Abstract
Background
Obesity is associated with hypertension, but the exact mechanism is not fully understood. Bariatric surgery significantly decreases weight and blood pressure (BP). Low plasma nitric oxide (NO) and raised asymmetric dimethylarginine (ADMA), an endogenous inhibitor of NO, concentrations are associated with both obesity and hypertension. Correlations between the changes in these parameters were studied after bariatric surgery.
Methods
Weight, BP, plasma ADMA and NO were measured in 29 obese patients (24 female, 5 male) before and six weeks after bariatric surgery.
Results
Patients were 39.2 ± 1.2 (mean ± SEM) years old and weighed 126 ± 3 kg. Six weeks after the surgery, patients had lost 10 ± 0.7 kg (P < 0.0001) and mean arterial pressure (MAP) decreased by 11 ± 1.0 mmHg (P < 0.0001). The plasma ADMA concentration decreased by 24 ± 2% from 5 ± 0.4 to 4.0 ± 0.3 μmol/L (P < 0.0001). The plasma total nitrite concentration increased by 15 ± 1% from 51.4 ± 2.6 to 60 ± 3 μmol/L (P < 0.0001). The correlation between the decrease of ADMA and increase of NO subsequent to weight loss was significant (P < 0.0001). However, MAP was not correlated to the changes in ADMA or NO.
Conclusions
After bariatric surgery, beneficial changes in BP, NO and ADMA occur, but our findings suggest that these BP changes are independent of changes in the NO–ADMA axis. Other causes for the changes in BP should therefore be considered.
Introduction
Obesity is responsible for an increasing burden of morbidity, mortality and health-care costs worldwide. 1 The increased mortality is mostly associated with insulin resistance (IR), type 2 diabetes and cardiovascular disease. Patients with body mass index greater than 40 kg/m2, or greater than 35 kg/m2 with obesity-related co-morbidities, can be considered for one of the following bariatric surgery operations: gastric banding, sleeve gastrectomy or Roux-en-Y gastric bypass. 2 Some co-morbid conditions secondary to morbid obesity may be completely resolved following these surgeries, whereas other conditions such as high blood pressure (BP) may be partially resolved. 3
Bariatric surgery can result in approximately 15–25% long-term weight loss. The Swedish obese subject studies reported that the hypertension in patients undergoing surgical intervention was improved in comparison with the cohort without surgery. 4 Obese patients treated by bariatric surgery have decreased IR possibly due to increased peripheral insulin sensitivity. 5
The association of obesity and hypertension has also been widely reported, although exact mechanisms are not known. Many theories implicating inflammatory markers and growth factors secreted by adipose tissue have been postulated. 6 Nitric oxide (NO) in the vascular endothelium is produced by endothelial NO synthase (eNOS) and regulates vasodilator tone and BP. 7 Asymmetric dimethylarginine (ADMA) is the by-product of histone protein methylation in the endothelium, acts as an endogenous NOS inhibitor 8 and has been identified as an independent cardiovascular risk factor. 9 It is also produced by human adipocytes. 10 Decreased NO and increased ADMA are associated with obesity, IR and high BP. Increased concentrations of ADMA could contribute to the development of endothelial dysfunction in obesity, while weight reduction is associated with improvement in endothelial function. 11–13
The aim of this study was to examine whether the improvement in BP is related to changes in the NO–ADMA axis. We therefore looked at weight loss, IR and BP before and six weeks after bariatric surgery.
Methods
This was a prospective study of 29 morbidly obese patients before and six weeks after bariatric surgery, measuring BP, homeostatic model of IR (HOMA-IR) and plasma NO and ADMA.
Ethical permission was obtained from Imperial College Healthcare NHS Trust (Ref: 04/Q0406/25), and it was conducted according to the principles of the Declaration of Helsinki. Twenty-nine patients between the ages of 18 and 65 y undergoing bariatric surgery were included.
Blood pressure
BP was measured before and six weeks after surgery by the same clinician using a spot vital signs device (Model 42MTB-E1-M; Welch Allyn, Buckinghamshire, UK). The average of three separate recordings was taken. Appropriate cuff sizes were chosen for each patient depending on their individual arm circumference. Mean arterial pressure (MAP) was measured by dividing pulse pressure (SP−DP) by three and adding it to the diastolic pressure:
14
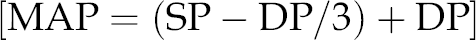
Sample collection
Plasma samples were collected in ethylenediaminetetraacetic acid vacutainers for the measurement of total nitrite and ADMA concentrations.
