Abstract
Background
Testosterone is measured for the investigation of female hyperandrogenism and male hypogonadism. Liquid chromatographytandem mass spectrometry (tandem MS) is becoming the method of choice but comprehensive reference ranges are lacking.
Methods
Testosterone was measured by tandem MS on 90 healthy women, 67 young healthy men and pregnant women (59 first trimester and 60 second trimester).
Results
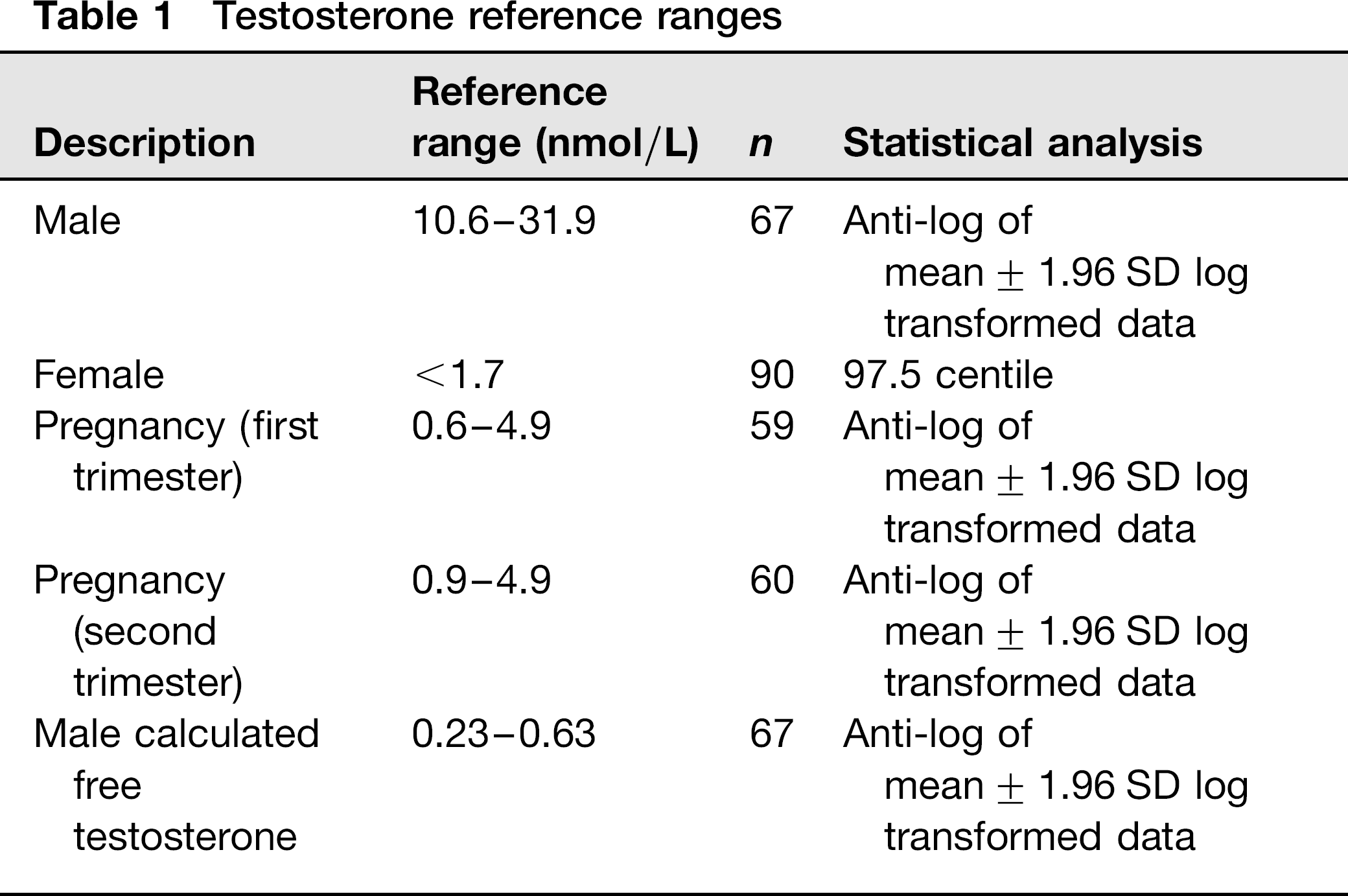
The male, male calculated free, first trimester and second trimester testosterone reference ranges (derived using the antilog of mean ± 1.96 SD of log transformed data) were 10.6-31.9, 0.23-0.63, 0.6-4.9 and 0.9-4.9 nmol/L, respectively. The female testosterone upper reference range limit, derived non-parametrically from the 97.5th centile, was < 1.7 nmol/L.
Conclusions
We have derived tandem MS testosterone reference ranges to support clinical services.
Introduction
Serum testosterone in adults is required for the investigation of hyperandrogenism in women 1 and hypogonadism in men. 2 Calculation of free testosterone may also be helpful in symptomatic men with a total testosterone between 8 and 12 nmol/L. 2 Although immunoassay is widely used by clinical laboratories to measure testosterone, there is interference and/or cross-reaction with structurally related steroids. This lack of specificity can result in marked differences in measured testosterone concentrations between immunoassay methods and constitutes a clinical risk. As a consequence, some laboratories now use more specific methods based on liquid chromatographytandem mass spectrometry (tandem MS). 3 While testosterone reference ranges for immunoassay are well established, there are fewer published for tandem MS.
Our aim was to derive reference ranges for testosterone measured by tandem MS for men, women and pregnant women. We have also derived a calculated free testosterone range in men.
