Abstract
Dihydrotestosterone (DHT) is the most potent natural androgen in humans. There has been an increasing interest in this androgen and its role in the development of primary and secondary sexual characteristics as well as its potential roles in diseases ranging from prostate and breast cancer to Alzheimer's disease. Despite the range of pathologies shown to involve DHT there is little evidence for measurement of serum DHT in the management of these diseases. In this review we describe the physiology of DHT production and action, summarize current concepts in the role of DHT in the pathogenesis of various disorders of sexual development, compare current methods for the measurement of DHT and conclude on the clinical utility of DHT measurement. The clinical indications for the measurement of DHT in serum are: investigation of 5a reductase deficiency in infants with ambiguous genitalia and palpable gonads; men with delayed puberty and/or undescended testes; and to confirm the presence of active testicular tissue. Investigation is aided by the use of human chorionic gonadotrophin stimulation. Due to paucity of published data on this procedure, it is important to follow guidelines prescribed by the laboratory performing the analysis to ensure accurate interpretation.
Introduction
Androgens are one of the main families of steroid hormones present in humans: progestogens, androgens, oestrogens, glucocorticoids and mineralocorticoids. The androgen group consists of four hormones: dihydrotestosterone (DHT), testosterone, androstenedione and dehydroepian-drosterone. The relative potencies of these androgens are 300%, 100%, 10% and 5% respectively; these relative potencies are only approximate due to variations in species studied and the assay methodology used. 1
The aim of this review is to describe the physiology of DHT production and action, to summarize current concepts in the role of DHT in the pathogenesis of various disorders of sexual development, to compare current methods for the measurement of DHT and to determine the clinical utility of DHT measurement. This is complementary to the review on hypogonadism in men. 2
Production of androgens
Androgen synthesis
The major synthetic pathway of DHT formation and metabolism is outlined in Figure 1. Testosterone is synthesized in men by the testis and in women either directly by the adrenals and ovaries, or by peripheral conversion of androstenedione. Testosterone is then irreversibly converted to DHT by the NADPH-dependent enzyme 5α reductase, 3 or aromatized to the potent oestrogen 17β-oestradiol. 4
Biosynthesis of androgens. The formation of the major hormone classes from cholesterol: P450scc, cholesterol side-chain cleavage; 3βHSD, 3β-hydroxysteroid dehydrogenase; P450c21, steroid 21 hydroxylase; P450c17α, steroid 17α hydroxylase/17,20 lyase; P450aldo, aldosterone synthase; P450c11, steroid 11 hydroxylase; 17 βHSD, 17 β-hydroxysteroid dehydrogenase; SRD5A1 /5A2, steroid 5α reductase type 1 /2; P450arom, aromatase. The backdoor pathway: within the adrenal gland, 17α -hydroxyl progesterone is reduced by 5α reductase then 3α reductase giving 5α -pregnane-3α,17α -diol-20-one, which is cleaved by the 17,20 lyase activity of CYP17A1 to androsterone. Once in the periphery androsterone is reduced by 17β-hydroxysteroid dehydrogenase yielding 5α-androstane-3α, 17β-diol, and subsequently metabolized by 3α hydroxysteroid dehydrogenase enzymes (such as 17β hydroxysteroid dehydrogenase -6) to DHT without testosterone or androstenedione as intermediaries7,8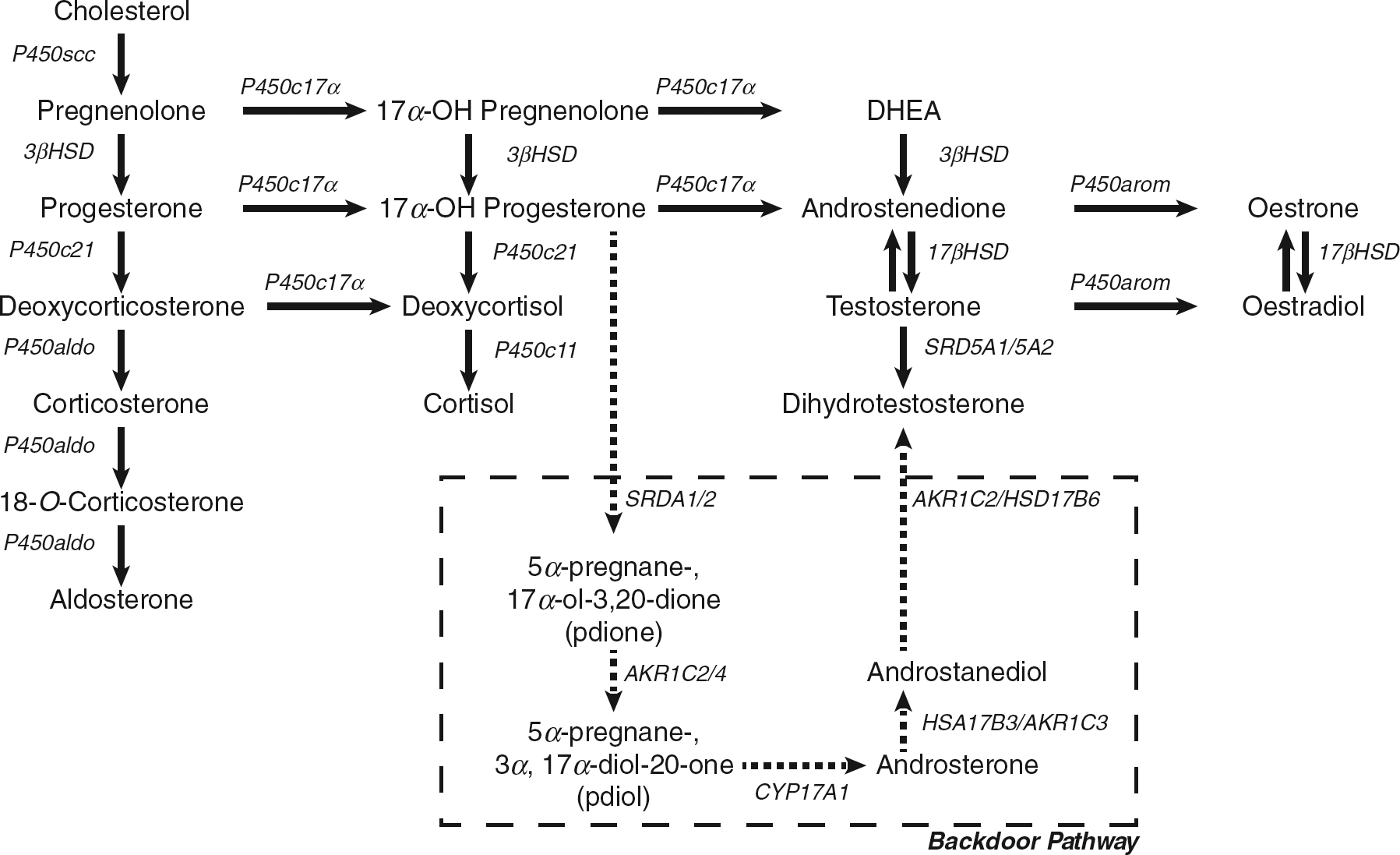
An alternative synthetic pathway, the ‘backdoor pathway’ has been proposed in female patients with congenital adrenal hyperplasia and other conditions.5,6 Examination of ratios of 5α -pregnane-3α,17α -diol-20-one metabolites to the classic steroid hormone pathway metabolites in urine steroid hormone profiles showed that patients with 21-hydroxylase deficiency have elevated ratios compared with controls. 6 This indicates the presence of a pathway that enables the production of DHT without testosterone or androstenedione as intermediaries.7,8 This pathway is also shown in Figure 1.
