Abstract
Commonly used methods for assay of haemoglobin A1c (HbA1c) are susceptible to interference from the presence of haemoglobin variants. In many systems, the common variants can be identified but scientists and pathologists must remain vigilant for more subtle variants that may result in spuriously high or low HbA1c values. It is clearly important to recognize these events whether HbA1c is being used as a monitoring tool or, as is increasingly the case, for diagnostic purposes. We report a patient with a rare haemoglobin variant (Hb Sinai-Baltimore) that resulted in spuriously low values of HbA1c when assayed using ion exchange chromatography, and the steps taken to elucidate the nature of the variant.
Introduction
The assay of haemoglobin A1c (HbA1c) has been established for many years as the best way of assessing glycaemic control in diabetes mellitus (DM). Trials have related clinical outcome in patients with both type I and type II DM to prevailing HbA1c levels and shown that therapy to reduce HbA1c results in improved patient outcome.1,2
HbA1c is formed as the result of non-enzymatic glycation of the haemoglobin β chain. The initial reaction is the formation of an unstable aldimine and occurs between the carbonyl group on glucose and the N-terminal valine of the haemoglobin β chain (‘labile HbA1c’). This aldimine undergoes Amadori rearrangement resulting in the formation of a stable Schiff's base. 3 Glycation occurs at a rate proportional to the prevailing glucose concentration. 3 This, coupled with the usual life span of the red cell (approximately 120 days), means that in those with normal red cell survival, the proportion of haemoglobin present as HbA1c gives an indication of the average level of glycaemia over the preceding three months. Individuals with unusually short or long red cell survival may show lower or higher HbA1c results, respectively, for any given level of glycaemia. 4
There are a large number of different clinical assays for HbA1c but those in routine clinical use are of three main types – affinity chromatography, immunoassay and ion exchange high-performance liquid chromatography (HPLC). All methods are prone to reporting spuriously low values in conditions associated with diminished red cell survival, e.g. spherocytosis. Many methods are also susceptible to some degree to the presence of haemoglobin variants. The most obvious example is β thalassaemia where diminished production of a normal β chain makes methods reliant on detection of the glycated β chain unusable.
In any method type, when the haemoglobin variant leads to diminished red cell survival, this results in a lower HbA1c for the prevailing level of glycaemia (simplistically, the red blood cells spend less time in the circulation exposed to glucose, resulting in diminished formation of HbA1c). In addition, interference may occur in ion exchange HPLC methods because of the chromatographic properties of the haemoglobin variant. Failure to resolve the variant haemoglobin components from HbA1c and/or HbA0 may cause problems with peak integration and spurious HbA1c results. Thus HbAE and HbAD traits have been shown to result in overestimation of HbA1c on the Bio-Rad Variant II Turbo system (Bio-Rad Laboratories, Hercules, CA, USA) while, in the same study, the same haemoglobinopathy traits caused negative interference on the Menarini HA 8160 analyser (A. Menarini Diagnostics, S.r.l., Firenze, Italy). 5 Both systems use cation exchange chromatography as a means of separating haemoglobins.
We report here the presence of a rare haemoglobin variant (Hb Sinai-Baltimore) that gives a spuriously low HbA1c value when assayed using cation exchange HPLC on the Variant II Turbo 2.0 analyser – apparently due to the chromatographic properties of the variant.
Case report
The patient was a 92-year-old man with presumed type II DM since 2007. The diagnosis was based on the finding of an elevated two-hour glucose result in a standard 75 g oral glucose tolerance test. The diagnosis was never confirmed and glucose results since that time have all been between 5.1 and 6.0 mmol/L. When tested, the ratio of albumin to creatinine in urine had been consistently less than 2.5 mg/mmol. Management of hyperglycaemia had been with diet alone. He had no other relevant co-morbidities and his only regular medication was longterm aspirin. He had had regular HbA1c assays performed on blood since at least 2007. Until 2010, these were performed using previous versions of Bio-Rad's Variant HbA1c analysis system. During this period, there was no evidence for the presence of a haemoglobin variant and HbA1c values fluctuated between 43 and 49 mmol/mol (National Glycohemoglobin Standardization Program 6.1-6.6%). Haematological indices showed normal haemoglobin with a red cell count of 3.86 × 1012 /L (reference interval 4.3-6.0), a mean corpuscular volume of 108 fL (80-99) and a mean corpuscular haemoglobin (MCH) of 35 pg (27-33). Values for iron, ferritin, transferrin and folic acid were normal other than a transferrin saturation of 56% (16-50). Serum vitamin B12 was 205 pmol/L (170-600). Mutation analysis of the HFE gene revealed no abnormalities. In 2010, the analytical system was changed to the Variant II Turbo 2.0 analyser that uses shorter chromatography times than previous versions of the analyser and at this time an abnormality was noted in this patient's chromatogram.
