Abstract
Background
Anti-thyroglobulin (Anti-Tg) assays show poor concordance.
Methods
We have investigated concordance and the causes of discordance between Abbott, Roche and Immulite Anti-Tg assays in 606 patients followed up for differentiated thyroid cancer (DTC). The reference range (RR) or lower reporting limit (LRL) was used to classify samples as negative or positive.
Results
Anti-Tg prevalence ranged between 6% and 55% depending on the method and cut-off. Concordance was 45% using LRL and 75% using RR. Specimens between the RR and LRL using the Immulite and Roche assays were identified that were positive by the Abbott assay and showed poor recovery of Tg in the Tg assay. This suggests misclassification using the RR. Anti-Tg International Reference Preparation (IRP) concentrations measured by the Roche and Abbott methods agreed well but patient samples did not. This is likely to be due to the heterogeneity of Anti-Tg. The Immulite assay appeared less sensitive than the Abbott and Roche based on investigations using the IRP and the low prevalence of Anti-Tg in the DTC patients (6–8%). Interference by Tg (>1000 μg/L) in the Roche assay was also identified as a cause of assay discordance.
Conclusions
Anti-Tg is used as a tumour marker for DTC and to predict interference in Tg assays themselves and hence inform clinicians of reported Tg concentrations. We have identified several causes of Anti-Tg assay discordance. This includes variation in assay sensitivity and interference from Tg, the heterogeneity of Anti-Tg and the use of different cut-offs to classify samples as antibody-positive or -negative.
Introduction
In recent years, there has been increasing recognition that serial quantitative thyroglobulin autoantibody (Anti-Tg) measurements are of use for the follow-up of differentiated thyroid cancer (DTC). 1,2 In addition, Anti-Tg is known to interfere in the measurement of thyroglobulin (Tg) by immunoassay and current guidelines recommend their measurement to alert the laboratory and clinicians to the risk of such interferences. 1,3,4 However, Anti-Tg assays are qualitatively and quantitatively different and are not interchangeable. 1,2,5–8 The prevalence of Anti-Tg varies with the method of analysis and cut-off used to classify samples as ‘positive’ or ‘negative’. 5,6,9 Discordance between Anti-Tg assays has been attributed to various factors including heterogeneity of Anti-Tg, differences in the specificity of circulating Anti-Tg for Tg antigen, Tg interference, differences in assay reagents (including the particular preparations of Tg employed) and the standards used by the various methods. 1,2,10
The aim of this study was to compare Anti-Tg assay performance in patients being followed up for DTC and to investigate the cause of any discordance found.
Materials and methods
Specimens
Anti-Tg was measured in 1030 sequential samples from patients being treated for DTC using the Abbott Architect (Abbott Laboratories, Abbott Park, IL, USA) and Roche E170 (Roche Diagnostics GmbH, Mannheim, Germany) assays following a change in laboratory methods. Of these, 606 were specimens from different patients with sufficient sample volume remaining in which to measure Anti-Tg using the Immulite Anti-Tg assay (Siemens AG, Erlangen, Germany).
Anti-thyroglobulin assays
The Abbott Architect Anti-Tg is a direct assay in which patient Anti-Tg binds to solid-phase Tg. After washing, human Anti-Tg is detected using acridinium-labelled anti-human IgG. The manufacturer's reference range (RR) is <4.11 kU/L. This was derived from 234 apparently healthy individuals with thyroid-stimulating hormone (TSH) within the RR. The manufacturer's stated sensitivity is ≤1 kU/L. Between-assay precision was 15.6% at 1 kU/L and 2.0% at 165 kU/L (n = 9). In our laboratory, we have adopted 2 kU/L as the lower reporting limit (LRL) for this assay.
