Abstract
Background
The worldwide epidemic of obesity is a major public health concern and is persuasively linked to the rising prevalence of diabetes and cardiovascular disease. Obesity is often associated with an abnormal lipoprotein profile, which may be partly negated by pioglitazone intervention, as this can influence the composition and oxidation characteristics of low-density lipoprotein (LDL). However, as pioglitazone's impact on these parameters within high-density lipoprotein (HDL), specifically HDL2&3, is absent from the literature, this study was performed to address this shortcoming.
Methods
Twenty men were randomized to placebo or pioglitazone (30 mg/day) for 12 weeks. HDL2&3 were isolated by rapidultracentrifugation. HDL2&3-cholesterol and phospholipid content were assessed by enzymatic assays and apolipoprotein AI (apoAI) content by single-radial immunodiffusion. HDL2&3 oxidation characteristics were assessed by monitoring conjugated diene production and paraoxonase-1 activity by spectrophotometric assays.
Results
Compared with the placebo group, pioglitazone influenced the composition and oxidation potential of HDL2&3. Specifically, total cholesterol (P < 0.05), phospholipid (P < 0.001) and apoAI (P < 0.001) were enriched within HDL2. Furthermore, the resistance of HDL2&3 to oxidation (P < 0.05) and the activity of paroxonase-1 were also increased (P < 0.001).
Conclusions
Overall, these findings indicate that pioglitazone treatment induced antiatherogenic changes within HDL2& 3 , which may help reduce the incidence of premature cardiovascular disease linked with obesity.
Introduction
The worldwide pandemic of obesity is a major public health concern and is strongly linked to the rising prevalence of diabetes and cardiovascular disease (CVD).1,2 Furthermore, the dyslipidaemia often found in association with obesity resembles that of type 2 diabetes, and is typically characterized by high levels of plasma triglycerides, mainly in very-low-density lipoproteins (VLDL). 3 The metabolic fate of these large, triglyceride-rich VLDL particles is shifted towards the production of small, dense low-density lipoproteins (LDL). 4 This has major implications for the processes of atherosclerosis, as the presence of small dense LDL has long been associated with increased cardiovascular risk. 5 In addition to increased levels of apolipoprotein B (apoB)-containing lipoproteins, obese individuals tend to have significantly lower levels of high-density lipoproteins (HDL), particularly HDL's larger subfraction, HDL2, which may be partly explained by increased adipocyte-derived cholesterol ester transfer protein. This enzyme can influence HDL remodelling and produce small, dense HDL3. 6 Additionally, HDL's major apolipoprotein, apoAI, is significantly reduced in the presence of obesity. 7 Obesity also affects other HDL-associated proteins, including paraoxonase-1 (PON-1).8,9 Normally, this enzyme functions to reduce oxidation products removed from LDL; 10 however, its activity may be reduced in the presence of obesity, meaning that LDL maintains a greater pro-oxidant load.8,9 Conversely, weight loss positively modulates the activity of this enzyme. 11
The dyslipidaemia of obesity can only be partly corrected by dietary or lifestyle interventions, thus suggesting that lipid-modifying therapies are appropriate in subjects at high risk. One such treatment, which may modulate the dyslipidaemia of obesity, is pioglitazone. Pioglitazone functions by acting as an insulin sensitizer, improving insulin sensitivity in skeletal muscle, hepatic and adipose tissues.12,13 Furthermore, pioglitazone also has lipid-modifying functions, as it can reduce serum triglycerides and small dense atherogenic LDL, while increasing serum HDL-cholesterol levels.12,14 This latter effect is thought to be due to a significant increase in HDL2-cholesterol. 15 Pioglitazone also limits malondialdehyde-LDL levels in humans 16 and can inhibit the oxidized-LDL up-regulation of matrix metalloproteinase-1 expression in vascular smooth muscle cells; 17 furthermore, pioglitazone decreases oxidized LDL. 18 However, to the authors’ knowledge, pioglitazone's influence on the oxidation potential of HDL2&3 is currently unknown.
Therefore, this pilot study aimed to examine if evidence-based interventions can be used earlier in the natural history of atherosclerosis, thus helping to prevent, delay or limit its development. Although the effects of pioglitazone on HDL have been characterized to some extent, no data currently exist to describe if compositional changes relate to the oxidation potential of HDL2&3. Therefore, this current study examined if pioglitazone functioned as a putative antioxidant towards HDL2&3. This was achieved by examining the oxidation potential of HDL2&3 by following the production of conjugated dienes and by examining the activity of HDL's main antioxidant enzyme PON-1.