Methods
Clinical HbA1c assay
Routine HbA1c analysis in this laboratory is carried out using cation exchange HPLC (Bio-Rad Variant II Turbo 2.0) as per the manufacturer's instructions. The sample is haemolysed and diluted on board the analyser and then applied to a 4.6 mm × 27.5 mm cation exchange column. The haemoglobin fractions are eluted using proprietary perchlorate buffer with a gradient of increasing ionic strength. The chromatography time is approximately 1.5 min and the column effluent is monitored at 415 and 690 nm. Clinical results are routinely reported in SI units (mmol HbA1c per mol total Hb - mmol/mol). In cases where there appears to be interference in quantification of HbA1c, we use an affinity chromatography method (Bio-Rad In2It) as backup. This system uses a single-test cartridge where penylboronic acid bound to a solid phase binds glycated haemoglobin in a haemolysed blood sample. After quantification of non-bound haemoglobin components (principally HbA0), glycated haemoglobin is eluted with sorbitol and quantified spectrophotometrically.
Identification of the haemoglobin variant and mutational analysis
Further haemoglobin analysis was initially carried out using the Bio-Rad Variant II β thal programme and cellulose acetate electrophoresis. Haemoglobin stability was assessed using the isopropanol test. Additional studies were carried out using electrospray ionization mass spectrometry (ESI-MS) of whole lysate, 6 the redissolved isopropanol precipitate and tryptic digests of the isopropanol precipitate. Finally, direct sequencing of the amplified β chain gene was carried out on an ABI 3130 × l genetic analyser using Big Dye terminators (Applied Biosystems, Foster City, CA, USA). 6
Results
In April 2010, it was noted that instead of a single HbA1c peak there were two peaks close together with elution times close to that expected of HbA1c (Figure 1). Integration showed that these peaks comprised 2.3% and 3.8% of the total Hb area. The integrator had identified the second peak as HbA1c and the result reported from the instrument was 18 mmol/mol (3.8%). Other peaks identified on this chromatogram (in addition to HbA0) were HbF (approximately 2.0% of total Hb) and labile HbA1c (approximately 2.3% of total Hb). Because of the abnormal appearances on HPLC and the fact that these peaks had the potential to interfere with HbA1c quantification, an alternative method was used to quantify HbA1c. Using affinity chromatography, HbA1c was 39 mmol/mol (5.7%). When this pattern of interference was confirmed in November 2011, additional detailed studies were carried out to determine the nature of the haemoglobin abnormality.