The Roche E170 Anti-Tg is a competitive assay in which patient Anti-Tg competes for binding to solid-phase Tg with ruthenylated human monoclonal Anti-Tg. The manufacturer's RR is <115 kU/L. An in-house RR of <46 kU/L was derived from the 97.5 percentile of 115 biochemically euthyroid (TSH 0.5–2.0 mU/L) male subjects aged less than 30 years, with no previous abnormal thyroid function tests or obvious history of thyroid disease or medication likely to affect the thyroid axis. The laboratory RR rather than the manufacturer's RR was used for the purposes of this study. The LRL was 10 kU/L. Between-assay precision was 5.4% at 80.8 kU/L and 5.9% at 238.6 kU/L (n = 14).
The Immulite Anti-Tg is a direct assay in which patient Anti-Tg binds to Tg solid phase. Bound Anti-Tg is detected with enzyme-labelled anti-human IgG. The manufacturer's RR is 40 kU/L. This was derived from serum samples taken from 117 apparently healthy adults with no history of thyroid disease with normal TSH and free T4. The LRL was 20 kU/L. Between-assay precision was 7.6% at 35.5 kU/L and 7.7% at 562.3 kU/L (n = 12).
All three Anti-Tg assays were standardized against the International Reference Preparation (IRP) MRC 65/93.
Investigations involving international reference preparations
Human serum Anti-Tg first IRP NIBSC Code: 65/93 was reconstituted in water (1 mL). Dilutions were made in phosphate-buffered saline. Anti-Tg was measured as per the manufacturer's instructions.
To investigate the effect of Tg on measurement of Anti-Tg, a pool of Anti-Tg and Tg-free patient serum was derived from specimens with Anti-Tg <10 IU/mL (Roche) and Tg <0.5 μg/L. Tg IRP BCR457 was reconstituted in water (1 mL) and spiked into the Anti-Tg and Tg-free serum. Anti-Tg was measured as per the manufacturer's instructions.
Thyroglobulin and thyroglobulin recovery
