Abstract
Background
Cardiac troponin (cTn) has become the standard biomarker for the diagnosis of acute coronary syndromes. False-positive cTnI results have previously been reported on the Beckman Coulter analysers, which were shown to be random, not reproducible and occurred more commonly than expected. Our investigation ensued after a patient sample with an inordinately elevated cTnI was analysed, followed by a series of false-positive results being reported. The implications of falsely elevated cTnI results on patient care could be considerable.
Methods
Multiple experiments with patient sample pools with concentrations below the 99th percentile to extremely high (0.025, 15, 175 and 884 μg/L) were conducted in varying sequences of high and low samples on the Beckman Coulter Access2, UniCel DxI600 and UniCel DxI800 analysers.
Results
Our results demonstrate a significant increase in cTnI concentrations in the negative pool after analysis of high pool samples in various sequences. This increase is sufficient to cause elevations above the 99th percentile cut-off and false-positive cTnI results. These findings were reproducible on all three analysers.
Conclusions
Our study is highly suggestive of carryover and cTnI reagent pack contamination by the pipettors on the Access2, DxI600 and DxI800 analysers when patient samples with extremely high cTnI concentrations are analysed, leading to potential false-positive cTnI results on subsequent samples.
Introduction
Cardiac troponin (cTn) has become the standard biomarker for the diagnosis of myocardial infarction. 1,2 The universal definition of myocardial infarction requires a rise and/or fall of cTn with at least one result being above the 99th percentile cut-off. 2 False-positive cardiac troponin I (cTnI) results have previously been reported on the Beckman Coulter 3,4 and other analytical platforms. 5 These false-positive results are random, not reproducible and the root cause remains elusive despite intensive investigation. 4,6 Although cTn measurement does not independently facilitate the diagnosis of acute coronary syndromes (ACS), the potential ramifications of falsely elevated cTnI results on patient care are considerable, including prolonged emergency department stay, repeat laboratory testing and invasive cardiac investigations. The US Food and Drug Administration offers assistance on how this issue should be dealt with. 7 An incident of an inordinately high cTnI (2100 μg/L) from a patient with a fatal myocardial infarction being followed by a series of false-positive cTnIs led to our hypothesis of probe or reagent pack contamination.
Methods
Patient serum pools were prepared from remaining samples submitted to our laboratory for testing with varying cTnI concentrations: negative, below the 99th percentile URL (0.025 μg/L); 15; 175; and 884 μg/L. One patient serum with an inordinately high cTnI of 2100 μg/L was also included. Patient serum pools were prepared, divided into aliquots and stored frozen at −20°C until use. Transport of frozen serum pool samples between laboratories was on dry ice. All experiments were completed within a six-week period. Three levels of CardioImmune® TL quality control (QC) material (Microgenics Corporation, Fremont CA, USA) with mean concentrations of 0.045, 0.60 and 8.88 μg/L on the Access2 and 0.059, 0.78 and 11.4 μg/L on the DxI, were included at the beginning of each run to ensure acceptable analytical performance. The Beckman AccuTnI reagent packs are sufficient for 50 tests; thus QC material and pool samples were placed in aliquot tubes and presented to the analysers in batches consisting of 50 tests.
The protocol consisted of multiple experiments (please see Supplementary data at
To determine if carryover and reagent pack contamination was unique to cTnI or a more universal problem, similar experiments were conducted with other analytes measured by immunoassay on these analysers. Experiments with low- and high-serum pools were performed with human chorionic gonadotropin (HCG), thyroid-stimulating hormone (TSH) and carbohydrate antigen 19-9 (CA19-9) (please see Supplementary data at
Results
Repeated analysis of the negative pool (×47) showed no significant change in the cTnI concentrations throughout the run (mean 0.021 μg/L, SD 0.0062 μg/L). Analysis of a negative pool sample followed by 10 pool samples of 175 μg/L demonstrated an increase in measured cTnI concentrations of the negative pool run after each 10 sequential analyses of the 175 μg/L pool (see Figure 1). This increase was progressive, with higher values measured after each sequence of 10 175 μg/L pool samples and was sufficient to exceed the 99th percentile cut-off (0.04 μg/L), causing false-positive results. This was evident on all three analysers. When the negative pool was repeatedly analysed utilizing the remaining tests left in the reagent pack, the measured cTnI levels continued to increase. Replacing the reagent pack returned measured results of the negative pool to baseline. No demonstrable increase in cTnI concentration of the negative pool was seen performing the same experiment using the pool of 15 μg/L instead of the 175 μg/L pool (results not shown).