Samples for ADMA and total nitrite were stored at −20°C, within one hour of collection until analysis. Samples were analysed in duplicate.
Measurement of NO
Total NO assay (R&D Systems, Minneapolis, MN, USA) was used as instructed by the manufacturer. This assay determines NO concentrations based on the enzymatic conversion of nitrate to nitrite by the enzyme nitrate reductase. The intra- and inter-assay coefficient of variation (CV%) of the assay as stated by the manufacturer was 2.5% and 4.6% for the concentrations ranging from 30 to138 μmol/L. The recovery throughout the range of the assay was 92–117%. The assay was linear up to 119 μmol/L, with a limit of detection of 0.09 μmol/L.
A dietary influence on plasma nitrite concentrations has been described if the samples are collected after less than 16 h of fasting. 15 Samples were collected in our study after a 12-h fast, but the duration of fasting was the same between collection of pre- and postbariatric surgery samples. In the absence of drastic changes in intake of dietary protein, the extent of interference of diet on plasma nitrite concentrations would be expected to be similar in the samples collected at the two time points.
Measurement of ADMA
ADMA was measured by enzyme-linked immunosorbent assay (ELISA) (Immunodiagnostics, San Diego, CA, USA). The manufacturer's instructions were followed. The manufacturer quoted 0.5% cross-reactivity with symmetric dimethylarginine with intra- and inter-assay CVs of 2–4% and 3–4.5%, respectively. The assay was linear up to 103 μmol/L, with a limit of detection of 0.05 μmol/L. The average recovery of the assay was 104%.
Homeostatic model of insulin resistance
HOMA-IR is closely associated with IR. Preoperative and postoperative fasting blood glucose and insulin concentration were measured. Insulin was measured by ELISA (Dako, Cambridge, UK). HOMA-IR was calculated by the formula: (fasting glucose × fasting insulin)/22.5. 16
Statistical analysis
All parameters were tested for normal distribution using the Kolmogorov–Smirnov test. Paired Student's t-tests were performed to evaluate the differences observed between the pre- and postsurgical values of the parameters studied. Correlations were studied using Pearson correlation and a value of P < 0.05 was taken as significant. The patient recruitment data are presented as mean ± 1 SD while baseline and postoperative changes in parameters are presented as mean ± SEM. All statistical analysis was carried out by the use of Analyse-It software, version 2.21 (Analyse-It, Leeds, UK).
Results
Patient demographics
BMI, body mass index
*Data presented as mean ± 1 SD
Parameters pre- and postbariatric surgery
BMI, body mass index; BP, blood pressure; ADMA, asymmetric dimethylarginine; NO, nitric oxide; HOMA-IR, homeostatic model of insulin resistance
Data presented as mean ± SEM
The average weight loss after the surgical intervention was 10 ± 0.7 kg (P < 0.0001).
MAP decreased by 11 ± 1 mmHg, systolic BP by 17 ± 2 mmHg and diastolic BP by 9 ± 1 mmHg (P < 0.0001). HOMA-IR decreased from 4.3 ± 0.5 to 2.1 ± 0.1 (P < 0.0001) after the surgery. Plasma ADMA concentration decreased by 24 ± 2% from 5 ± 0.4 to 4.0 ± 0.3 μmol/L (P < 0.0001), whereas plasma NO concentration increased by 15 ± 1% from 51 ± 2.6 to 60 ± 3 μmol/L (P < 0.0001).