Tg was measured using BRAHMS Tg-PluS DYNOtest immunometric assay (BRAHMS GmbH, Hennigsdorf, Germany). Test samples were set up in triplicate. Duplicate analysis was used to quantify Tg. Tg standard for recovery was added to the third tube. Samples with Tg <2 μg/L were spiked with 2 μg/L Tg standard, and samples with Tg > 2 μg/L were spiked with 100 μg/L Tg standard as per routine laboratory practice and Tg assay performed as per the manufacturer's instructions. Between-assay precision was 4.9% at 6.3 μg/L and 5% at 124.7 μg/L (n = 36).
The RR for Tg recovery was 74–115%. This was derived from 95% confidence intervals of 2251 Anti-Tg-negative (Roche Anti-Tg assay) serum samples. Recovery <74% indicates non-quantitative recovery due to assay interference.
Results
Prevalence of Anti-Tg in patients with differentiated thyroid cancer
Samples were classified as antibody-positive or -negative using either the reference range or lower reporting limit, N = 606 subjects
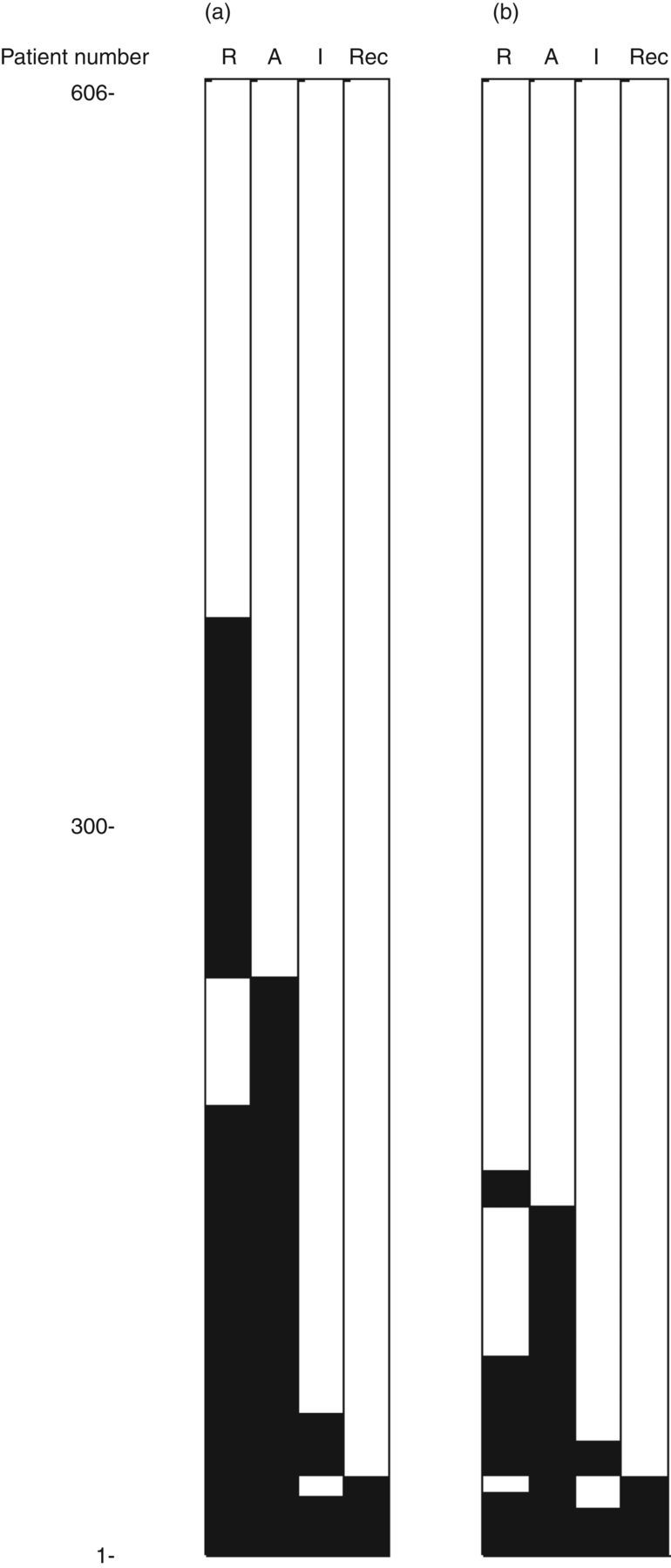
Concordance between Anti-Tg assays and Tg recovery. Roche (R), Abbott (A) and Immulite (I) Anti-Tg assays and recovery of Tg (Rec) in Brahms Dynotest Tg assay are shown. Positive and negative Anti-Tg are shown as black and white horizontal bars, respectively, for each consecutive patient 1–606. (a) Classification of samples based on lower limit of reporting: Roche <10, Abbott <2 and Immulite <20 kU/L. (b) Classification of samples based on reference range: Roche <46, Abbott <4 and Immulite <40 kU/L
All 16 specimens with Anti-Tg concentrations between the LRL and the RR using the Immulite assay were positive by the Roche and Abbott assays (using either LRL or RR) and five of these 16 specimens (31%) demonstrated poor recovery of Tg (Figure 1). Similarly, we identified six samples between the LRL and the RR of the Roche assay which were positive by the Abbott assay and had poor recovery of Tg in the Tg assay. It is noteworthy that we used our in-house RR for classification of antibody status for the Roche assay. Use of the manufacturer's RR would result in identification of a further seven samples negative by the Roche assay but positive by the Abbott and with poor recovery of Tg.
