Abstract
Lactic acidosis is a rare complication of haematological malignancies with a poor prognostic outcome and unclear aetiology. Possible mechanisms include high rate of glycolysis by cancer cells, in part due to overexpression of hexokinase II. The insulin-like growth factor (IGF)-system has an important role in normal as well as tumour cell growth. We present a case of a 79-year-old man with a diffuse large B-cell lymphoma and lactic acidosis. Initially, the patient was successfully treated according to the R-CHOP scheme. After recurrence of disease, the patient was treated according to a protocol of the Dutch-Belgian Haemato-Oncology Group (HOVON-85 study). Eleven months after completion of the last therapy, the patient still appeared to be in complete remission. Serum levels of IGFs, pro-IGF-IIE[68-88], IGF binding proteins (IGFBPs)-1 to - 4 , acid labile subunit (ALS), as well as ternary IGF-I-IGFBP-3-ALS complex formation, were determined in samples taken before, during and after treatment, respectively. Before treatment patient's serum concentration of the growth hormone-dependent parameters of the IGF-system and IGF-II were clearly reduced when compared with patient's values during remission of disease. On the other hand, during acidosis a relatively higher proportion of IGFs is present in binary complexes, instead of 150 kDa complexes, that may allow an increased access of IGFs to target cells including the malignant ones. Pretreatment serum levels of IGFBP-1 and -2 were elevated, decreased during therapy and normalized at remission. Especially IGFBP-2 seems a suitable marker for disease activity.
Introduction
The association of lactic acidosis and lymphoma was first described in 1964. 1 Lactic acidosis is a rare complication of haematological malignancies with a poor prognostic outcome and unclear aetiology. 2 Possible mechanisms include high rate of glycolysis by cancer cells, in part due to overexpression of hexokinase II.3–5 Hexokinase II can be overexpressed by insulin and insulin-like growth factors (IGFs), resulting in increased glycolysis and lactate production. 6 Other possibilities include increased production of lactate by tumour microembolism and decreased degradation of lactate by extensive liver metastases.2,7 Thiamine deficiency also can be involved in increased lactate production.8,9 Tumour necrosis factor-alpha may play a role by reducing the activity of pyruvate dehydrogenase, thereby accumulating pyruvate and increasing lactate production. 10
The IGF-system has an important role in normal as well as tumour cell growth. 11 The liver is the major source of IGF-I and IGF-II proteins in the blood circulation. Under normal circumstances, most of the IGFs exist in a 150 kDa ternary complex with IGF binding protein (IGFBP)-3 and the acid labile subunit (ALS). 11 The latter two proteins are also mainly produced by the liver, and their synthesis together with that of IGF-I, is regulated by growth hormone (GH). The remainder of the circulating IGFs is bound to one of the six IGFBPs, designated IGFBP-1 to -6, and less than 1% is present in the free form. The ternary complexes are thought to regulate the availability of IGFs to the target tissues, only the 40-50 kDa binary complexes and free IGFs can pass from the vascular compartment into the interstitial space. 11 Locally produced and circulating IGFs can be released from their binding proteins by IGFBP proteases and bind to the cellular membrane IGF-I receptor (IGF-IR), resulting in stimulation of proliferation and inhibition of apoptosis through the phosphatidyl-inositol 3'-kinase (PI3K/Akt) and mitogen-activated protein kinase (MAPK) pathways. 12 Several of the IGFBPs, e.g. IGFBP-2 and -3, are not only involved in the regulation of IGF bioavailability but also exhibit intrinsic, IGF-independent, biological effects. In this article we will focus on the role of IGF and IGFBPs associated with lactic acidosis.