Results of the same negative pool analysed on the Access2 (blue diamonds), DxI600 (red squares) and DxI800 (green triangles) before and after analysis of sequences of 10 replicates of a high pool (175 μg/L) (grey arrows). The results demonstrate a gradual elevation of the measured cTnI of the negative pool, even when no further high pool samples are analysed. The dashed line indicates the 99th percentile cut-off. cTnI, cardiac troponin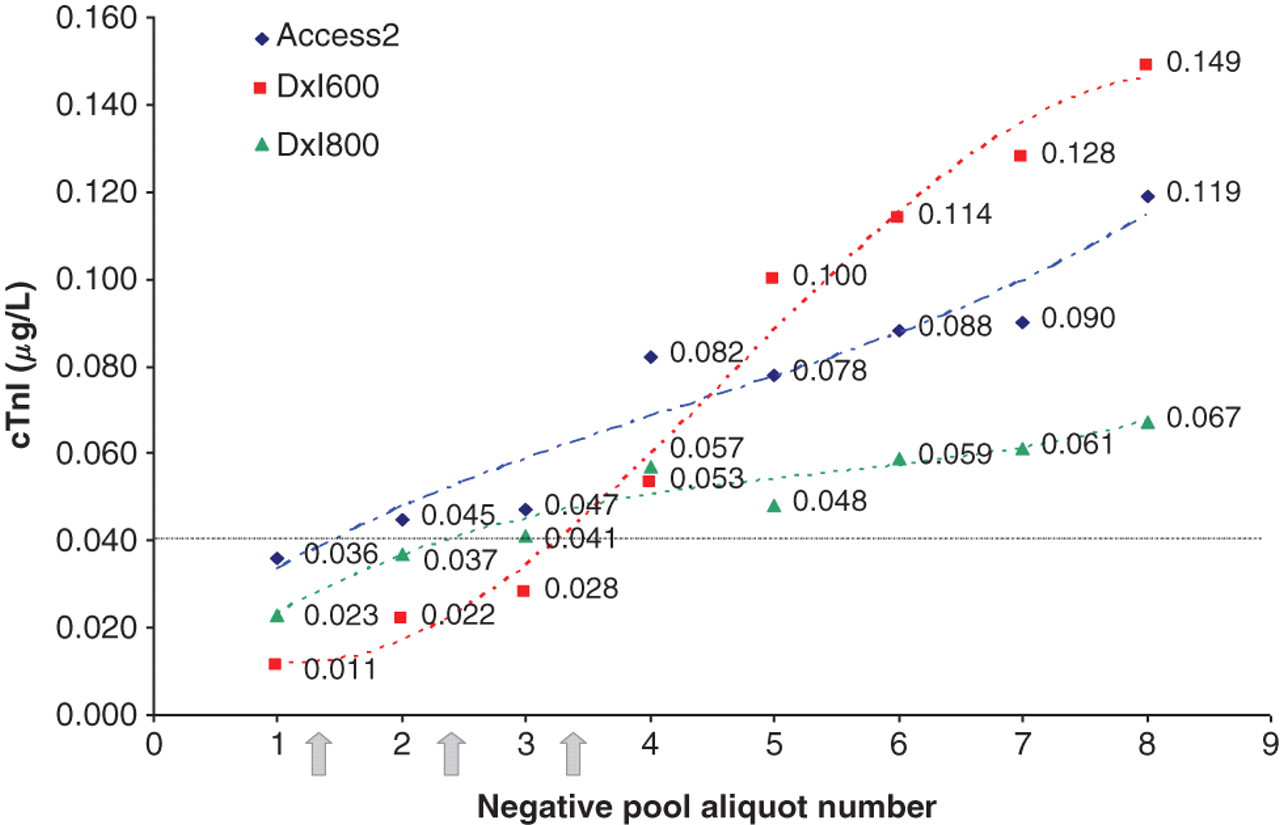
Analysis of an extremely elevated cTnI (884 μg/L) in triplicate after analysis of three QC and three negative pool samples, caused results of the negative pool to increase from a mean of 0.024 to 0.139 μg/L directly after analysis of the three 884 μg/L pool samples, decreasing thereafter, but remaining falsely elevated with a mean of 0.045 μg/L (see Figure 2 and Table 1). Analysis of the same negative pool with the same reagent pack (both stored sealed at 4°C) after overnight refrigeration, showed a further increase in measured cTnI from the mean of 0.045 μg/L after contamination to 0.126 μg/L.