Measurement of Anti-Tg concentration in a serial dilution Anti-Tg reference preparation
Anti-thyroglobulin serum human 1st International Reference Preparation NIBSC Code: 65/93 was diluted 1:10 in phosphate-buffered saline and then further diluted as required. Anti-Tg measurement by Immulite was performed after 1:100 dilution in sample diluent as per the manufacturer's instructions
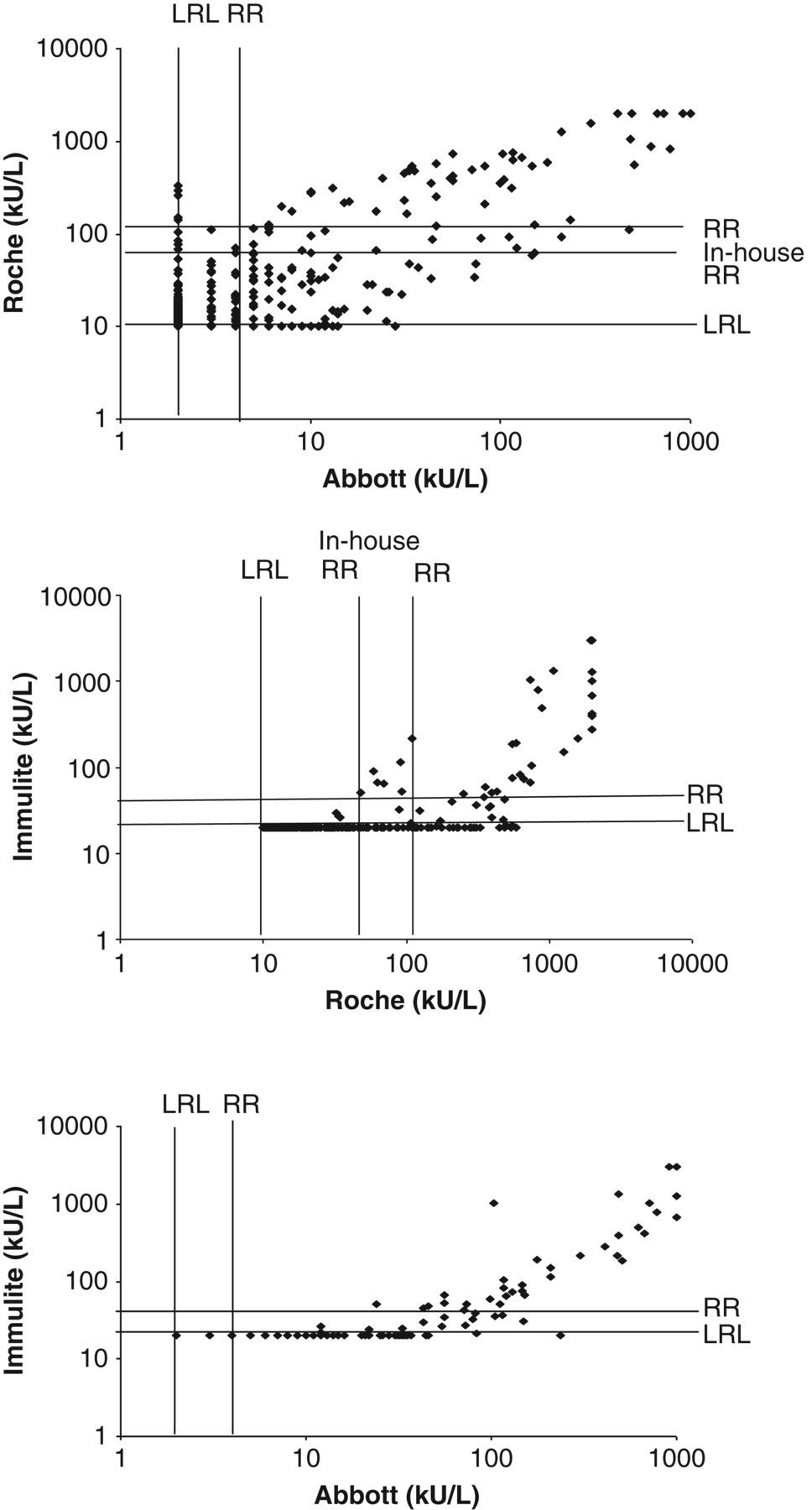
The relationship between Anti-Tg concentrations measured by different methods. The lower reporting limit (LRL) and manufacturers’ reference range (RR) are shown. An in-house reference range (in-house RR) is also shown for the Roche assay
Effect of Tg on measurement of Anti-Tg
Pooled serum negative for Anti-Tg and Tg (Tg-Ab <10 IU/mL by Roche and Tg <0.5 μg/L) was spiked with Tg reference material BCR457. Anti-Tg was measured in serial dilutions of the pool
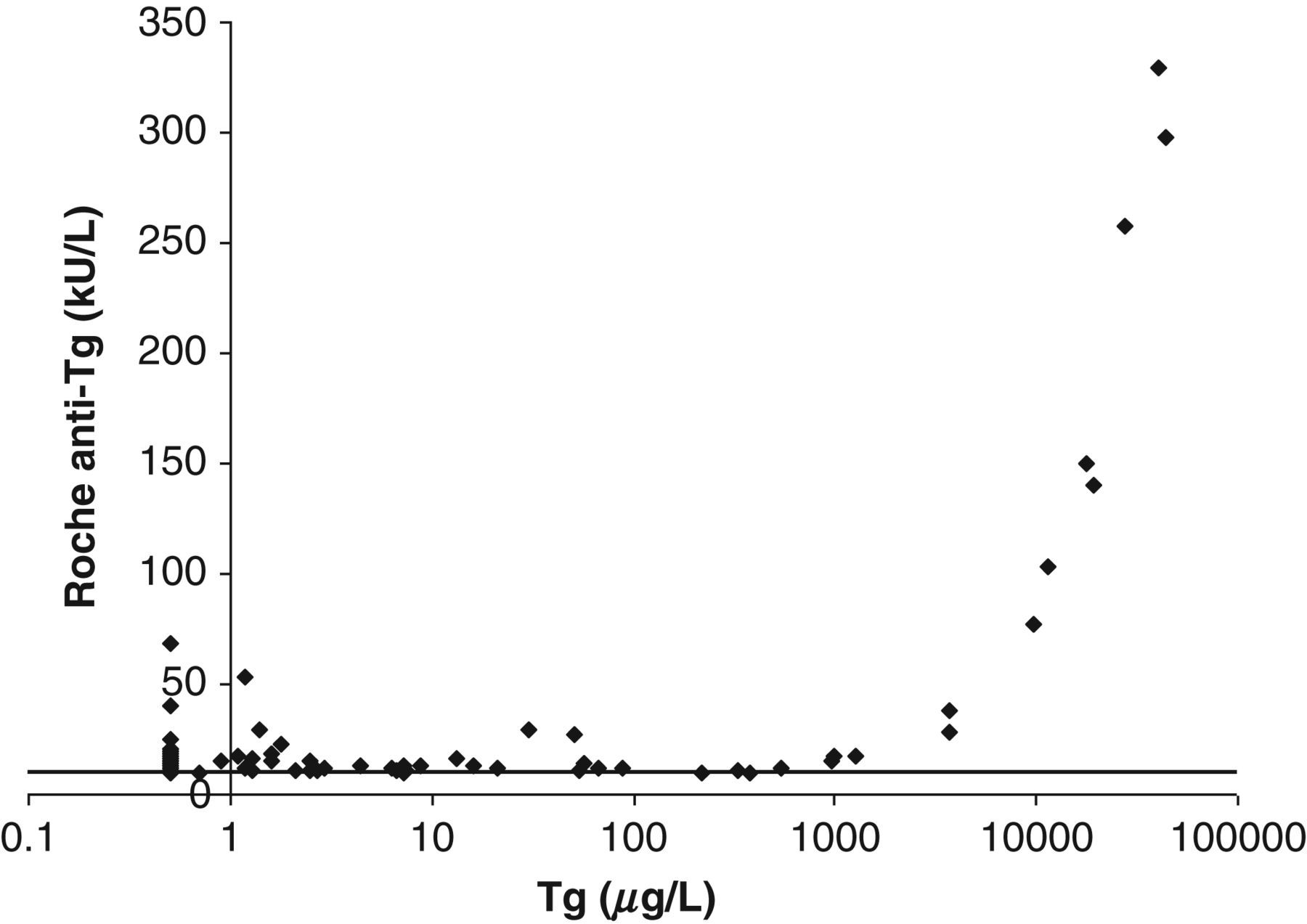
Relationship between Tg concentration and Anti-Tg concentration in the Roche assay. Data from samples which were Anti-Tg-positive by Roche but -negative using Abbott and Immulite assays are shown. Lower reporting limit was used to classify samples
Discussion
We have found significant differences in the prevalence of Anti-Tg (6–55%) in DTC patients depending on the assay and cut-off used. Prevalence of Anti-Tg positivity was particularly low using the Immulite assay (6–8% depending on whether LRL or the assay or manufacturer's cut-off were used to classify samples). This is significantly lower than the prevalence of 14–55% and 23–39% found using the Roche and Abbott Anti-Tg assays, respectively. Prevalence of Anti-Tg in patients followed up for DTC has previously been reported as 20–30%, 2 but it is acknowledged that the choice of cut-off, as well as the choice of method, affects the performance of the assay. 5,6
