Abstract
Background
A failure of urine ammonium to increase during acidosis indicates impaired renal acidification, and the urinary ammonium concentration is therefore a useful investigation in determining the cause of a metabolic acidosis. However, urine ammonium measurements are not widely available in routine diagnostic laboratories. This has led to the use of urine anion or osmolar gaps, which are unsatisfactory as surrogates for urine ammonium measurement.
Methods
We evaluated the adaptation of two widely available automated plasma ammonium assays for measurement of urinary ammonium.
Results
Both assays showed good recovery and linearity in urine samples spiked with ammonium chloride, and acceptable precision. Urine ammonium concentrations estimated from urinary anion and osmolar gaps showed poor agreement with measured urine ammonium concentrations.
Conclusions
Direct urine ammonium measurements are easily performed with modern autoanalysers by simple adaptation of routine plasma ammonium assays. The use of urine anion and osmolar gaps should be abandoned where direct measurement is available.
Introduction
The main renal homeostatic response to a metabolic acidosis is an increase in acid excretion as urinary ammonium. 1 The measurement of urinary ammonium is therefore a useful investigation in determining the cause of a metabolic acidosis. To diagnose a renal tubular acidosis, it is necessary to establish that there is a defect in acid excretion. Although urine pH is widely used for this purpose, it is not always a reliable index of acid excretion, and the urine ammonium concentration is a more definitive measurement. 2,3
However, urine ammonium measurements are not widely available in routine diagnostic laboratories, probably because the published methods are manual and time-consuming. Because of this, the use of the urinary anion gap or urinary osmolar gap to estimate the urine ammonium concentration is advocated. 4 However, the urinary anion gap has been shown to be poorly correlated with urine ammonium concentration. 5,6 While the urine osmolar gap has shown a statistical correlation with ammonium concentration, 5,6 the agreement is poor in many individual cases, 5 making this method of estimating urinary ammonium unreliable in the clinical setting.
We therefore investigated whether routine automated plasma ammonium assays could be used for urinary ammonium measurement.
Methods
We measured urinary ammonium concentrations by adaptation of automated plasma ammonium assays on the Roche Modular (Roche Diagnostics, Auckland, New Zealand) and Abbott Architect ci8200 analyzers (Abbott Diagnostics, Auckland, New Zealand). Both Roche and Abbott analyzers use an enzymatic assay using glutamate dehydrogenase. Urine samples were diluted 1/200 in normal saline (Baxter Healthcare Pty Ltd, Old Toongabbie, NSW, Australia), and then assayed as for plasma ammonium samples. Urine Na+, K+, Cl−, glucose and urea were measured using a Roche Modular analyser and an Abbott Architect ci8200. Urine osmolality was measured using freezing-point depression with a Fiske Mark 3 osmometer (John Morris Scientific Pty Ltd, Auckland, New Zealand) and a John Morris Model 3320 osmometer.
The urine anion gap was calculated as: AG = Na+ + K+ − Cl–. The estimated ammonium concentration from the osmolar gap was calculated as estNH4 + = 0.5(osmolality − 2[Na+ + K+] − urea − glucose). All concentrations are expressed in mmol/L.
Results
Blank samples (normal saline) showed ammonium concentrations less than 0.5 mmol/L. Urine samples spiked with ammonium chloride showed good linearity up to 200 mmol/L (Figure 1a). Mean (±SD) recovery of added ammonium was 93.9% (±4.1%) on the Roche analyzer and 92.0% (±11.7%) on the Abbott platform. Analysis of samples assayed in duplicate showed a coefficient of variation of 8.7% (n = 15) for the Roche Modular and 4.7% (n = 12) for the Abbott Architect.