5α Reductase
There are two isoforms of the 5 α reductase enzyme: both are microsomal NADPH-dependent proteins that reduce the double bond at the 4-5 position in a variety of C19 and C21 steroids. They differ in regard to their optimum pH, chromosome location and affinity for testosterone. Type 1 5α reductase has an alkaline optimum pH of 8 and is active over a broad pH range 6.0-8.5. 9 Type 2 5α reductase has an acidic optimum pH of 5.5 and in contrast to the type 1 isoform is only active over an extremely narrow pH range. 9 The gene encoding type 1 5α reductase, SRD5A1, is located on chromosome 5 at 5pl5.5 10 while that encoding the type 2 enzyme, SRD5A2, is located on chromosome 2 at 2p23. 11 Type 2 5α reductase has a 10-15-fold higher affinity for testosterone than the type 1 enzyme. 12
The two enzymes are differentially expressed and regulated in regard to tissue distribution and timing of expression throughout life (see Table 1).13,14 The ontogeny of the 5 α-reductase isoforms has been elegantly clarified by Thigpen et al. 11 who have demonstrated the dynamic expression in humans. The type 1 enzyme is not detectable in the fetus, appears transiently in newborn skin and scalp, and is permanently expressed in the skin, including pubic skin, only after puberty.15,16 There is no difference in type 1 expression between balding and non-balding scalp. 11 The type 2 enzyme has been found to be transiently expressed in the newborn's skin and scalp, 11 the adult scalp 17 and in pubic skin. 18 Within the male reproductive organs the type 2 enzyme has been localized to the testis, epididymis and vas deferens, while type 1 appears more abundant in the seminal vesicles and ventral prostate. 12
Tissue locations of 5 α-reductase isoenzymes 169

The liver is the main source of circulating DHT in both genders, as it contains both type 1 and type 2 isoforms of 5α reductase. 11 The DHT found circulating in blood is predominantly from type 2 5α reductase activity (70-80%), with the remaining 20-30% from type 1 5α reductase. 3
The androgen receptor
The androgen receptor is a member of the nuclear receptor superfamily and binds both testosterone and DHT. These receptors reside in the cell cytoplasm bound to heat shock proteins, until testosterone or DHT diffuse into the cell and bind the receptor resulting in dissociation of the heat shock protein. This enables the ligand-androgen receptor complex to enter the nucleus where it is phosphorylated, dimerized and binds to androgen response elements in the promoter regions of androgen-regulated genes after co-activator recruitment, thereby affecting gene transcription. 19 This is illustrated in Figure 2.