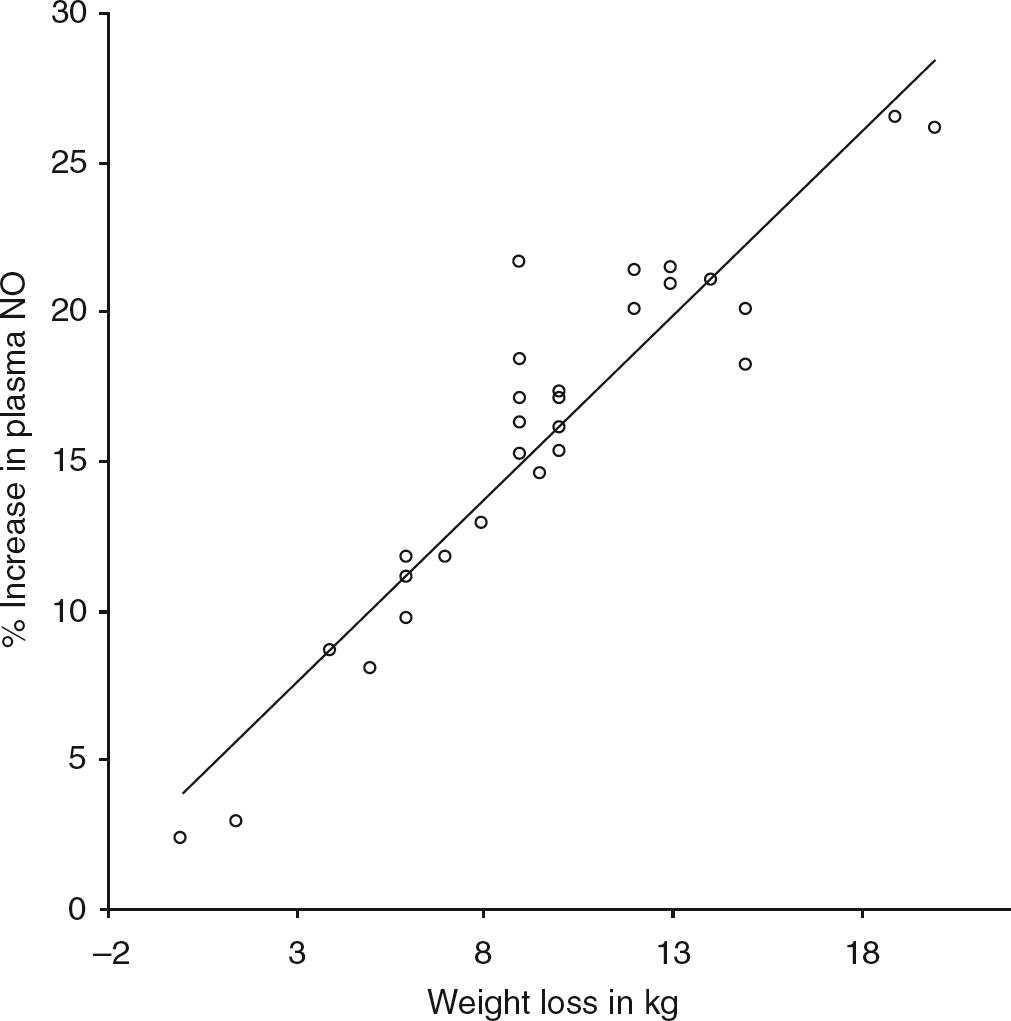
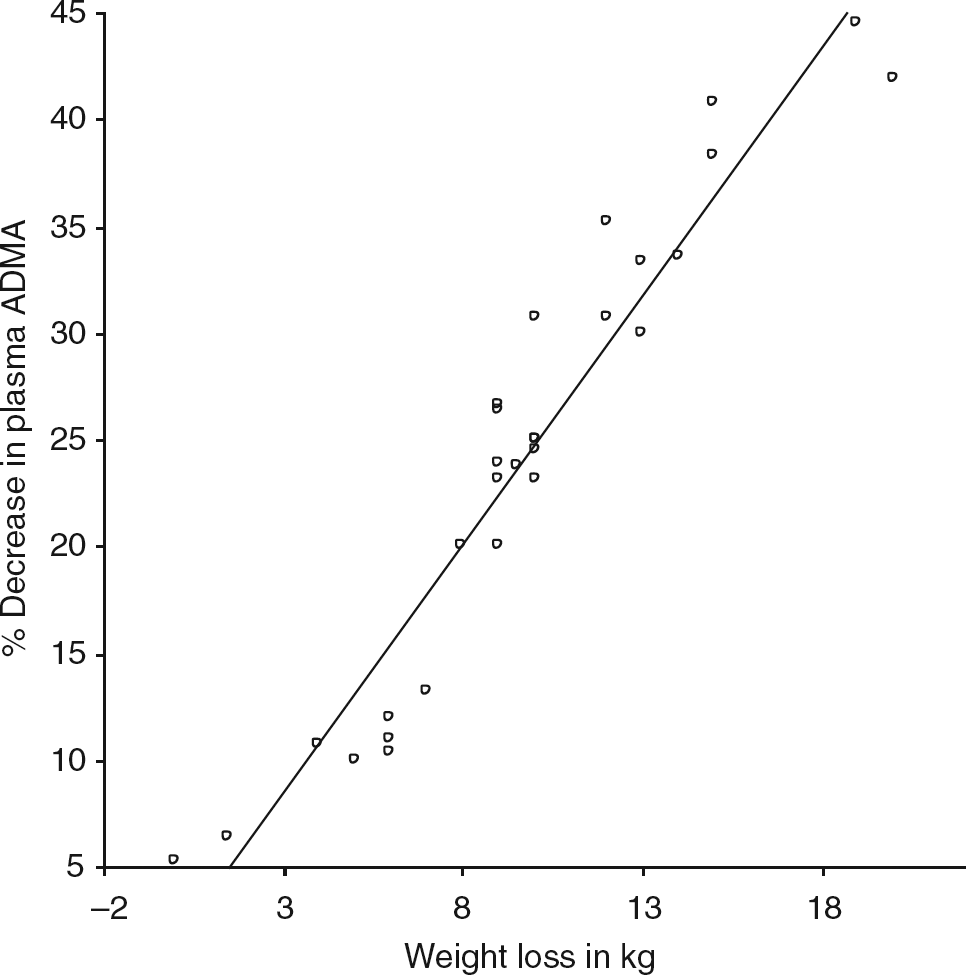
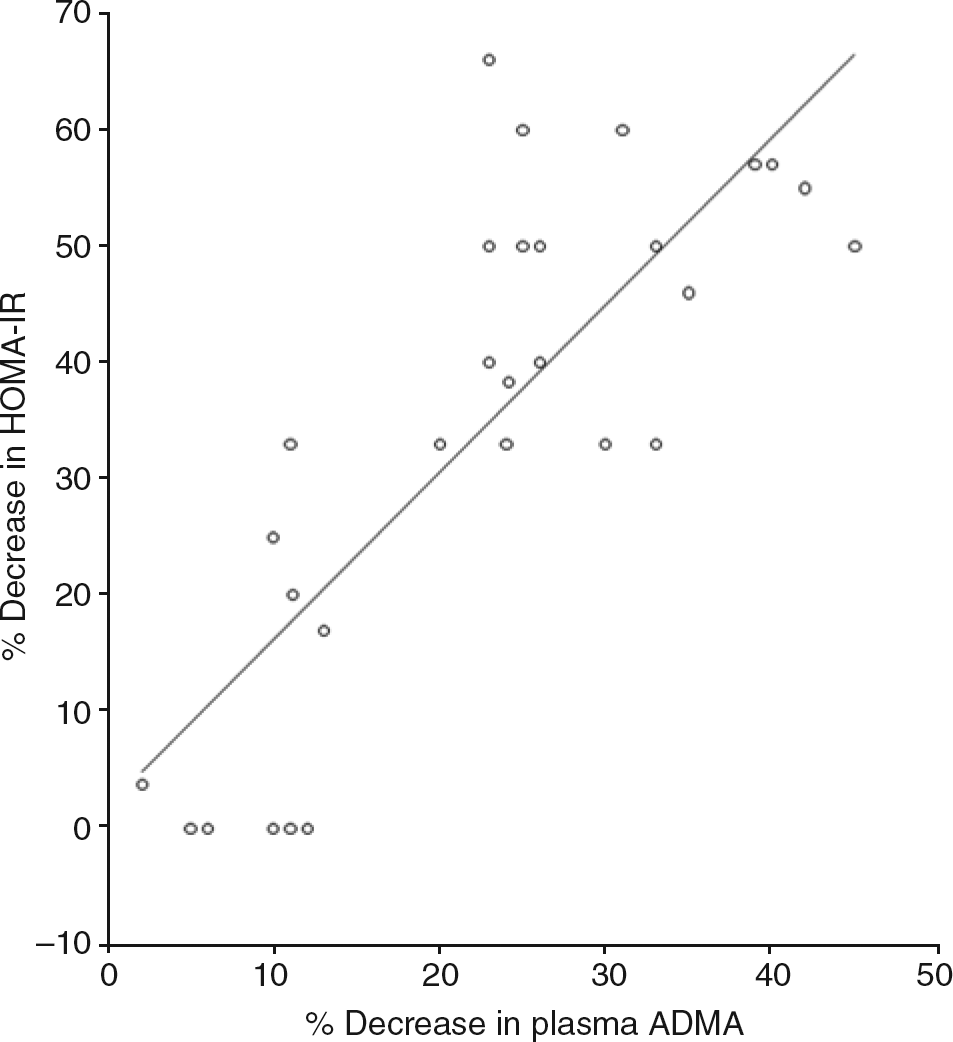
The study was not powered to detect differences between the three operations. Significant correlations were observed between the percentage decrease in plasma ADMA concentration with the percentage increase in plasma NO concentration (Figure 1) and weight loss (Figures 2 and 3). The decrease in HOMA-IR correlated with the decrease in ADMA (Figure 4) (P < 0.0001), but not the increase in plasma NO (r = 0.31, P = 0.09).