(a) Urine ammonium measurements on six urine samples spiked with ammonium chloride. Closed symbols: measurements on the Roche Modular; open symbols: measurements on the Abbott Architect. (b) Comparison of urine ammonium estimated by anion gap with measured urine ammonium. (c) Comparison of urine ammonium estimated by osmolar gap with measured urine ammonium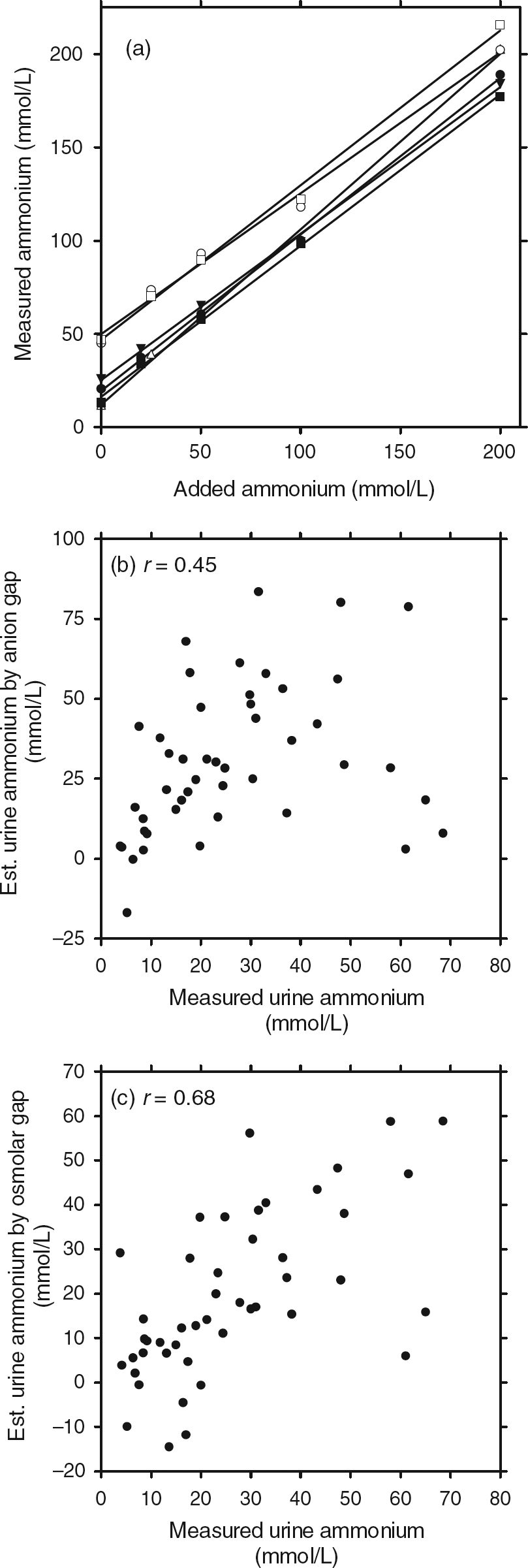
Stability studies showed that urine ammonium concentrations were stable in samples left at room temperature for up to 24 h and at 4°C for up to seven days. Significant increases in ammonium concentration occurred only in those samples where bacterial growth was apparent after more than two days at room temperature.
In 46 randomly selected urine samples, the urinary anion gap showed poor agreement with measured urine ammonium concentrations (r = 0.45; Figure 1b). Although the osmolar gap showed a positive correlation with the measured ammonium concentration (r = 0.68; Figure 1c), in only 27 out of 46 samples was the agreement within 50%.
Discussion
Many routine diagnostic laboratories do not offer urinary ammonium measurements, despite the fact that this is a useful parameter for the diagnosis of renal tubular acidosis. We demonstrate that measurement of urinary ammonium concentration is easily accomplished using two modern routine automated plasma ammonium assays, and it is likely that most routine enzymatic plasma ammonium assays could be adapted for urine ammonium measurement.
Preanalytical factors are an important source of error for plasma ammonium measurements, because plasma ammonium concentrations are low (micromolar range) and are therefore sensitive to contamination and to spurious elevations due to in vitro generation of ammonium from endogenous precursors such as glutamine. Because urine ammonium concentrations (millimolar range) are approximately 1000-fold higher than plasma concentrations, contamination is not a significant problem, nor is generation from precursors, as long as bacterial growth is prevented. The only significant preanalytical source of error for urine ammonium measurement appears to be bacterial growth with urea-splitting organisms, which is easily prevented by prompt delivery of samples to the laboratory and refrigeration if necessary.
In conclusion, urine anion and osmolar gaps are not accurate enough to be reliable surrogate measures, and should be abandoned in favour of direct urinary ammonium measurement.
DECLARATIONS