Case report
A 79-year-old man presented with progressive shortness of breath, excessive sweats and swelling of his abdomen. On physical examination a dyspnoeic man was seen. His blood pressure was 106/69 mmHg, pulse rate 112 beats/ min and temperature 36.9°C. There were no enlarged lymph nodes. His abdomen was swollen. Liver nor spleen was palpable. Examination of chest and extremities showed no abnormalities. Computed tomography (CT) scan of the abdomen showed massive ascites and a process in the colon transversum. Cytological examination of ascitic fluid was compatible with non-Hodgkin's lymphoma (NHL). Colonoscopy and gastroscopy were performed. Histological examination of biopsies of a stenotic lesion in the colon and of a pathological structure in the stomach showed diffuse large B-cell lymphoma (DLBCL) in both locations. CT scan of the thorax showed multiple mediastinal and hilar lymphnodes. Bone marrow examination showed no evidence of malignancy. At presentation, both lactate dehydrogenase (LDH) activity and the level of lactate in patient's serum were strongly elevated, i.e. 1507 U/L and 2.4 mmol/L, respectively. The patient had no medical history of insulin resistance or diabetes type 2. Fasting serum levels of insulin and C-peptide were 13 mU/L (normal values: <27 mU/L) and 800 pmol/L (normal range: 400-1500 pmol/L), respectively, with a normal blood glucose level of 7.2 mmol/L. The diagnosis was DLBCL stage IV, being localized in the digestive tract, thoracic nodes and peritoneal surface, and complicated by the presence of ascites and lactic acidosis.
The patient deteriorated progressively and within seven days serum lactate and LDH activity had risen dramatically (Table 1). The patient was treated with classical chemo-immunotherapy according to the R-CHOP scheme (Rituximab-Cyclophosphamide, Doxorubicin, Vincristine and Prednisolone), supported by the administration of G-CSF (granulocyte-colony stimulating factor), with cycles every two weeks. In the following weeks patient's condition improved.
Patient's serum levels of IGFs, IGFBPs, ALS and various other laboratory parameters at various time points
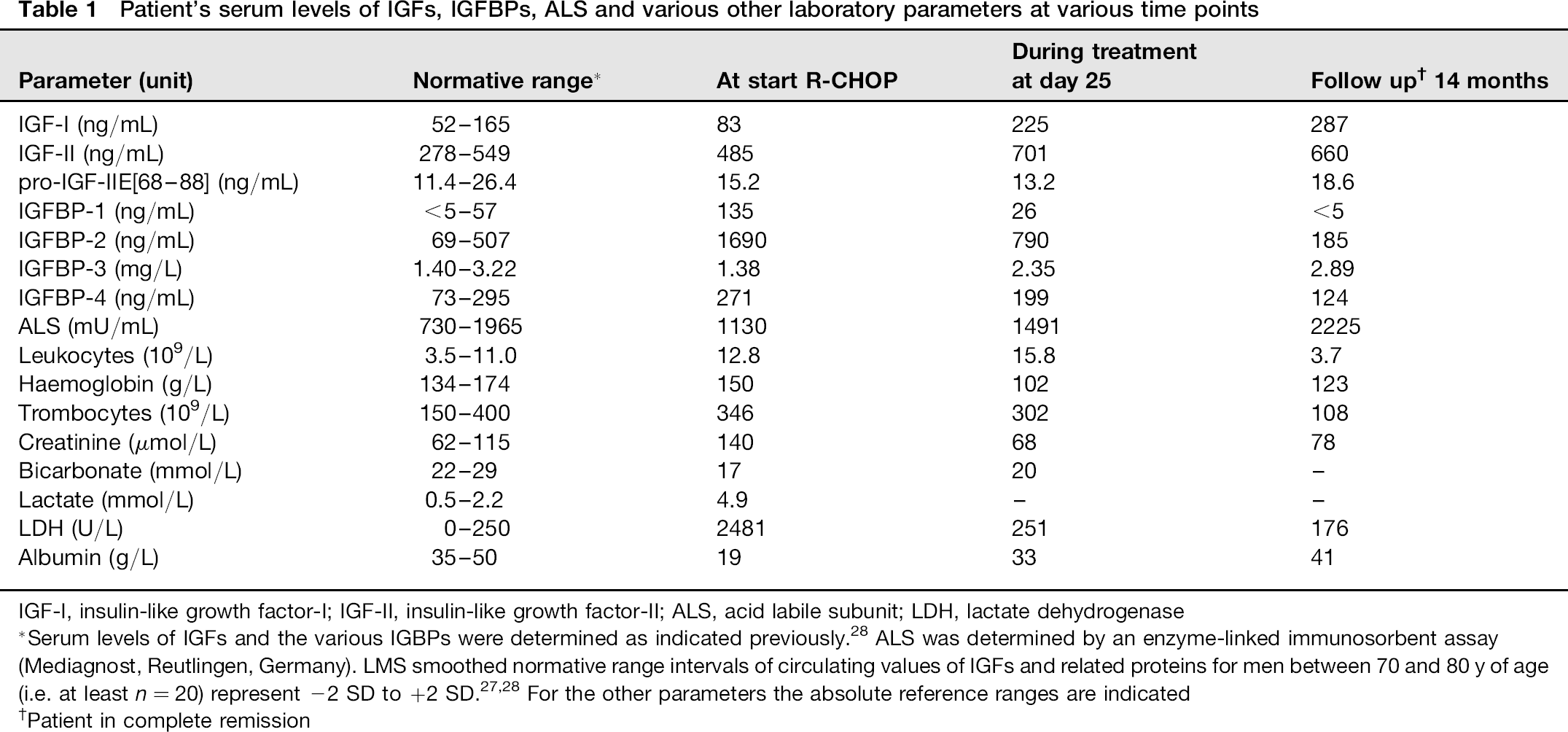
IGF-I, insulin-like growth factor-l; IGF-II, insulin-like growth factor-ll; ALS, acid labile subunit; LDH, lactate dehydrogenase
Serum levels of IGFs and the various IGBPs were determined as indicated previously. 28 ALS was determined by an enzyme-linked immunosorbent assay (Mediagnost, Reutlingen, Germany). LMS smoothed normative range intervals of circulating values of IGFs and related proteins for men between 70 and 80 y of age (i.e. at least n = 20) represent -2 SD to +2 SD.27,28 For the other parameters the absolute reference ranges are indicated