Correlation between decrease in plasma asymmetric dimethylarginine (ADMA) and rise in plasma nitric oxide (NO). 95% CI: 0.86, 0.96; r = 0.92; P < 0.0001 Correlation between weight loss and rise in plasma nitric oxide (NO). 95% CI: 0.41, 0.82; r = 0.67; P < 0.0001 Correlation of weight loss and plasma asymmetric dimethylarginine concentration (ADMA). 95% CI: 0.64, 0.90; r = 0.81; P < 0.0001 Correlation of percentage decrease in asymmetric dimethylarginine (ADMA) and homeostatic model of insulin resistance (HOMA-IR). 95% CI: 0.62, 0.9; r = 0.8; P < 0.0001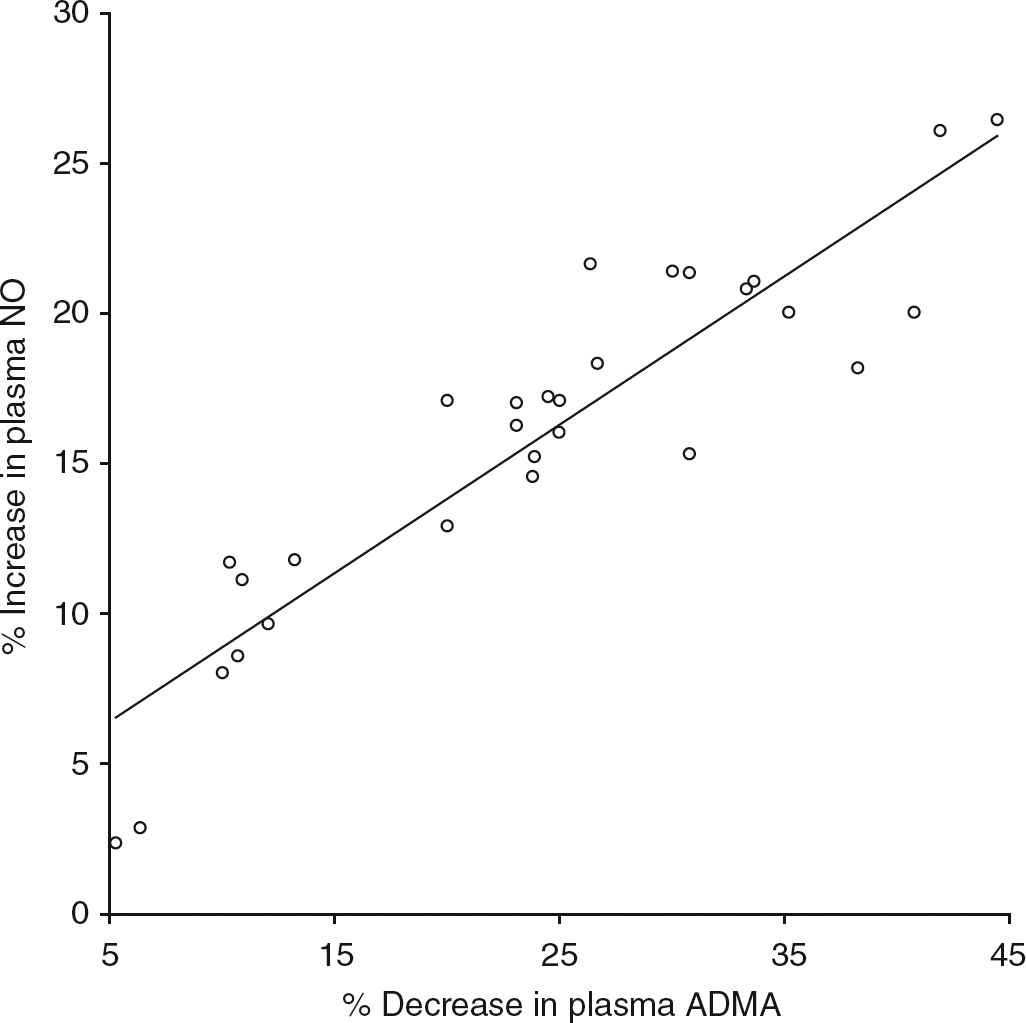
Pearson correlation between the observed changes in parameters with those of plasma NO and ADMA postbariatric surgery
ADMA, asymmetric dimethylarginine; NO, nitric oxide; BP, blood pressure
Discussion
We demonstrated a significant relationship between the decrease in ADMA and increase in plasma NO concentration in obese patients following bariatric surgery. ADMA may have been responsible for the low baseline concentration of NO by inhibiting eNOS activity in vascular endothelium in morbidly obese patients. The significant correlation between the decrease in ADMA and weight loss after bariatric surgery in our patients is in keeping with the findings of Krzyzanowska et al. 17 and is also consistent with the role of adipocytes producing ADMA. Although BP did decrease significantly, we were unable to confirm our hypothesis that changes in ADMA and NO were associated with the changes in BP.