Androgen receptor (AR) binding. Sex hormone binding globulin transports most testosterone around the circulation to the target cell where it is reduced to dihydrotestosterone (DHT). The DHT can bind the AR on dissociation of a heat shock protein. The AR then dimerizes and is phosphorylated (P) before transfer into the nucleus. The AR complex binds to the upstream region of target genes and after recruitment of co-activators, such as AR-associated co-regulator 70, can initiate gene transcription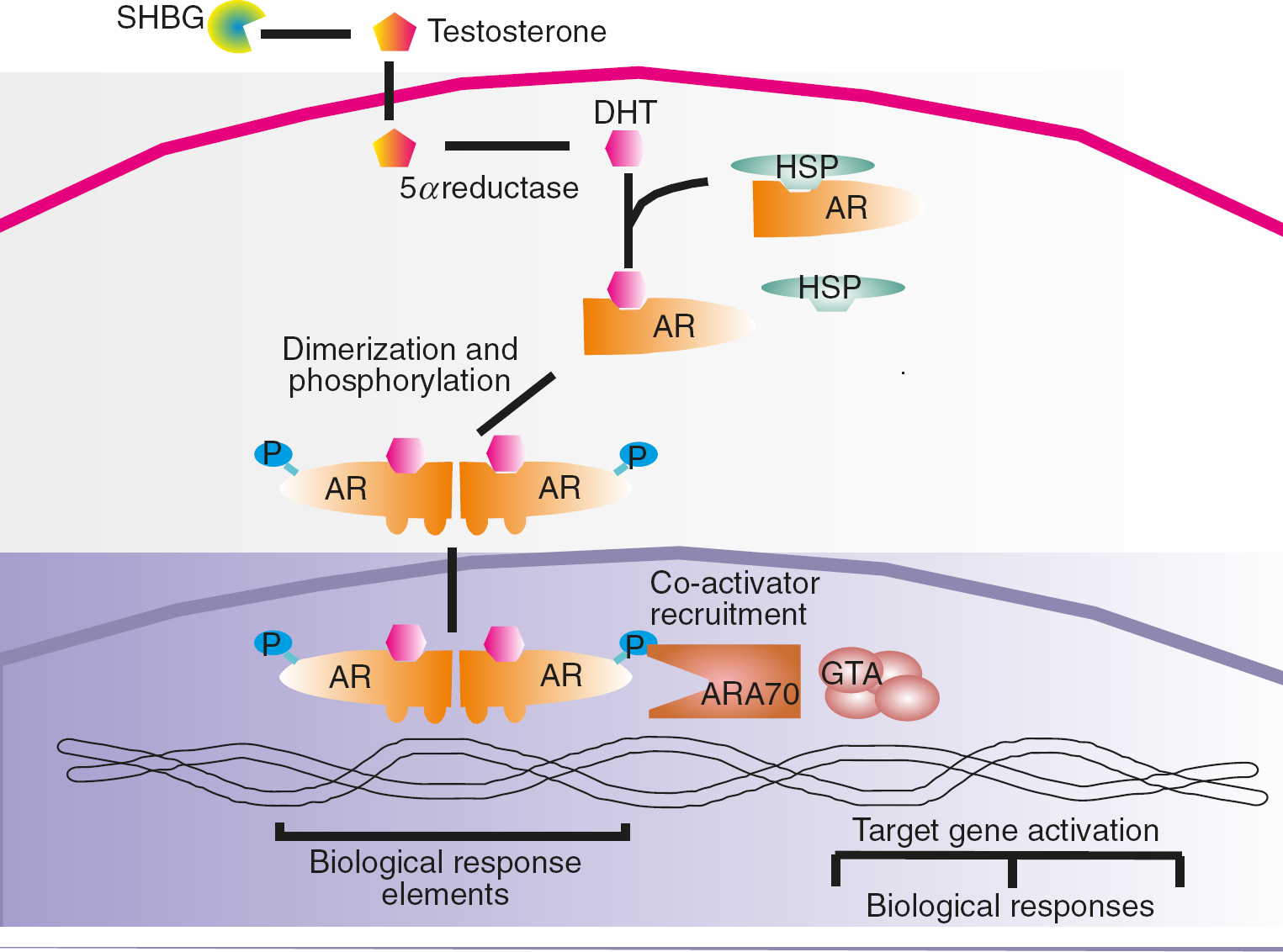
DHT has a more potent androgenic effect than testosterone because its binding affinity to the androgen receptor is approximately double that of testosterone and, moreover, it has a dissociation rate about a fifth of that of testosterone. 20
DHT metabolism and excretion
DHT is metabolized primarily by 3α-hydroxysteroid dehydrogenase reduction in the liver or the target androgen tissue with subsequent glucuronidation or sulphation and excretion in the urine.21–24 The formation of these conjugates and their clinical significance has already been reviewed, with studies of these end products showing that they have no or little androgenic function. 25
Physiology of DHT
Neonate
Male sexual development in the fetus requires two complementary processes: regression of the Müllerian ducts under the influence of anti-Müllerian hormone (AMH), and stimulation of the androgen receptor by testosterone and DHT. 4 Testosterone secretion by Leydig cells begins at about 60 days postconception under stimulation by placental human chorionic gonadotrophin (hCG). At about 16 weeks, testosterone rises to adult concentrations as the fetal pituitary begins to secrete luteinizing hormone (LH). The local formation of DHT by 5α reductase type 2 results in differentiation of the urogenital sinus and the male phe-notypic external genitalia. The action of 5α reductase type 2 on progesterone in the blood and amniotic fluid protects the male fetus from the actions of progesterone binding to the androgen receptor thereby inhibiting formation of the female phenotype. 26
Male childhood
Following birth there is a gradual increase in gonadotrophin release in men with a consequent rise in testosterone that peaks in the first 2-6 months of postnatal life. 27 Testosterone secretion and therefore DHT subsequently both fall to undetectable levels after this and remain undetectable until the adrenarche at about six years of age. 27
Male puberty
Prior to the physical onset of puberty, an increase in the secretion of gonadotrophin releasing hormone (GnRH) pulses from the hypothalamus is associated with a consequent increase in LH secretion, particularly during sleep. 28 This stimulates an increase in gonadal steroid secretion, principally testosterone from the testis. 29
During late puberty the daytime secretion of LH increases. This is in turn followed by an increase in plasma testosterone secretion, with the greatest increase seen in pubertal Tanner stage 2. At the end of puberty, testosterone in men shows a marked diurnal variation,30–34 despite LH and follicle stimulating hormone (FSH) being secreted throughout the day. 28
Adult male
Testosterone continues to be the principal testicular androgen, released in episodic pulses. As mentioned above, a diurnal rhythm is also detected with an elevation of testosterone secretion through the night with peak serum testosterone at 6-9 00, with a subsequent fall during the day.30–34 Approximately 95% of plasma testosterone is synthesized in the testis with the remainder being adrenal in origin. Serum DHT concentration is approximately l/10th that of testosterone while androstenedione is about one-fifth. Circulating DHT is principally derived (70%) from conversion of testosterone by non-gonadal tissues, with 30% being directly secreted by the testis or adrenals: the prostate does not contribute to circulating DHT. 35
Elderly males and the andropause
There is much controversy over the existence of the andropause. It is well accepted that there is a decrease in serum testosterone concentration with age associated with an accompanying drop in the amplitude of the diurnal variation. 36 This decrease in testosterone is paralleled by a paradoxical increase in sex hormone binding globulin (SHBG) concentration thereby decreasing the free and bioavailable testosterone concentrations. 37 The effect of the ‘andropause’ on DHT concentrations is unclear, particularly since DHT binds to SHBG. Some studies report a decline,38–40 others report no fall with age41–45 and one study actually reports an increase. 36
Female puberty and the adult female
DHT does not appear to play any significant role in female puberty. The predominant androgens are androstenedione and testosterone, which is primarily derived from peripheral conversion of androstenedione.46–48 During the female adult life, testosterone production from both ovaries and the adrenals falls so that by 50 y the circulating concentrations are half those seen at age 20 y. 49 Circulating DHT is almost entirely produced by peripheral conversion and has a circulating concentration of 3-10% that of testosterone. 50 DHT fluctuates within narrow ranges throughout the menstrual cycle but with no consistent relationship to the phase of the cycle. 48
Menopause