Patient in complete remission
After six cycles of chemo-immunotherapy according to R-CHOP therapy and two more Rituximab infusions, a CT scan showed complete remission. Unfortunately, two months after the therapy had been completed, there was a recurrence of disease in the supraclavicular lymph nodes. The patient was treated according to a protocol of the Dutch-Belgian Hemato-Oncology Group (HOVON-85 study; R-PECC; Rituximab, Prednisolon, Etoposide, Lomustine and Chloorambucil) after written informed consent. According to protocol he received four cycles of R-PECC once monthly followed by Ibritumomab Tiuxetan. Again, this resulted in complete remission. At the last visit for follow-up, 11 months after completion of therapy, the patient appeared to be still in complete remission.
Serum levels of GH, IGF-I, IGF-II, pro-IGF-II[68-88], several IGFBP's and IGF-IGFBP complex formation were determined in samples taken before, during and after treatment (Table 1). Before treatment of NHL and during lactic acidosis, patient's serum levels of the GH-dependent parameters of the IGF-system and total IGF-II, as well as 150 kDa complex formation between ALS, IGF-I and IGFBP-3 or -5 (Figure 1) were reduced when compared with patient's values during remission of disease. In contrast, pretreatment serum levels of the other IGFBPs were either in the supra- normal range (IGFBP-4) or elevated (IGFBP-1, and -2).
Serum IGFBP-1, which is subject to acute metabolic regulation, was high during lactic acidosis. At the same time, his serum level of pro-IGF-IIE[68-88] was within the normal range (Table 1), suggesting that high molecular forms of IGF-II did not play a significant role in the pathology of disease.
During treatment and remission, patient's serum levels of the GH-dependent parameters of the IGF system and total IGF-II had increased markedly, whereas the levels of IGFBP-1, -4 and, especially, IGFBP-2, declined. During complete remission, most of the IGF-I appeared to be sequestered in 150 kDa ternary complexes, instead of binary ones, as observed for normal serum (Figure 1).
The different molecular size classes of endogenous IGF-IGFBP complexes in serum, equilibrated with 125l-hlGF-l, were determined by neutral gel filtration through a 1.6 × 60 cm Hi-Load Superdex-200 column, as described previously.