In 1996, Steinberg et al. 18 demonstrated that obesity might have a negative impact on NO signalling, resulting in impaired endothelium-dependent vasodilation in these patients with metabolic dysfunction. Weight loss increased NO and improved arterial distensibility, indicating improved endothelial function. 19 Our patients also showed a significant decrease in HOMA-IR postbariatric surgery. The good correlation between the changes in HOMA-IR and ADMA in our patients agrees with previous findings suggesting plasma ADMA is an independent indicator of HOMA-IR. 20
The information about interaction between IR and the ADMA–NO axis in obesity-associated hypertension is limited. Inhibition of NOS impairs muscular microcirculation, thus blunting muscle glucose uptake in response to insulin.
21
On the other hand, induction of hypertension by the administration of the eNOS inhibitor NG-monomethyl-
Weight loss improves HOMA-IR and NO in the obese, but interactions with obesity-associated hypertension have never been tested. Studies in rats have shown a significant improvement in BP after removal of ADMA-induced NO suppression. 26 Our results, however, indicate that decreased suppression of NO synthesis induced by weight loss in obese patients did not correlate with decreases in MAP.
The mechanism of weight loss-induced decreases in BP is not clearly understood. In our study, the ADMA–NO axis does not appear to play a major part in reduction of BP. Insulin elevates the sympathetic nervous system activity that in turn augments the renin–angiotensin system (RAS), leading to sodium and water retention. 6 Sympathetic tone withdrawal after gastroplasty could also be one of the mechanisms responsible for the decrease in BP that is independent of the renin–aldosterone axis.
Bariatric surgery has been shown to decrease the concentrations of fasting insulin significantly, 27 but is also associated with reduction in intra-abdominal pressure. 28 Moreover, decreases in RAS activity could result in natriuresis and thus a reduction in BP in obese hypertensive subjects. 29 It is possible that the RAS could also be having a hypertensive effect by increasing plasma ADMA concentration and that angiotensin-converting enzyme inhibitors decrease ADMA in plasma. 30
Gastric bypass surgery has been shown to improve obesity-induced complications in which raised concentrations of ADMA have been implicated; these include hyperlipidaemia, IR, increased carotid artery intimal medial thickness and renal impairment in obese patients. 31 Although all our patients showed a decrease in systolic and diastolic BP, this did not correlate with the corresponding changes either in plasma NO or in ADMA concentrations. These results agree with the findings of Krzyzanowska et al., 17 where no significant correlation was noted between the changes in ADMA and BP after weight loss postvertical banded gastroplasty. There could be an indirect action of NO and ADMA in lowering the BP or improvement in BP during the initial six weeks postbariatric surgery could be due to mechanisms different from those involving endothelial function. However, it is difficult to dissociate the improvement in endothelial function from a role in long-term improvement in BP.
We acknowledge the fact that we did not verify the assay manufacturer's data in our laboratory. The samples were stored at −20°C within an hour of collection and analysed within nine months of collection. Nitrite and nitrate have been found to be stable in frozen plasma for up to one year, 32 whereas ADMA is stable for 72 months. 33
The limitations of our study are: first, the numbers of patients in each group were not enough to warrant subgroup analysis and second, the analysis of NO and ADMA was done at only one time point after the surgery (six weeks) and thus the dynamics of changes occurring after the surgery could not be studied.
In conclusion, we have confirmed the associations between the NO–ADMA axis with weight loss and improvement in IR. We know that there is a postoperative natriuresis and it is probable that the drivers for changes in BP are multiple, and that while the natriureis may be an important early key driver for reduction in BP, once the intravascular volume has reset itself, the changes in ADMA and NO could become more important.
DECLARATIONS