From the time women enter menopause until the age of 80 y, testosterone concentrations remain stable or may even increase slightly.51,52 There is no evidence of a fall in DHT concentrations throughout female adult life. In women who have undergone an oophorectomy, testosterone concentrations drop by 50%. 51 Women who have undergone a premature menopause show an equal fall in testosterone concentration and have similar values to women undergoing a natural menopause. 53
Clinical utility of DHT
Disorders of sex development
Historically terms such as intersex and Hermaphrodite were employed for disorders of sexual development. This terminology compounded the lack of understanding of the molecular mechanisms of these disorders and caused offense to patients due to the negative connotations of the words. In 2006 a consensus conference in Chicago agreed upon a re-classification based on chromosomal gender. 54 The term DSD (disorders of sex development) was agreed upon as a title for the collection of disorders and is defined as a congenital condition in which the development of chromosomal, gonadal or anatomical sex is atypical. 55 There are a large number of DSD (see Table 2) and only those in which DHT may play a role are covered here.
A selection of the disorders of sex development (adapted from ref. 54 )

DSD, disorders of sex development; AMH, anti Müllerian hormone; LH, luteinizing hormone
46, XY DSD: disorders of androgen action
Males born with defective androgen signalling pathways present with varying degrees of under-masculinization ranging from male phenotype (partial androgen insensitivity syndrome, with infertility or a minor degree of hypospadias) to a completely female phenotype (complete androgen insensitivity syndrome). Individuals are 46 XY males. These three conditions were previously classified as male pseudohermaphroditism or intersex before a full understanding of the molecular action of androgens was established. The term XY DSD is now in use 55
Androgen receptor defects can be classified as absent, having reduced affinity or quantity, and receptor-positive androgen resistance. More than 300 mutations in the androgen receptor are known to give rise to androgen resistance. 56 These mutations are found throughout the gene, with the most common mutation being single base substitutions. There is no genotype:phenotype relationship with particular mutations resulting in different phenotypes even within a family. 57 The typical postpubertal individual with an androgen receptor defect has a serum testosterone concentration at or above the adult male upper limit with raised LH and normal or only slightly raised FSH. The serum oestradiol and SHBG are both raised. 56
Due to the similarity in the clinical presentation of disorders of androgen action and disorders of androgen synthesis it is important to distinguish between them for clinical management. The ability of the pituitary-gonadal axis can be tested by stimulation with hCG (see Measurement of DHT) and androgen resistance can be tested by a short course of stanozolol (200 μg/kg/day for 3 days). Androgen resistance in the liver manifests by a failure of SHBG to fall after eight days (normal subjects show a fall of 45-55%). 58
46, XY DSD: disorders of androgen synthesis
This covers a spectrum of disorders resulting from deficiencies in any of the five hormones necessary for the production of testosterone or developmental abnormalities in the testis (see Figure 1). 4
5α Reductase type 2 deficiency
Classically men with 5α reductase type 2 deficiency present with ambiguous genitalia, the testes may be undescended as inguinal or labial masses; there is a labia-like scrotum, a urogenital sinus with a blind vaginal pouch and a clitoris-like phallus. 59 Wolffian structures differentiate normally and affected individuals have epididymes, vas deferens, seminal vesicles and a small prostate. 60 Virilization occurs at puberty with a male pattern of musculoskeletal development (although to a lesser degree than unaffected brothers), enlargement of the phallus and descent of the testes into a hyperpigmented scrotum. Affected males have normal spermatogenesis (if the testes are descended) and are fertile, 61 with a male sex drive.
The clinical spectrum of 46XY individuals with 5α reductase type 2 deficiency has subsequently been shown to be very wide and should include female phenotype with little or no virilization. While a female phenotype is still believed by some to be rare 62 a recent study found 41% (10 out of 23) of 46XY DSD adult females attending a DSD clinic had 5α reductase type 2 deficiency. 63
First published reports of affected males showed that cutaneous androgenization (i.e. acne, body hair and male pattern baldness) did not develop. 59 Subsequent studies show normal 5α reductase type 1 activity9,64 and normal sebum excretion and acne in affected individuals,65,66 although to date male pattern baldness has not been observed. 60
Affected 46XY individuals have high normal to elevated serum testosterone with decreased DHT concentrations and therefore elevated testosterone/DHT ratios.60,62,67,68 This is the basis of biochemical diagnosis with an elevated T:DHT ratio, with or without hCG stimulation. LH and FSH are elevated two to three-fold. 69 Partial enzyme deficiencies can be difficult to diagnose prepubertally with short hCG stimulation tests giving T:DHT ratios found to be within the normal range,67,70 but false-positive results have been reported in boys aged 6-9 y. 71
Females (46XX) with 5α reductase deficiency are phenoty-pically normal and are detected biochemically due to investigation of delayed onset menarche and decreased body hair growth 72 or as part of family studies of siblings of children with DSD.