Chromatogram for Variant II Turbo assay for haemoglobin A1c (HbA1c) in the patient reported. HbA1c has been identified as the peak at 0.59 min (HbA1c = 18 mmol/mol; 3.8%). The peaks at 0.51 min (identified as labile A1c) and 0.59 min comprise 2.3% and 3.8%, respectively, of the total haemoglobin. Integration software identified the peaks (in order of elution time) as: HbA1a (0.17 min), HbA1b (0.23 min), HbF (0.28 min), labile A1c (0.51 min), HbA1c (0.59 min), P3 (0.79 min), P4 (0.86 min), ‘Unknown’ (0.93 min) and HbA0 (1.06 min)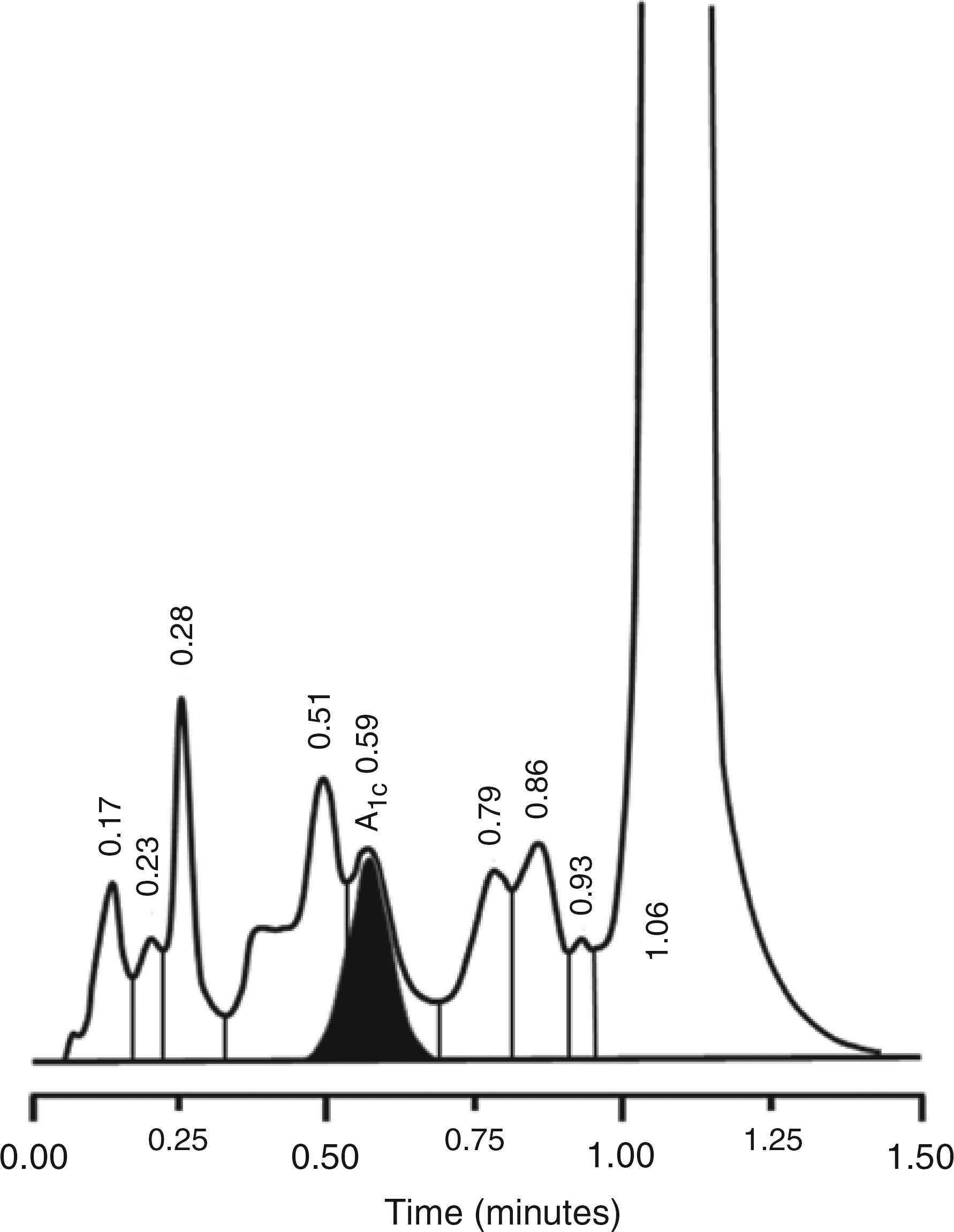
The Bio Rad Variant II β thal programme revealed an essentially normal profile as did cellulose acetate electrophoresis. However, the isopropanol test showed a mild instability; going cloudy at 20 min and forming a fine precipitate at 30 min. ESI-MS of whole lysate revealed an abnormal β chain comprising 33% of the total and with a mass 42 Da lower than that expected (Figure 2). This variant was enriched to a level of 72% in the redissolved isopropanol precipitate, confirming its instability. Further examination of tryptic digests of the precipitate indicated diminished quantities of the M + 1 and M + 2 ions of peptide β-3, which were replaced by ions of 42 and 21 m/z less. Taken together, these findings suggested a point mutation of one of the three glycine residues in peptide β-3 to valine. This was confirmed by direct sequencing of the amplified β chain gene which showed the patient was heterozygous for a mutation of GTG (Val) → GGG (Gly) at codon β18.