Materials and methods
Study protocol
Men were recruited from a primary care setting by an invitation letter from their general physician. The sole inclusion criterion was a body mass index of ≥28 kg/m2. The exclusion criteria included clinical or electrocardiogram evidence of CVD, hypertension (>160/90mmHg), impaired glucose tolerance (fasting plasma glucose >7.0 mmol/L) or diabetes (all subjects had a fasting blood glucose measurement and two-hour oral glucose tolerance test), severe dyslipidaemia (total cholesterol >7 mmol/L; triglycerides >5 mmol/L), current or previous history of smoking, use of vasoactive drugs/vitamins, or a first-degree family history of CVD. A baseline assessment included anthropometric measurements and venous blood sampling. Participants were randomized (single-blinded) to either a placebo group (one tablet daily, n = 10) or a pioglitazone group (30 mg daily, n = 10). Participants adhered to their treatment regimen for 12 weeks and were asked to maintain their normal diets. At the end of the 12-week intervention, subjects attended the outpatient clinic for the same protocol as the baseline assessment. The 20 male volunteers successfully completed the study and compliance was assessed by returned tablet counting. The study was approved by the Queen's University Belfast Ethical Committee, and each subject gave written informed consent.
Blood sampling
Fasting blood samples were collected into either ethylene-diaminetetraacetic acid (EDTA) or serum collection tubes. Plasma and serum was recovered by low speed centrifugation at 1100 g maximum for 20 min in a Beckman J-6B centrifuge (Beckman Coulter, High Wycombe, UK), and stored as 2 mL aliquots at - 75°C.
Routine laboratory analyses
Fasting plasma glucose, insulin, glycated haemoglobin (HbA1c) and full lipid profile (total cholesterol, HDL, LDL and triglycerides) were assessed by routine methods in the Belfast Link Labs, Belfast Health & Social Care Trust. The HOMA-IR (homeostasis assessment of insulin resistance: a measure of insulin sensitivity) was calculated as (fasting plasma glucose [mmol/L] × fasting serum insulin [mU/L])/22.5. 19
Isolation, composition and oxidation potential of HDL2&3
HDL's two major subfractions, HDL2&3, were isolated from either EDTA-plasma or serum using rapid flotation ultracentrifugation. 20 The total cholesterol and phospholipid content of HDL2&3 were assessed using commercially available kits on an ILab600 Autoanalyser (Instrumentation Laboratory, Warrington, Cheshire, UK). The concentration of apoAI was assessed by single radial immunodiffusion.20–22 The arylesterase activity of PON-1 was assessed in serum and HDL2&3, isolated from serum, by monitoring the hydrolysis of phenyl acetate spectrophotometrically. 23
HDL2&3 oxidation
Small contaminating molecules which co-isolate with HDL2&3 were removed by subjecting the subfractions to size-exclusion chromatography on a pre-packed column of Sephadex G25 (PD10 Columns; GE Healthcare Life Sciences, Little Chalfont, Buckinghamshire, UK). Prior to oxidation and to allow for their standardization, the total protein concentration of HDL2&3 was assessed by the Bradford assay (Bio-Rad, Hemel Hempstead, Hertfordshire, UK). Purified HDL2 (50 μg/mL total protein) and HDL3 (200 μg/mL total protein) were then oxidized by 5 μmol/L copper chloride (final concentration). The production of conjugated dienes was followed spectrophotometrically at 234 nm in a thermostatically controlled (37°C) automated plate reader. The kinetic parameter time at half maximum (t1/2max), an equivalent of lag time, was evaluated using computer software (SoftMax Pro Version 4.8; Molecular Devices Corp., Wokingham, Berkshire, UK). 20
Statistical analysis
All results were assessed for skewness using histogram comparisons. Normally distributed paired samples were analysed using a paired t-test. Between-group analyses were assessed by an unpaired t-test when normally distributed, and by the Mann-Whitney U test when not normally distributed. Statistical tests were performed using the Statistics Package for Social Sciences (SPSS for Windows), v17 (SPSS Inc., Chicago, IL, USA). Results are presented as mean (standard deviation) for the normally distributed results and when not normally distributed as median (interquartile range). A P value less than 0.05 was considered significant.