Spencer et al. 5 have recently reported that the use of a cut-off, which is designed to identify patients with autoimmune thyroid disease, rather than the detection limit, can cause misclassification of samples as antibody-negative. Our findings are in agreement. We identified a number of samples between the RR and the LRL of both the Immulite and the Roche Anti-Tg assays which were positive using one or more of the other assays and also demonstrated poor recovery of added Tg.
Although all three assays were standardized against the Anti-Tg IRP, concentrations of this material measured by the Immulite assay were approximately half of that expected. Measurement of Anti-Tg in serial dilution of the Anti-Tg IRP suggested that the Immulite assay was less sensitive than the Roche and Abbott Anti-Tg assays. However, it should be borne in mind that there is a potential for matrix effects on these experimental results as the analysis was performed in phosphate-buffered saline and not a serum matrix. Nevertheless, these findings, together with the lower prevalence of Anti-Tg found in DTC patients’ serum, suggest that Immulite assay is not as sensitive as the other assays tested and may account for some of the discordance between the methods. The Roche and Abbott assays appear to be very sensitive, measuring down to expected concentrations of IRP of 25 and 3 kUL, respectively, compared with 50 kU/L for the Immulite assay. This results in detection of high prevalence rates of 55% and 39%, respectively, of Anti-Tg in our DTC patients using the LRL. The significance of such high prevalence rates of Anti-Tg is unclear.