Methods
Subjects
The female testosterone range was derived using serum from 90 healthy volunteer women with regular menstrual cycles (menses every 27-32 d), age range 16-45 y (mean age 29 y). Their healthy state was determined by history, physical examination and hormonal evaluation (thyroid function, prolactin and 17-hydroxyprogesterone). Subjects with signs of hirsutism or with a personal history of diabetes or hypertension, or a family history of polycystic ovarian syndrome (PCOS) were excluded.
Testosterone and calculated free testosterone ranges were determined using serum from 67 healthy Caucasian men aged between 20 and 40 y. 4 Patients were screened for diabetes using fasting plasma glucose. Those with dysglycaemia or significant abdominal obesity were excluded.
Pregnancy-associated ranges were derived from surplus anonymized serum samples from pregnant women attending the University Hospital of Wales for antenatal screening. These included 60 samples taken in the second trimester (15-18 weeks) and 59 in the first trimester (8-12 weeks). Serum was taken off the clot for the first trimester and from serum separator tubes for the second trimester.
Measurement of testosterone
Samples were prepared by protein crash. Briefly, 150 μL of a pre-prepared protein crash mixture was added to serum/ standards (100 μL). The protein crash solution for 100 samples contained 35 μL D3-testosterone (Sigma-Aldrich, St Louis, MO, USA; 98% purity), 5 mL of 0.3 mol/L Zn2S04 (Sigma-Aldrich) and 20 mL methanol (Fisher Scientific, Loughborough, UK; HPLC-grade). Samples were mixed and centrifuged. Supernatant (20 μL) was injected onto the liquid chromatographytandem mass spectrometry (LC-MS/MS) column. The standards were made up in-house and validated using gas chromatography mass spectrometry (GC-MS) targeted standards (Welsh External Quality Assurance Scheme [WEQAS] Laboratories, University Hospital of Wales, Cardiff, UK).
The LC-MS/MS instrument (Quattro™ Premier XE triple quadrupole tandem mass spectrometer; Waters Ltd, Watford, UK) was operated in positive ion electrospray mode (ESI+ve). Chromatographic separation was achieved using a Waters BEH C18 column (1.7 μm, 2.1 mm × 50 mm). The mobile phases were (A) water and (B) methanol both containing 2 mmol/L ammonium acetate (Sigma-Aldrich, >98%) and 0.1% formic acid (Fisher Scientific; 98/100%). The flow rate was set to 0.4 mL/min at 50% B which was increased to 98% B by 1.5 min. The retention time for testosterone and D3-testosterone was 1.23 min.
The instrument was run in multiple reaction monitoring mode with the testosterone (m/z 289.20) and D3-testosterone (m/z 292.20) both having daughter ions of m/z 97.20 and 109.10. The source probe temperature was 130°C, deso-lvation temperature 400°C, cone voltage 29 V, collision energy 21 eV and the desolvation nitrogen gas flow 3 L/h. The lower limit of reporting was 0.7 nmol/L (coefficient of variation = 7%). The interassay imprecision (n = 10) was 9.4% at 1.8 nmol/L, 6.7% at 17.8 nmol/L and 6.4% at 37.9 nmol/L. The agreement between tandem MS and a GC-MS reference method results was tandem MS = 1.0677GC-MS-0.1598, r2 = 0.9653 for female samples (n = 30) and tandem MS = 1.004GC-MS-0.0776, r2 = 0.9903 for male samples (n = 30).
Calculation of free testosterone
Sex hormone-binding globulin (SHBG) and albumin were measured using Abbott Architect™ (Abbott Diagnostics, Maidenhead, UK). Between-assay imprecision was 1.4% at 37 g/L albumin and 5.7% and 4.5% at 13.48 nmol/L and 49.41 nmol/L SHBG, respectively (n = 22). The Vermeulen equation was used to calculate free testosterone. 5
Statistical analysis
Data were analysed using SPSS 16 (SPSS Inc., Chicago, IL, USA).
Results
Data were not normally distributed, as assessed using the Kolmogorov-Smirnov test. The male testosterone, calculated free testosterone and pregnancy trimester-specific data-sets were normalized by log transformation. Reference ranges were then calculated from the anti-log of mean ± 1.96 SD of these log transformed data. Female testosterone data were not normalized by log transformation; the upper reference range was therefore derived non-parametrically using the 97.5 centile. Reference ranges are shown in Table 1.
Testosterone reference ranges
Discussion
High-quality method-specific reference ranges are required to support tandem MS testosterone methods used to provide clinical services. We report a testosterone reference range on a female population in whom PCOS has been carefully excluded. Our reference range in this population was found to be <1.7 nmol/L. This agrees well with that reported by Fanelli et al., 6 who have also used a carefully selected cohort of healthy women. In our laboratory, we had previously established a provisional reference range of < 1.9 nmol/L on surplus samples from 241 blood donors. Experience with this assay had led us to consider that this was too high. Since 5-10% of the population may have PCOS, a true female reference range should be derived in a population in which PCOS has been excluded. Haring et al., 7 have also reported a higher female testosterone reference range (<1.97 nmol/L) in a cohort where PCOS has not been rigorously excluded.
We have also established pregnancy-associated reference ranges and consider that these will be of use when it is necessary to investigate patients who develop signs of hyperandrogenism during pregnancy. The literature on pregnancy-associated testosterone reference ranges is limited and none, to our knowledge, derived using tandem MS. We were prompted to derive these ranges to assist in the investigation of a local patient with hyper-reactio luteinalis. PCOS has not been excluded in the pregnant cohorts used to derive our reference ranges; however, it is proportionally less common in a pregnant population (since they frequently have anovulatory subfertility) so less likely to be an important factor.
Finally, we have derived a male total testosterone reference range which agrees well with other recently published data7-8 and a reference range for calculated free testosterone. In summary, we report adult reference ranges for tandem MS measurement of testosterone to support clinical services.