Results
Subject physical and biochemical characteristics
A summary of the anthropometric and clinical measurements is given in Table 1. Participants in both groups were matched for all baseline parameters, with the exception that baseline glucose was lower in the subjects randomized to the pioglitazone group (P = 0.011). Baseline measurements of glucose homeostasis (glucose, insulin and HOMA-IR) were well within the normal ranges, indicating that none of the individuals had overt type 2 diabetes (Table 1). Pioglitazone intervention produced a beneficial effect to both systolic and diastolic blood pressure (pre versus post, P<0.05 for both comparisons). Although fasting plasma glucose was not affected by either treatment (P
Physical and biochemical characteristics of study cohort
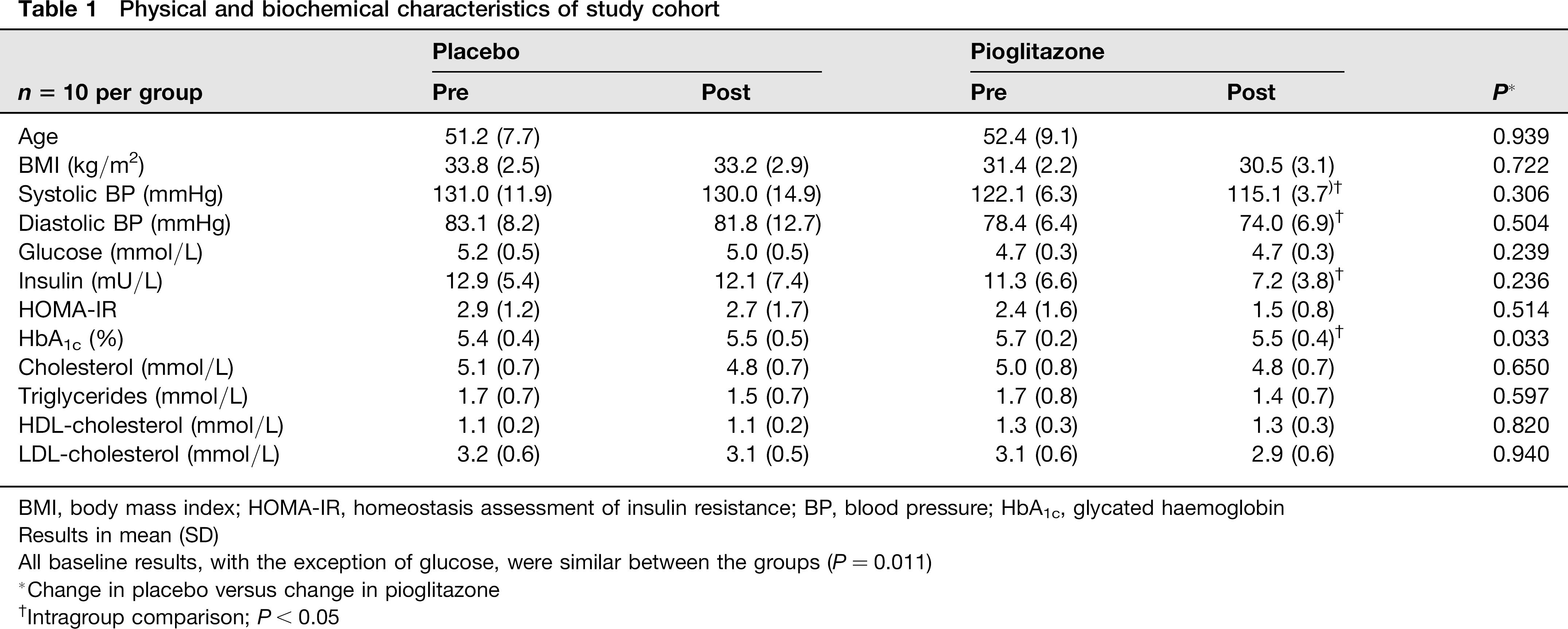
BMI, body mass index; HOMA-IR, homeostasis assessment of insulin resistance; BP, blood pressure; HbA1c, glycated haemoglobin Results in mean (SD)
All baseline results, with the exception of glucose, were similar between the groups (P = 0.011)
Change in placebo versus change in pioglitazone
Intragroup comparison; P< 0.05
Composition analyses of HDL2&3
Total cholesterol, phospholipid or apoAI were not affected by placebo intervention (pre versus post, P > 0.05 for all comparisons; Table 2). However, subjects randomized to the pioglitazone treatment exhibited significant increases in total cholesterol, phospholipid and apoAI within HDL2, while total cholesterol decreased and phospholipid and apoAI increased within HDL3 (pre versus post, P < 0.05 for all comparisons). Furthermore, intergroup comparisons between the placebo and pioglitazone groups supported the intragroup analyses (P < 0.05 for all cases; Table 2).