Despite the fact that Anti-Tg IRP concentrations measured by the Roche and Abbott methods agreed well with expected values and with one another, this was not the case for patients’ samples. Patients’ results were so discordant that it was not possible to use a correction factor to convert results from the old method to the new Roche or Abbott following the change in the laboratory method. Anti-Tg antibodies are not unique molecular entities, but rather mixtures of immunoglobulins that only have in common their ability to interact with Tg, and this heterogeneity in Anti-Tg is the likely cause of the discordance seen in the patients’ samples.
Tg interference in the Roche Anti-Tg assay was identified as a cause of discordance between the Anti-Tg assays tested. Positive interference by Tg in the Roche Anti-Tg assay was noted for patients’ samples with Tg greater than 1000 μg/L. This was confirmed by measurement of serial dilutions of the Tg IRP which showed a proportional relationship between Tg and apparent measured Anti-Tg. Anti-Tg assays are known to be prone to interference by high concentrations of Tg. 2,10
In summary, we have found a number of potential causes for discordance between Anti-Tg assays in our DTC patients’ samples. These include characteristics of the Anti-Tg assay such as assay sensitivity and the degree of interference from Tg, the chemical heterogenity of Anti-Tg and the particular cut-off selected to classify samples as antibody-positive or -negative. Measurement of Anti-Tg is used not only as a tumour marker for DTC but also to predict interference in Tg assays themselves and hence inform clinicians of reported Tg concentrations. However, it is clear that measurement of Anti-Tg is far from straightforward.
DECLARATIONS