Transformed electrospray ionization mass spectra of redissolved isopropanol precipitate from the subject (a). (b) and (c) are subject and control lysates, respectively. The subject showed the expected normal α (15,126 Da) and β chains (15,867 Da) together with an additional β chain with a mass decrease of 42 Da suggestive of a Val → Gly point mutation. The low expression level of the variant (33%) suggested decreased stability that was confirmed by the examination of isopropanol precipitates, which showed enrichment of the 15,825 Da signal to 72% of the total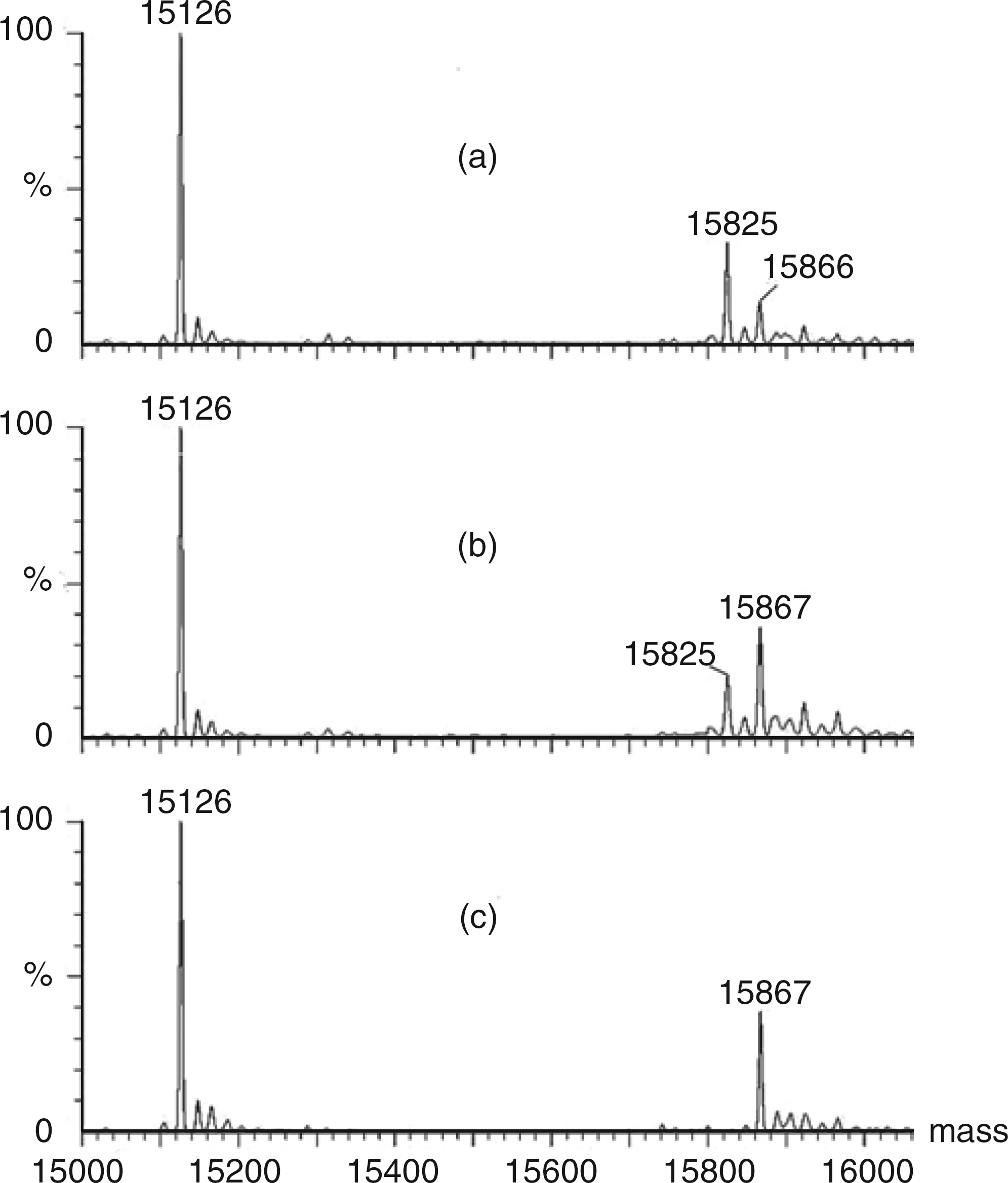
Discussion
Ion exchange chromatography methods for HbA1c quantification are known to be susceptible to interference from variant haemoglobins. Variants that are relatively common are often easily recognized. This may be because of their characteristic migration (the Variant II Turbo 2.0 analyser identifies abnormal haemoglobins such as labile HbA1c, HbF and HbC while carbamylated Hb, HbS, HbE and HbD are labelled as abnormally migrating variants). Alternatively, the laboratory may be alerted to the presence of a variant haemoglobin by results that appear implausible – when trouble shooting often identifies that a variant haemoglobin may be present.