29
The 125l content of each 1.2 mL fraction was counted in a γ-counter, (a) Column profile for age matched pooled normal adult serum (i.e. obtained from three men between 72 and 81 y of age), (b) Patient's serum during acidosis (solid line) and at remission of disease (dotted line). IGF-IGFBP, insulin-like growth factors-IGF binding proteins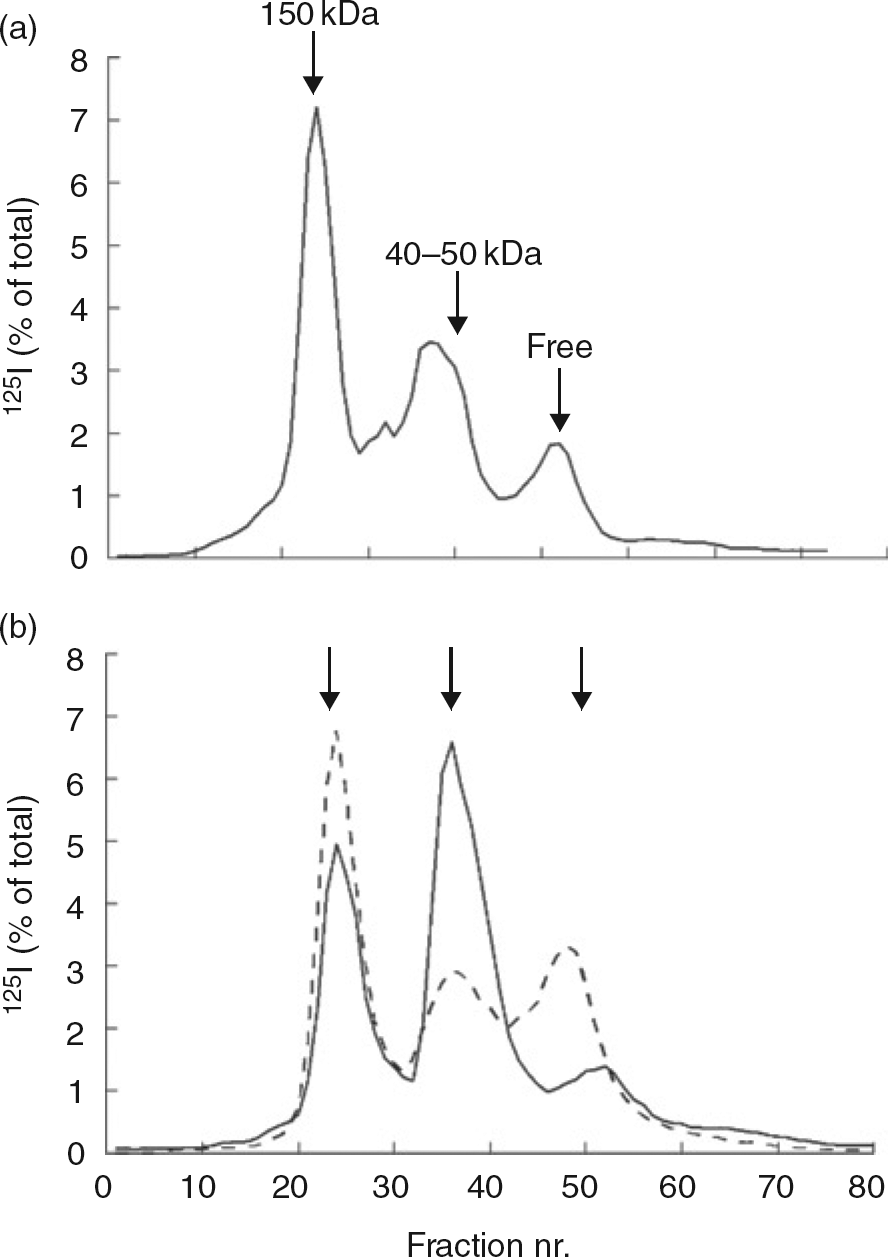
Discussion
It is well known that cancer cells can maintain a very high rate of glycolysis even in the presence of oxygen. Several factors may contribute to a high glycolytic activity of malignant cells. It has been proposed that the production of high molecular weight precursor forms of IGF-II (big IGF-II) by malignant lymphocytes exert profound endocrine and autocrine insulin-like effects, after binding to the insulin receptors, e.g. increasing cell proliferation, glucose uptake and glycolysis. 13 Ultimately, this may lead to hypoglycaemia, a condition known as non-islet cell tumour induced hypoglycemia (NICTH). In addition, excessive production of big IGF-II reduces serum levels of total IGF-I, IGFBP-3 and ALS, whereas that of IGFBP-2 increases. This picture resembles that seen in our patient before treatment. However, the level of pro-IGF-IIE[68-88], a crucial diagnostic biomarker for NICTH, 14 in patient's serum was not elevated and he did not show hypoglycaemia.