Two experiments with the measured cTnI results of the negative pool plotted before and after a single patient sample with a cTnI of 2100 μg/L (blue) or 884 μg/L (green) in triplicate was analysed. Open arrows indicate when the elevated cTnI sample was analysed. Open points indicate results obtained on the same negative pool 24 h after analysis of the elevated cTnI sample, using the same reagent pack. The dashed line indicates the 99th percentile cut-off. Only results of the negative pool are shown. cTnI, cardiac troponin Mean results of the negative pool analysed on the DxI600 and DxI800 before and after analysis of the 884 g/L pool in triplicate, as well as after refrigeration of the reagent pack for 24 h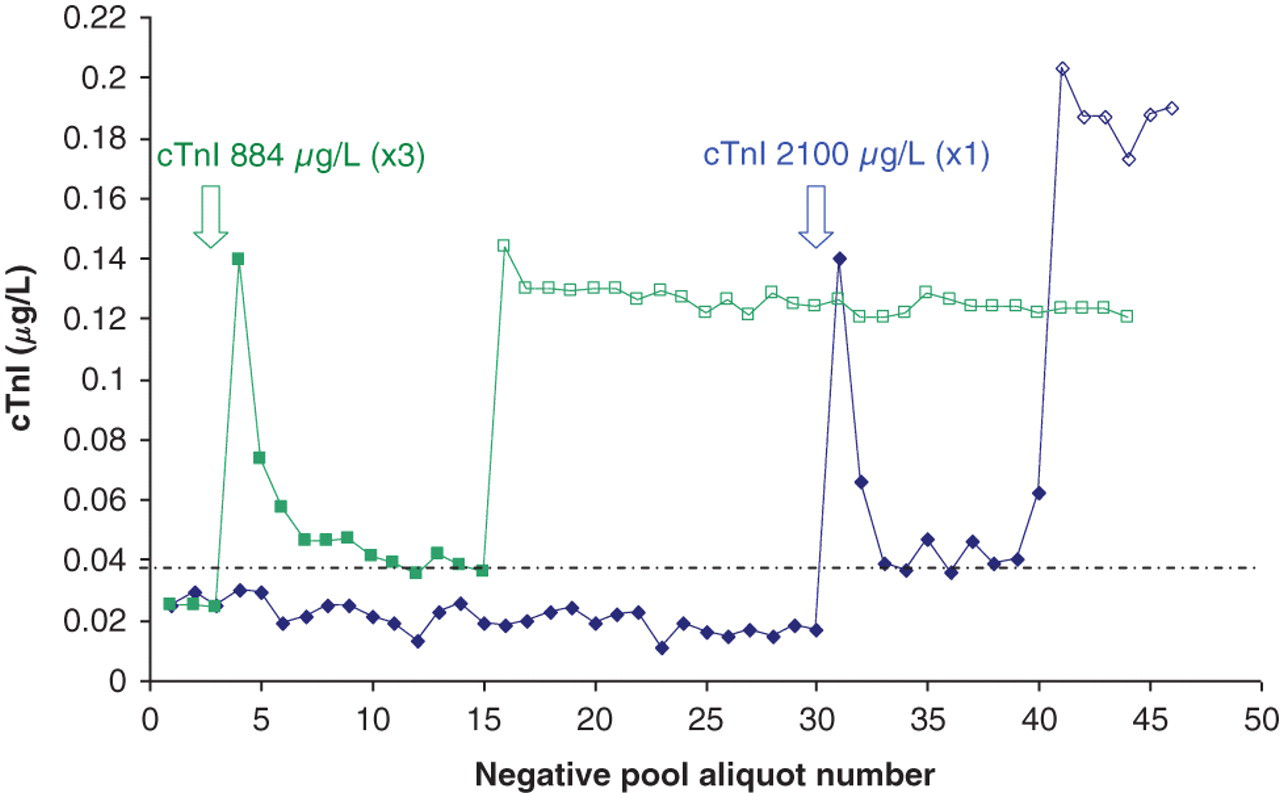
A single analysis of an inordinately elevated cTnI (2100 μg/L) followed by multiple negative pool samples demonstrated elevated results that followed the same pattern as the experiment using the 884 μg/L pool in triplicate. These elevated results were reproducible when the same pipettor that was used to aspirate the 2100 μg/L sample was specified, as well as when a different pipettor that had not come into contact with the 2100 μg/L sample was specified. The results were higher when the same pipettor was used, but both pipettors showed falsely elevated results. Even specifying a third pipettor on the DxI800 that was not used previously gave falsely elevated results. As before, when the reagent pack and negative pool used for this experiment were stored refrigerated overnight, and the negative pool was analysed the next day, higher cTnI results were obtained (see Figure 2).