In this instance, inspection of the chromatogram revealed an abnormality and raised the suspicion that an abnormal Hb may be present. This led to further investigations that identified the variant as Hb Sinai-Baltimore (β18Val → Gly).
There has been only one previous report of this mutation 7 where it occurred in a black infant together with HbS in a ratio of 55:45. He had mild anaemia (100 g/L), marked microcytosis and hypochromia. The variant was described as moderately unstable, but this result would have been compromised by the high level of HbF present (13.8%). The child's mother who was heterozygous for the variant alone (35% variant, 65% HbA) had normal haemoglobin, but moderate microcytosis and hypochromia. This was however thought to be due to heterozygosity for the α3.7 deletion. The presence of HbS trait in the child and α-thalassaemia in the mother obscured these haematological findings. So the identification of our case, lacking additional haemoglobin mutations, allowed a clearer definition of the phenotype associated with the β18Val → Gly mutation itself.
Our patient had normal haemoglobin, macrocytosis, slightly low red cell count and a slightly elevated MCH. Also there was clear molecular instability as evidenced by isopropanol precipitation and the level of the variant, which comprised only 33% of the total β chains. The large hydrophobic (CH, CH3, CH3) side chain of β18 valine is very highly conserved and evolution has tolerated only minor variation to a similar Met or Leu residue in a limited number of species. The change here to a much smaller glycine (H) would be expected to decrease molecular stability. This may lead to a slight shortening of red cell life span (particularly during episodes of pyrexia, for example) and result in a lowering of HbA1c.
The valine to glycine substitution that occurs in Hb Sinai-Baltimore will not affect the overall charge of the variant haemoglobin and so it may be expected that glycated Hb Sinai-Baltimore will elute close to HbA1c. It is possible that this is the peak seen in Figure 1 at 0.51 min that has been labelled ‘labile A1c’.
The alternative method (affinity chromatography) used in this case to assay HbA1c is less likely to be affected by abnormal haemoglobin variants as it detects all glycated haemoglobins. The total glycated haemoglobin (including glycated variants) is bound to phenylboronic acid on solid support before being washed off with a sorbitol buffer. Thus it generates a result for total glycated haemoglobin in relation to total haemoglobin.
It should go without saying that those providing clinical assays for HbA1c should be aware of the potential for haemoglobin variants to produce spurious HbA1c results in their specific method. Equally importantly, those managing patients with diabetes must be aware of the possibility of haemoglobin variants interfering in their patient's results – particularly in those areas of the world where the prevalence of haemoglobin variants is high. While the prevalence of Hb Sinai-Baltimore appears to be very low, this report serves to remind laboratory workers, pathologists and clinicians that caution is required when interpreting HbA1c results in individuals with haemoglobinopathies – even when the haemoglobinopathy itself may be of little clinical significance.
We described the identification of Hb Sinai-Baltimore in a patient with DM in whom the first indication of an abnormality was a spuriously low HbA1c result on cation exchange chromatography. Although this variant results in decreased stability of the haemoglobin β chain, it appears that the low reported results for HbA1c are mainly due to chromatographic changes consequent on the presence of Hb Sinai-Baltimore. Given the serum glucose concentrations reported over this period, repeat assay using affinity chromatography appears to give a more plausible result.
Declarations
Footnotes
Acknowledgements
None