Compositional parameters for HDL2
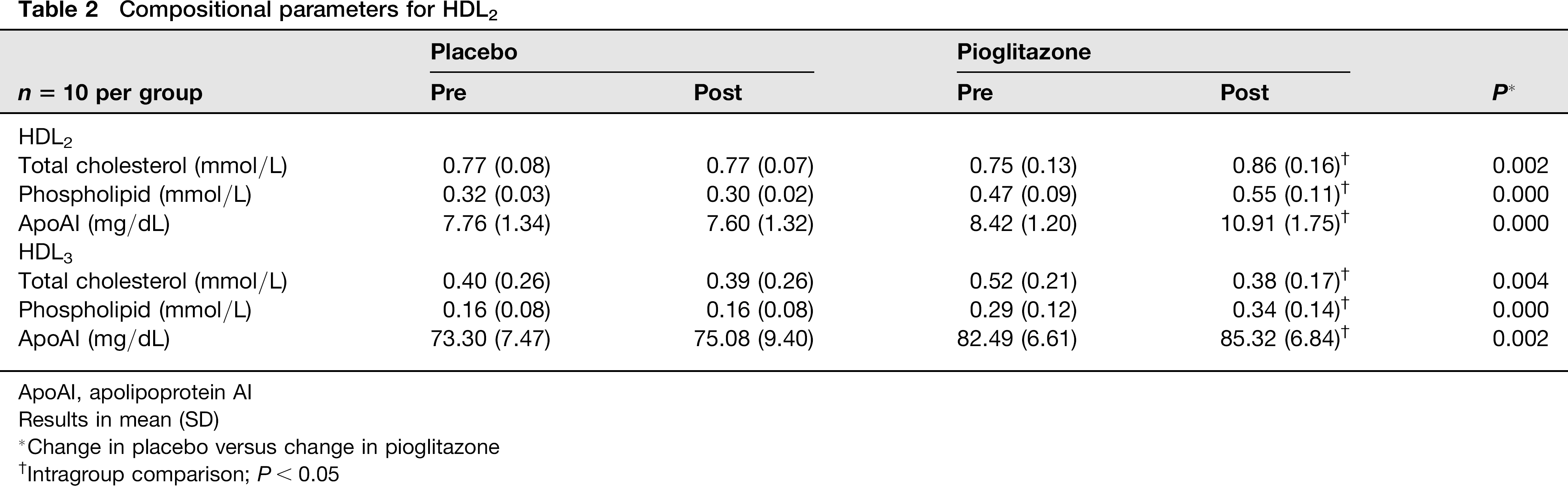
ApoAI, apolipoprotein AI
Results in mean (SD)
Change in placebo versus change in pioglitazone
Intragroup comparison; P< 0.05
Oxidation potential of HDL2&3
Baseline t1/2max was similar between the groups (P > 0.05). The oxidation potential of HDL2&3 was unchanged in the placebo group (Table 3). However, following pioglitazone treatment, HDL2&3 were protected against oxidation, showing an increase in t1/2max (pre versus post, P < 0.05 for both comparisons). Comparison between the groups identified that t1/2max increased in HDL2&3 in the pioglitazone group versus the placebo group (P < 0.05 for HDL2&3; Table 3).