The relatively lower levels of IGF-I, and presumably also those of the GH dependent IGFBP-3 and ALS, as encountered in patient's serum during lactic acidosis, could have been caused by partial resistance to the hepatocellular action of GH. It has been shown that the primary defect in the GH/IGF axis occurs via an impaired IGF-1 response to circulating growth hormone with consequent diminution of normal negative feedback inhibition of IGF-1 on GH.15,16 Although single measurements of the GH level in patient's serum did not reveal elevated values, it is not known whether the expression of the GH receptor by target tissues was reduced or the signal transduction pathway disturbed.
The reduced formation of 150 kDa complexes could also have been caused by the reduction of pH. Molecular modelling of ALS at neutral pH, predicted 20 leucine-rich repeats arranged in a doughnut-like shape with patches of functional electronegative regions within the centre of the cavity where ALS interacts with IGF-IGFBP-3 (or IGFBP-5) binary complexes. 17 Hence, it may well be that during acidosis the interaction of ALS with the binary complexes is hampered. Another possibility is that the elevated concentration of IGFBP-2 competes with ALS-IGFBP-3 for IGF-I binding. In both cases, as a consequence, a relatively higher proportion of IGFs is present in binary complexes that may allow an increased access of IGFs to target cells including the malignant ones. Obviously, this would have had an unfavourable effect on the proliferation of tumour cells, deregulation of the glycolysis through increased expression of hexokinase II and aggravation of acidosis.
Acidification also leads to activation of cathepsin D, a proteinase which stimulates the proteolysis of IGFBP-3, leading to a decreased affinity of IGFBP-3 to IGF-I, resulting in an increased binding to IGF-IR. 18 However, during acidosis, our patient did not show an increase of proteolysis of serum IGFBP-3 (as revealed by Western blotting of pretreat-ment serum, data not shown). During remission of disease, patient's serum levels of IGFs, and ALS remained above the respective normative ranges. We do not have a definite explanation for this finding. It may well be that patient's own normal levels lie above the respective statistically processed reference ranges. If true, than this patient was and remains at a high risk for developing cancer. 19
Serum IGFBP-1, which is acutely regulated by insulin, was elevated before treatment but decreased in response to R-CHOP therapy. Evidence is accumulating that the serum level of IGFBP-1 may be predictive of outcome in critically ill patients, suggesting a possible prognostic role for this protein. 3 Similarly, the IGFBP-2 level in patient serum decreased during treatment and appeared to be within the normative range at complete remission. Since circulating IGFBP-2 levels are less susceptible to acute metabolic regulation, this protein could be a more suitable marker for disease activity than IGFBP-1.
Malignant lymphocytes may produce IGF-binding protein (IGFBP-2) in excess. 20 Various studies suggest that overexpression of IGFBP-2 has a tumourigenic potential. 21 Namely, in addition to IGF inhibitory effects, IGFBP-2 also exhibits IGF-independent intrinsic biological activities. IGFBP-2 has been shown to increase cell mobility and proliferation. An IGFBP-2 mediated loss of phosphatase and tensin homologue expression (PTEN), a tumour suppressor and P13K/Akt pathway activation, has been found for different types of cancer cells.22,23 In addition, IGFBP2 has been shown to stimulate the expansion of haematopoietic stem cells through activation of the p42/44 MAPK pathway and expression of several Hox genes. 24
We hypothesize that in our patient IGFBP-2 produced by malignant cells induced proliferation or expansion of these cells, leading to the excessive production of lactate, acidosis and subsequent effects on several parameters of the IGF system, as described above.
In general, the serum level of IGFBP-4 is up regulated during critical illness. 25 The same tendency was found for our patient. At present it is not clear which roles IGFBP-4 may play in NHL with acidosis. The expression of IGFBP-4 in cultured murine mandibular condyles appears to be increased by acidosis. 26 In addition, increased levels of IGFBP-4 have been found in patients with acute lymphoblastic leukaemia. 27
Further investigation is needed to understand the involvement of the IGF-system in NHL complicated by acidosis. This may contribute to an effective treatment of this disease.
Declarations
Footnotes
Acknowledgement:
Melanie van Berkum (Department of Metabolic and Endocrine Diseases, University Medical Center, Utrecht, The Netherlands) conducted part of the measurements of the various components of the IGF system.