Over 50 different total and partial gene deletions, nonsense and mis-sense mutations have been reported throughout the gene (Human Gene Mutation Database at the Institute of Medical Genetics in Cardiff http://www.hgmd.cf.ac.uk: srd5A2 gene). In two individuals the entire coding sequence of the gene was found to be deleted.9,70,73–75 The variation in mutations, and the resultant amount of functional activity of the gene remaining, account for the wide spectrum of phenotypes, from apparently female to male with hypospadias or microphallus.70,75–77 Certain mutations are found to be specific to different ethnic populations, which probably resulted from a founder effect with inbreeding. 60 There is no clear genotype: phenotype relationship with one mutation resulting in phenotypic variation ranging from female phenotype to microphallus and hypospadias. 62
46 XY DSD: other
Persistent Müllerian duct syndrome
This is a rare form of familial male DSD. In this condition 46 XY males have a uterus and Fallopian tubes in addition to the Wolffian structures that include the seminal vesicles, epididymis and vas deferens. 78 It is a heterogeneous condition caused by defects in the synthesis or action of anti-Müllerian hormone, such that patients can have a normal or absent concentration of the hormone. 79 In the normal male fetus anti-Müllerian hormone transcripts are expressed in the testicular tissue from eight weeks onwards where it promotes degradation of the Müllerian ducts that become the fallopian tubes, uterus and upper portion of the vagina in females. 80
Hypospadias, micropenis and cryporchidism
Hypospadias is defined as a displacement of the urethral meatus from the tip of the phallus to the ventral surface; it occurs in isolation or with cryptorchidism and/or micropenis. Mutations or abnormalities in the 5α reductase genes or the androgen receptor are rare in isolated hypospadias.77,81–83 The aetiology is believed to be predominantly multifactorial. 84
Micropenis is an anatomically normal penis that has a length less than 2.5 standard deviations below the mean for age. It is apparent that any defect resulting in an inadequate testosterone concentration can result in micropenis and androgen production defects would appear to be the dominant cause since mutations or abnormalities in the 5α reductase genes or the androgen receptor are rare. 85
Cryptorchidism describes an incompletely descended testis. It can present either unilaterally or bilaterally. It has many causes including defects in androgen production and action, and is common with an incidence of 1-1.6% in the newborn. 85
Androgenic alopecia
There is a paradox in the relationship between the increased growth of hair on the body at the same time as loss of hair on the scalp, particularly since they are both believed to be due to the same stimulus, i.e. androgens. Androgenic dependent alopecia, with a temporal recession of the frontal hairline and diffuse hair-loss on the crown, is reported to affect around 50% of Caucasian men over 40 y of age. 86 Many believe that this phenomenon is DHT dependent - particularly as it is not believed to occur in subjects with 5α reductase deficiency. Although most studies have found no correlation between serum androgen concentrations and androgenic alopecia:87–89 associations with both DHT 90 and testosterone 91 have been reported. Therapeutic studies with finasteride, a specific and potent inhibitor of 5α reductase type 2, however, does support a role for DHT as administration has been shown to improve hair growth and reduce scalp DHT in both animal studies and men.92–96 This is consistent with studies showing the presence of 5α reductase type 2 in hair follicles. 97
Genetic analysis suggests a polygenic aetiology for androgenic alopecia,98–100 which may involve the androgen receptor.100,101 Studies on the scalp showed subtle differences in androgen interconversion and androgen receptor numbers between balding and non-balding areas. 102 There is now a genetic test for particular variants of the androgen receptor gene known to have an increased risk of developing androgenic alopecia. 100
The development of androgenic alopecia in women is less well understood with both androgen-dependent and independent factors contributing. 100 Female pattern baldness was found to be associated with increased production rates of testosterone but not DHT, 103 and female hair loss can be associated with other signs of hyperandrogenism (e.g. hirsuitism, polycystic ovary syndrome [PCOS]). However, as with men, no apparent relationship with serum androgens has been found.104–106 While finasteride treatment is contraindicated in fertile females due to the risks of feminization of a male fetus; in postmenopausal women finasteride treatment for one year did lower plasma DHT levels yet did not slow the progression of hair loss. 107
Acne
Androgens trigger and aggravate acne but acne is likely to be multifactorial. 108 There is conflicting data on the role of 5α reductase in the human sebaceous gland. Whereas type 1 5α reductase has been demonstrated by immunocytochem-istry in the glands in acne prone areas of skin, 109 Dijkstra et al. 110 found that sebaceous glands predominantly convert testosterone to androstenedione. There is no known correlation between serum DHT concentrations and sebum secretion rates or the extent or severity of acne. 111 Males with 5α reductase deficiency, and those on finasteride (an inhibitor of 5α reductase) for benign prostatic hyperplasia, did not decrease sebum production compared with a control group. The same study showed that androgen insensitive subjects had no sebum production with results identical to preadrenarchal children. 64 More recently in a small study Kohler 112 showed that inhibition of 5α reductase with finasteride seems to help some female patients with acne.