Change in time at half maximum (t1/2max) and PON-1 activity for HDL2&3
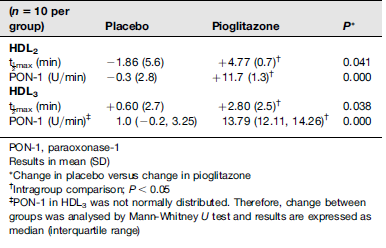
PON-1, paraoxonase-1
Results in mean (SD)
Change in placebo versus change in pioglitazone
Intragroup comparison; P <0.05
PON-1 in HDL3 was not normally distributed. Therefore, change between groups was analysed by Mann-Whitney U test and results are expressed as median (interquartile range)
The activity of PON-1 in HDL2&3 was unaffected by the placebo intervention (P > 0.05 for both; Table 3). However, following pioglitazone treatment, PON-1's activity was increased in both HDL2&3 (P < 0.05 for both comparisons). In addition, the increase in PON-1 activity in the pioglitazone group was maintained in the between-group comparisons (placebo versus pioglitazone, P < 0.001 for HDL2&3; Table 3).
Discussion
This study has shown for the first time that pioglitazone treatment significantly altered the composition and oxidation potential of HDL2&3 in an obese cohort. Obesity is often associated with low levels of HDL, which are in turn reflective of a general state of dyslipidaemia. This can be further complicated by insulin resistance - a condition which is typically managed through the use of drugs which modulate glucose homeostasis. The results presented here indicate that in addition to normalizing insulin sensitivity and blood pressure, pioglitazone may also augment the composition and the antiantioxidant capacity of HDL2&3, which would enhance their antiatherogenic potential.
Overall, pioglitazone treatment exerted its greatest effect towards HDL2, as intervention led to a significant increase in its lipid content and, therefore, size. This was supportive of the literature 15 and, by implication, suggests an improvement in reverse cholesterol transport and antiatherogenic potential. However, this increase in lipid occurred in the absence of any change to serum HDL-cholesterol. Although, this anomaly can be explained by the significant decrease in lipid within the HDL3 subfraction, which taken together would produce little or no change in serum HDL-cholesterol.
Pioglitazone treatment also augmented apoAI levels within HDL2&3, which would indicate enhanced apoAI transcription. This finding is again consistent with the literature, where pioglitazone increases apoAI transcription via a peroxisome proliferator-activated receptor-response in hepatocytes. 24 Furthermore, the increase of apoAI in HDL3, together with its decrease in lipid, would suggest that pioglitazone had stimulated a decrease in HDL3 size, together with an increase in particle number. This might also enhance reverse cholesterol transport, as these smaller HDL subfractions are involved in the early steps of reverse cholesterol transport, and a reduction in their number is reflective of a less efficient reverse cholesterol transport system. 25
Furthermore, this study has identified for the first time that following pioglitazone treatment, HDL2&3 displayed enhanced oxidative protection. This was shown by their increased resistance against copper-mediated oxidation and increased PON-1 activity.
This oxidative protection may be related to several processes. First, the increase in t1/2max in HDL2 may simply be reflective of its increased size, as larger lipoprotein subfractions are known to be more resistant to oxidation.26,27 However, as the t1/2max was also prolonged in HDL3, which was not enriched with lipid or increased in size, this explanation seems unlikely. Second, pioglitazone may have the ability to function as a chain-breaking antioxidant, similarly to, for example, lycopene and alpha-tocopherol.28–30 This suggestion is based on the fact that pioglitazone has a similar ring structure to that of alpha-tocopherol, which may allow it to become incorporated within HDL2&3. This would enable pioglitazone to donate a hydrogen atom during fatty acid oxidation, similarly to other hydrogen-donating antioxidant molecules. Unfortunately, we did not measure pioglitazone within HDL2&3, nor did we perform any in vitro experiments to assess its antioxidant properties. Third, the added protection against lipid peroxidation may simply be related to the increase in PON-1 activity, as the main function of this enzyme is to inactivate peroxidized lipid 10 to innocuous compounds. 31 Therefore, we suggest that one or both of these latter explanations seem likely; however, as there is little or no research in this area this would require further investigation.
Conclusion
This pilot study has shown for the first time that pioglitazone treatment influenced both the composition and oxidation potential of HDL2&3 in an obese population. In doing so, this drug may not only promote cholesterol efflux through the reverse cholesterol transport pathway, but may also protect the HDL against peroxidation in vivo, which would synergistically confer a significant cardioprotective effect. Furthermore, we suggest that these findings, together with pioglitazone's ability to normalize insulin sensitivity and reduce blood pressure, may be a useful treatment to reduce the likelihood of obese subjects from developing diabetes and its subsequent sequelae of CVD.
DECLARATIONS
Footnotes
Acknowledgements:
None.
