state after decompression
Subject 2, TASL-MRI. Upper panels: at rest - on both sides, the tlnalamus, cerebrum, and occipital lobe were fed by the vertebrobasilar system. The basal ganglia were fed by the ipsilateral carotid artery. The cerebral spheres, but not the occipital lobe, were also fed by the ipsilateral carotid artery. Middle panels: during compression - the right thalamus continued to be fed only by the vertebrobasilar system. The right basal ganglia were fed by the vertebrobasilar system. The original perfusion territory of the right carotid artery was fed mostly by the vertebrobasilar system. Lower panels: after decompression - the distribution of the perfusion territory recovered to the resting state after decompression
Left: Subject 1, 2DPC-MRA; Right: Subject 2, 2DPC-MRA; Left side: anterior-posterior direction; Right side: right-left direction; Upper panels: at rest; Middle panels: during compression; Lower panels: after decompression. Subject 1 - at rest, 2DPC-MRA showed bilateral posterior-to-anterior flow through the Al segment and bilateral anterior-to-posterior flow through the P-COMs. During compression, 2DPC-MRA showed anterior-to-posterior flow through the right Al segment and posterior-to-anterior flow through the right P-COM. After decompression, the directions of blood flow recovered to those in the resting state. Subject 2 - at rest, 2DPC-MRA showed posterior-to-anterior flow through the well-formed left Al segment. Flow was not visualized in the narrow right A1 segment or in the hypoplastic P-COMs. During compression, 2DPC-MRA showed no flow in the right Al segment or in either P-COM, as in the resting state. Flow through the left Al segment did not change. After decompression, the directions of blood flow recovered to those in the resting state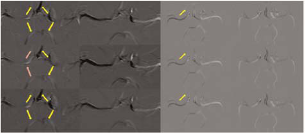
During compression, in Subject 1, the right thalamus was fed only by the vertebrobasilar system, and the right basal ganglia (with the exception of the right head of the caudate nucleus) were fed by the vertebrobasilar system. The right head of the caudate nucleus continued to be fed by the left carotid artery,
The original perfusion territory of the right carotid artery was fed mostly by the vertebrobasilar system, and part of the frontal lobe was fed by the left carotid artery (Fig. 2, middle panels). In Subject 2, the right thalamus continued to be fed only by the vertebrobasilar system. The right basal ganglia were fed by the vertebrobasilar system. The original perfusion territory of the right carotid artery was fed mostly by the vertebrobasilar system (Fig. 3, middle panels). In Subject 1, 2DPC-MRA showed anterior-to-posterior flow through the right Al segment and posterior-to-anterior flow through the PCA (Fig. 4, left middle panels). In Subject 2, the narrow right A1 segment and hypoplastic P-COMs continued to have no blood flow, and flow through the left Al segment did not change (Fig. 4, right middle panels).
After decompression, the distributions and directions of blood flow immediately recovered to their resting states (Figs. 2–4, lower).
Time-SLIP MRA was performed on one volunteer (Subject 1). The inversion time when contralateral flows were best visualized was 1200 ms. Vessels from the posterior circulation were extended and well-defined. Small vessels extending downward from the parietal portion were visualized. These small vessels appeared to have anastomosed with the middle cerebral artery. Compression of the right carotid artery revealed pial vessels and dural collateral circulation. This suggests that the top portion of the cerebral lobe was fed by the contralateral carotid artery, whereas the bottom portion was fed by the posterior circulation (Fig. 5).
Time-SLIP MRA. Left panels: 600 ms; Right panels: 1200 ms; Upper panels: at rest; Lower panels: compression. With manual right carotid artery compression, pial vessels and dural arteriolar vessels anastomosing with cortical vessels were visualized. Red arrows: from the contralateral carotid artery; yellow arrows: from the posterior circulation
Discussion
In Subject 1, TASL-MRI showed that during manual compression of the carotid artery the right thalamus was fed only by the vertebrobasilar system, and the right basal ganglia were fed by the left carotid artery. In Subject 2, blood flow to the right thalamus did not change and it continued to be fed only by the vertebrobasilar system. The right basal ganglia were fed by the vertebrobasilar system. Studies conducted using TASL-MRI have shown that there are individual variations in the distribution of the perfusion in the basal ganglia and thalami (5,9). In particular, the distribution of cerebral blood flow in the basal ganglia and thalami in patients with carotid artery stenosis is different from that in healthy controls (9). This suggests variability in the perfusion territories of the deep-brain structures and the importance of collateral circulation.
In Subject 1, the direction of blood flow changed from anterior to posterior through the right Al segment, and from posterior to anterior through the right PCA. In the majority of patients with unilateral ICA occlusion, the Al segment shows retrograde flow (19). This is consistent with previous reports that have also demonstrated reversibility. In Subject 2, although the right P-COM was poorly developed, when the carotid artery was compressed the original perfusion territory of the right carotid artery was fed mostly by the vertebrobasilar system. It is thought that this is due to the development of collateral circulation-like pial vessels (10,19). Time-SLIP MRA showed that, with manual compression of the right carotid artery, the vessels of the posterior circulation were extended and well described and they appeared to anastomose with the middle cerebral artery. This suggests pial vessels and dural arteriolar anastomoses from the posterior circulation.
A limitation of this study is that the TASL-MRI took a long time to perform (6.8 min). This long inspection time made it difficult to completely occlude the right carotid artery and was uncomfortable for the volunteers. In both subjects, TASL-MRI showed a partial perfusion territory in the left internal carotid artery (Figs. 2 and 3, purple). Incomplete compression of the right carotid artery might have affected the change in the contributions to the perfusion territory, not only in the basal ganglia or thalami, but also in the cerebral hemisphere. Although these were model cases of a partial internal carotid artery stenosis, the changes and recovery of the perfusion territory could still be visualized.
A further limitation was that ASL uses a relatively short-term bolus. The bolus is generated by the inversion of blood water, which decays with T1 relaxation of the blood. Loss of this bolus, and therefore loss of the labeling signal, also occurs as a result of blood water exchange at the tissue level. With severe obstruction of the main brain-feeding arteries and the subsequent presence of collateral flow, arrival of labeled blood at the brain tissue may have been delayed. This would result in under-estimation of the flow territory. However, a previous study of patients with ICA occlusion demonstrated that a delay of 1600 ms, which is close to the delay used here, is a good trade-off between signal-to-noise ratio, wash-out of the tracer, and T1 relaxation (20).
Finally, our study was of a small number of subjects. More study subjects are needed, especially those with variations in the anatomic features of the circle of Willis.
In conclusion, in this pilot study, by using TASL-MRI and 2DPC-MRA we were able to visualize dynamic changes in the perfusion territories of individual brain-feeding arteries and flow direction in the circle of Willis during manual compression of the carotid artery, and after decompression, in healthy volunteers.