Hirsutism
Hirsutism is defined as the growth of hair in women on the face and body which appears in the same pattern and with the same temporal development as in men. Such hair growth in women is caused either by an underlying state of androgen overproduction, possibly due to body mass, 113 or enhanced local sensitivity to androgens. Type 1 5α reductase has been shown in the skin of both hirsute and non-hirsute women. 15 Numerous attempts have been made to demonstrate increased androgen interconversion in the skin. Studies demonstrating this increased enzyme conversion in the skin may have been confounded by an increase in the volume of skin organelles as they were based on the patients total mass of skin and no normalization for skin structural content was performed. When enzyme activities are standardized by protein content or DNA, the differences disappear.114–116 Attempts to assess 5α reductase activity in hirsute patients by looking at metabolites of DHT in the circulation and urine have been unsuccessful. 117 Studies looking at differential expression of 5α reductase isoenzymes in hair follicles have been hampered by detection of 5α reductase type 2 which is constitutively expressed in the hair follicle in vivo but not during in vitro culture. 118
The majority of women with hirsutism with or without the PCOS have normal concentrations of circulating androgen hormones, 119 yet increased 5α reductase activity has been demonstrated in ovarian tissue and in the urine metabolites of women with PCOS. 120 The concentrations of DHT in serum from female patients are at the lower limit of currently available immunoassays and, therefore, distinguishing between possible normal and pathogenic concentrations is not possible. Newer mass spectrometric methods may enable this differentiation but currently DHT is not recommended as part of the investigations for PCOS. 121
Gynaecomastia
Gynaecomastia is the development of palpable mammary glands in men, resulting in breast enlargement. Physiologically, gynaecomastia occurs in neonates, at puberty and in old age. Pubertal gynaecomastia is a benign breast enlargement and has a prevalence of 4-65%. 122 The pathogenesis is still undetermined; classically it has been thought to be a result of the physiological imbalance between androgens and oestrogens that occurs during the hormonal turbulence at these ages. 123
The role of DHT is not clearly known but men with 5α reductase deficiency do not develop gynaecomastia.68-124 Serum DHT concentrations are lower in individuals with pubertal spontaneous gynaecomastia compared with healthy young volunteers 125 and treatment with DHT does appear to improve the condition.126,127 Although serum DHT concentrations rise during treatment, DHT falls to pretreatment values two months after the last injection but without recurrence of breast development over the following 15 months. 126 In many cases individuals undergoing treatment for benign prostatic hyperplasia (BPH) with finasteride, a 5α reductase inhibitor which lowers DHT, develop gynaecomastia. 128
Carcinoma of the prostate and benign prostatic hypertrophy
Benign prostatic hyperplasia (BPH) is very common in men, with a prevalence of >75% in men aged over 50 y. 129 Since the prostate requires DHT for its development and the prostate is a primary target for androgens, it would be expected that serum concentrations would be higher in BPH but it is unclear whether this is the case since there are reports supporting and contesting this notion.129–132 This may be due to a lack of a uniform approach to the measurement of DHT and the 5α reductase enzyme. Androgens are needed to develop these disorders. BPH is androgen-dependent as it does not occur in subjects castrated prior to the onset of puberty, or in individuals with type 2 5α reductase deficiency. 60
It is clear that treatment with finasteride results in a decrease in DHT but not testosterone. This treatment benefits the symptoms of BPH by improving urine flow and decreasing prostatic volume. 41 The mechanism for this is through the reduction in DHT and therefore androgen receptor stimulation within the prostatic stromal cells. 133 Some subjects experience mild symptoms of hypogonadism.134,135
Along with decreasing prostatic volume, finasteride also decreases the risk of prostate cancer - as demonstrated in the Prostate Cancer Prevention Trial. Current research involves the dual 5α reductase inhibitor, dutasteride, which results in a greater suppression of DHT than gained by finasteride. It is being investigated in the four year REduction by DUtasteride of prostate Cancer Events trial (REDUCE). Several recent literature reviews concluded that prostate disease development was associated with over expression of both 5α reductase isoenzymes, particularly the type 1 isoenzyme, with higher concentrations (3-4 times) of the enzyme present in malignant disease compared with BPH, 136 suggesting that a dual inhibitor would be more effective than finasteride alone. 137
There is no evidence to support any link between DHT concentrations and the diagnosis of BPH, 129 or indeed the risk of prostate cancer, the metastatic status of prostate cancer, anti-androgen treatment or survival.138–141 Further work involving direct determination of androgen levels in prostate tissue compared with serum may aid our understanding of these processes and the role of DHT. 142
Carcinoma of the breast
Breast tumours have been demonstrated to synthesize testosterone and DHT. 143 Local production was postulated to play a role in breast tumour growth since serum concentrations of DHT were significantly lower than in tissue. 143 Subsequent research has demonstrated that DHT can inhibit the growth of breast cancer cell lines in vitro.144,145 There is currently no evidence to support the measurement of DHT concentrations in these patients.
Alzheimer's disease
Recent studies have suggested that serum testosterone and DHT concentrations are lower in men with Alzheimer's disease. 146 Administration of testosterone, but not DHT, improved working memory and decreased hippocampal nerve growth factor protein in aged rats, while having no effect on beta-amyloid. Others believe that there is a significant association between the changes in circulating dehy-droepiandrosterone level and changes in the incidence of Alzheimer's disease (among other conditions) and other age-related changes. 147 As yet this research is in its infancy and a correlation between serum DHT concentration and Alzheimer's disease remains unclear.
Sexual dysfunction
As mentioned previously there is controversy over reported changes in DHT concentrations with age in men (section Elderly males and the Andropause). Therefore any sexual dysfunction associated with older age and the andropause cannot currently be related to DHT.
The role of testosterone in causing sexual dysfunction in women is unclear. In one study, premenopausal women with complaints of sexual dysfunction had lower adrenal androgen precursors and testosterone than age-matched controls with no sexual complaints. 148 Although this finding suggests a role for testosterone in female libido, the definition of androgen deficiency in women is not clear.
Fat mass and insulin resistance
DHT was negatively associated with different measures of fat mass and insulin resistance in humans. 149 A randomized control trial involving inhibition of 5α reductases suggests that there is no significant role for DHT in modulating bone mass, haematopoiesis or lipid metabolism in adult men. 150
Diagnostic utility
Measurement of DHT
Historically DHT quantitation has been performed by radioimmunoassay (RIA) using solvent extraction and oxidation of testosterone. The two laboratories offering DHT in the UK both use in-house RIA methods that are unpublished. There are several suppliers of commercial kits for DHT but these represent only two methods (i) a competitive RIA using iodinated DHT and (ii) a competitive enzyme-linked immunosorbent assay using horseradish peroxidase labelled DHT. All of the RIA assays use serum as the preferred sample type.
In the last few years liquid chromatographytandem mass spectrometry (LC-MSMS) methods have been developed that measure DHT or DHT in combination with other steroids in plasma, urine or tissue samples. These use solid-phase extraction, liquid:liquid extraction, precipitation or derivatization prior to analysis.151–159
Information regarding analytical variability is lacking as there is no external quality assurance programme. It is very method-dependent and the laboratory used should be contacted for this information. DHT concentrations obtained by LC-MSMS are approximately 60% those obtained by RIA in the one study that compared the 2 methods. 154 This is consistent with experience of quantitation of other sex steroids, where immunoassay can give an over estimation of steroid concentrations due to positive interference by other steroids.160–164 The major molecular interference in the DHT assays is testosterone -which is removed by potassium permanganate treatment.
Interference in the RIA methods by heterophilic antibodies would be minimized by the extraction protocols and obviously would not be a problem in LC-MSMS. Drug interferences in the assays are those which lower the concentration of DHT endogenously; 5α reductase inhibitors such as Finasteride or testosterone inhibitors which deplete the testosterone pool from which DHT can be made. Testosterone and testosterone esters do not appear to interfere in the LC-MSMS methods assuming the correct sample preparation and transitions are chosen. The specificity of the LC-MSMS methods is dependent on the method used and evaluation data on the ratio of the relative intensities of multiple mass transitions is not available and would need to be established before DHT by LC-MSMS is used in clinical applications. 165
hCG stimulation test
The hCG stimulation test has three main indications in the differential diagnosis of DSD: (1) to confirm the presence of active testicular tissue, (2) to aid in the diagnosis of a male with delayed puberty and/or undescended testes and (3) to aid diagnosis in an infant with ambiguous genitalia and palpable gonads. The test is recommended for the diagnosis of 5α reductase deficiency. 166 Several different protocols have been recommended but most series consist of only a few children. Moreover, it is important to note that false-positive test results have been reported in boys tested in the age range of approximately 5-10 y. 62 A further proviso must include caution in the interpretation due to the variable accuracy and performance of both testosterone and DHT analytical methods since testosterone assays are prone to interference by steroid precursors which might be of higher concentration in cases of DSD and secondly, DHT assays are not subject to external QA schemes.
We advocate the following protocol. On day one, blood is taken for testosterone, androstenedione and DHT. This is followed by the subcutaneous or intramuscular administration of 1500 units (infants) or 5000 units (over 2 y) of hCG. On day 4, blood is taken for testosterone, androstenedione and DHT.
hCG stimulates androgen production and the use of the protocol above will demonstrate whether androgens are adequately produced and if there is any deficiency in 17β hydroxysteroid dehydrogenase (rise in androstenedione but not testosterone) or 5α reductase (rise in testosterone but not DHT). The actual ratios of androstenedione to testosterone and testosterone to DHT which are used to diagnose enzyme deficiency will depend on the assays employed. In a series of 55 cases reported by Maimoun et al. 62 only 24 cases had hCG tests with testosterone and DHT measured and no data was given on the nature or quality of the assays employed over the 20 y of the study. This may explain the variable performance of the dynamic test. However, despite this it should be noted that 5/24 cases of genetically confirmed Type 2 5α reductase deficiency had post-hCG T/DHT ratios that would be considered to exclude the disorder (see Table 3).
Summary of human chorionic gonadotrophin tests from review of 5α-reductase deficiency 62
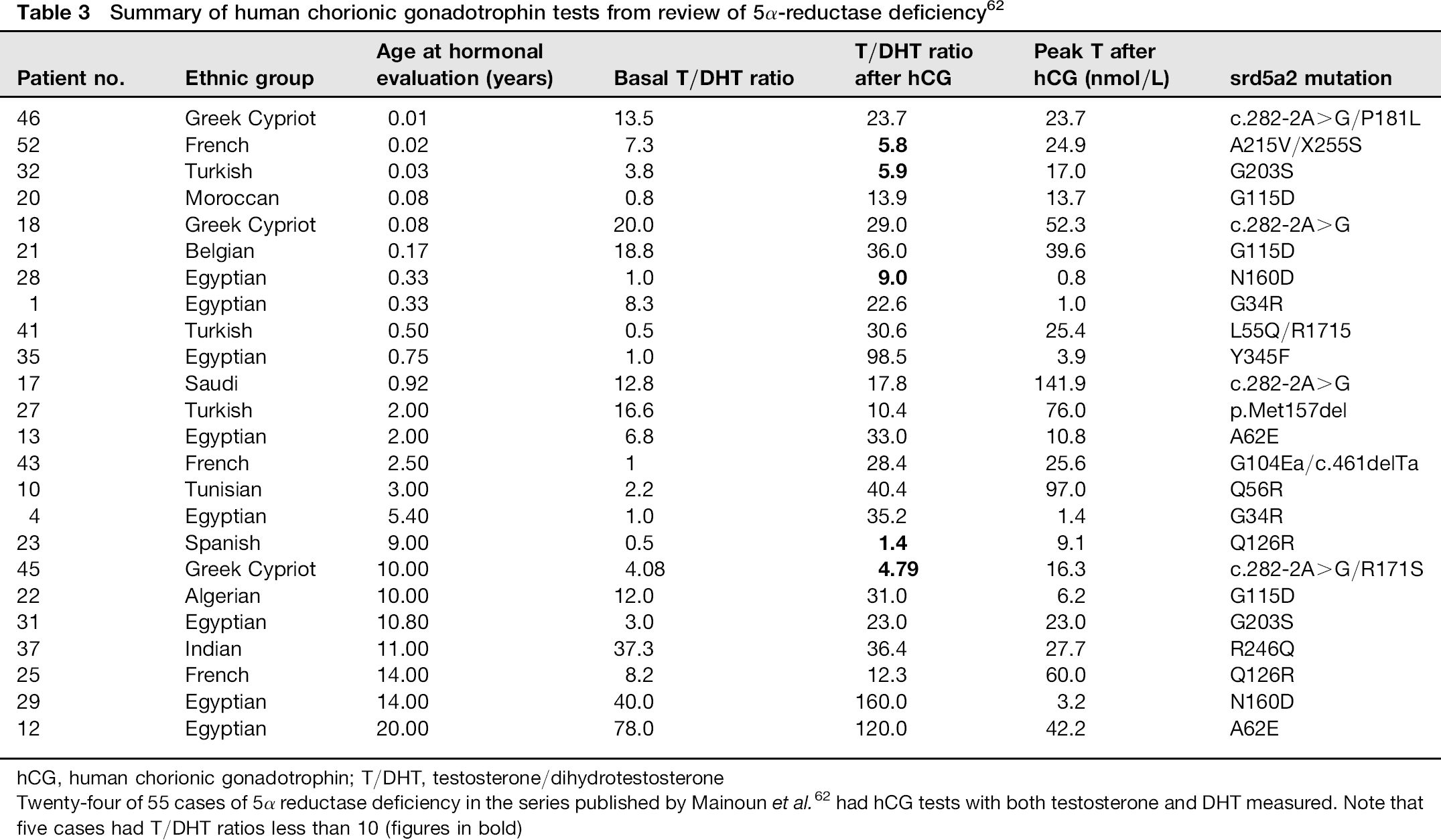
hCG, human chorionic gonadotrophin; T/DHT, testosterone/dihydrotestosterone
Twenty-four of 55 cases of 5α reductase deficiency in the series published by Mainoun et al. 62 had hCG tests with both testosterone and DHT measured. Note that five cases had T/DHT ratios less than 10 (figures in bold)
Urine steroid profiling
Urine steroid profiling in the UK is available using gas chromatography (GC) and gas-chromatography-mass spectrometry (GC-MS) by the Supra Regional Assay Service (SAS). It provides another means of investigating, and diagnosing some disorders of steroid production and metabolism. The biochemistry and clinical indications for urine steroid profiling have previously been reviewed in this journal.167,168 For the investigation of some gonadal and most adrenal disorders, including 5α-reductase deficiency a random spot urine can be used. However the test cannot be used until 1-3 months of age as the diagnostic ratios of 5α:5β reduced metabolites of Cortisol are not detectable until this age (Dr N Taylor, personal communication, 2011). In a pubertal child there is a characteristic distribution of androgen metabolites when the defect is present. 63 Unlike in the hCG test 17β hydroxysteroid dehydrogenase deficiency cannot be detected using a urine steroid profile.
Conclusion
DHT has a role in various pathologies and the clinical indications for its measurement in serum are the diagnosis of 5α-reductase deficiency in an infant with ambiguous genitalia and palpable gonads, in a male with delayed puberty and/or undescended testes and to confirm the presence of active testicular tissue, all in combination with an hCG stimulation test. Typical values of serum DHT are shown in Table 4, and as discussed above these are method dependent. Measurement of serum DHT levels is rarely of use in women.
Typical concentrations of serum dihydrotestosterone (DHT)
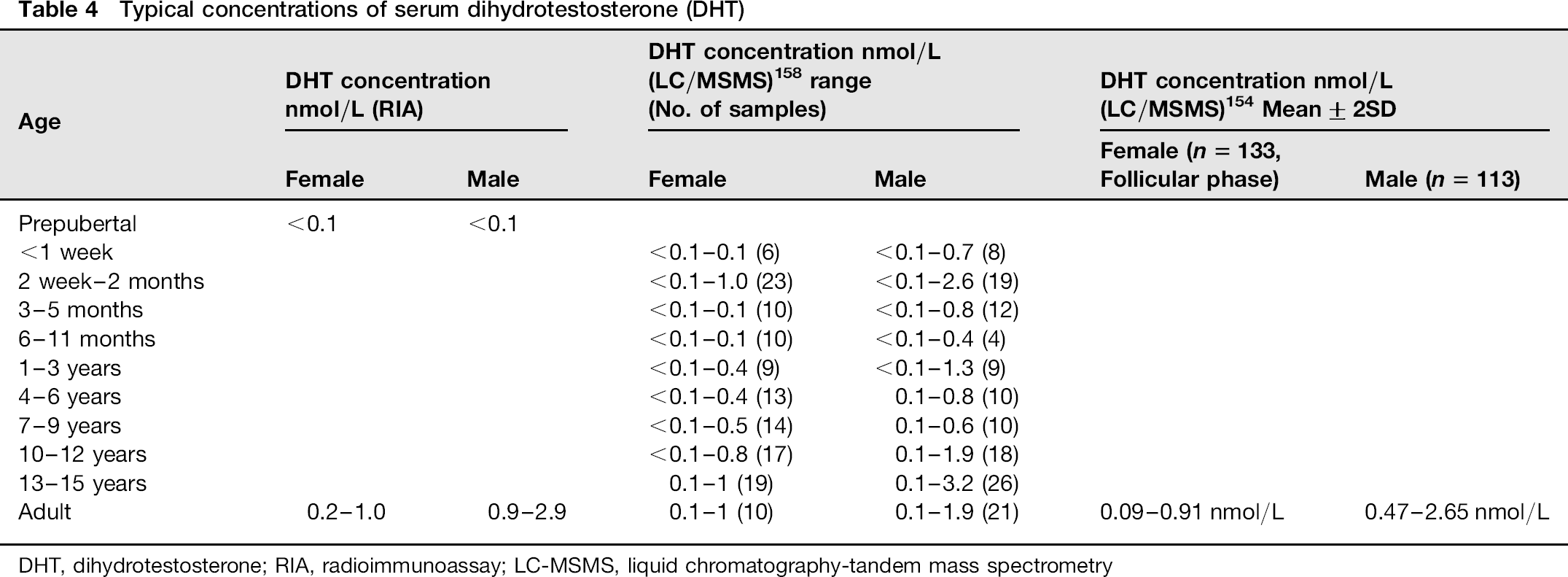
DHT, dihydrotestosterone; RIA, radioimmunoassay; LC-MSMS, liquid chromatographytandem mass spectrometry
Declarations
Footnotes
Acknowledgements:
Norman Taylor for advice on the interpretation of urine steroid profiling.