Experiments with HCG, TSH and CA19-9 were also conducted. HCG showed a statistically significant increase of the negative pool (from a mean of 0.09–0.22 IU/L, P = 0.00002, Student's t-test) after analysis of a high pool five times. No statistical difference was shown for TSH or CA19-9 (P > 0.05).
Discussion
The AccuTnI assay is a heterogeneous enzymatic ‘sandwich’ immunoassay. On the Access2, DxI600 and DxI800, patient serum is transferred from the primary tube to a reaction vessel by a sample probe. The serum is then transferred to a second reaction vessel by the pipettor. The Access2 has a single sample/reagent pipettor, whereas the DxI600 has two, and the DxI800 has four sample/reagent pipettors which are selected at random by the analyser. Of note is that the same pipettor that aspirates patient sample is used to aspirate reagent. Following a wash, the pipettor transfers reagent, containing monoclonal anti-cTnI antibody (MAb) conjugated to alkaline phosphatase, as well as paramagnetic particles coated with MAb directed to another antigenic site on cTnI molecules, to the reaction vessel. After incubation, a magnetic field retains the paramagnetic particles with the attached ‘sandwich’ while unbound material is washed away. Addition of a chemiluminescent substrate initiates the generation of light, which is measured and directly related to cTnI concentration in the serum sample. 8 Reaction vessels are single use and disposable. Carryover is claimed to be <10 ppm. 9 Our study suggests that carryover is between 5–10 times greater.
In this study, analysis of a negative pool sample followed by 10 175 μg/L pool samples demonstrated a significant increase in the cTnI concentrations measured on the negative pool. This increase was progressive, with higher values measured after each sequence of 10 175 μg/L pool samples and was sufficient to exceed the 99th percentile cut-off (0.04 μg/L), causing false-positive results. The increase was not only a trend upwards, but also beyond what could be explained by analytical variation. When the negative pool was repeatedly analysed for the last five tests remaining in the pack, the measured cTnI levels consistently increased, instead of the postulated plateau at an elevated level.
Contamination near the end of the pack, when remaining reagent volume is low, is expected to have amplified effects when compared with contamination that occurs at the beginning of a pack. However, an unexpected finding was the further elevation of measured cTnI on the same negative pool and reagent pack after 24 h had elapsed. When we used half a reagent pack (25 tests) analysing QC and negative pool, then analysed the 884 μg/L pool in triplicate, followed by another 10 replicates of the negative pool, removed the pack from the analyser, sealed it, placed it in the refrigerator and measured negative pool samples until completion of the pack the following day, a significant increase in measured cTnI on the negative pool was noted (from a mean of 0.024 μg/L before contamination, to 0.055 μg/L after contamination, to 0.188 μg/L the following day) (see Table 1). We postulate that there is Ag–Ab complex formation in the reagent pack following contamination, which is time-dependent and reaches equilibrium after several hours. To allow a shorter time to obtain a result, the Beckman AccuTnI quantitates cTnI at a specified time after introduction of the Ag when equilibrium has not yet been reached.
Our experiments suggest that reagent pack contamination occurred, with the pipettor the only source of carryover. The first few results after the insult were higher than subsequent ones which may be explained by carryover from the contaminated pipettor to the next reaction vessel. However, different pipettors gave consistently elevated results on the negative pool, indicating that the reagent had been contaminated.
Our findings of carryover and reagent pack contamination did not extend to other immunoassays (HCG, TSH or CA19-9) performed on these analysers. Although HCG showed a statistically significant increase of the negative pool after analysis of the high pool five times, this was considered clinically insignificant as HCG results <5 IU/L are considered negative (when used as a pregnancy test). No statistical difference was shown for TSH or CA19-9.
Although the initial cTnI experiments with 10 175 μg/L samples followed by a negative sample are highly improbable in routine laboratory testing, later experiments with only a single extremely high sample demonstrated that false-positive results of samples analysed subsequently can occur. This may occur in the emergency department setting as demonstrated by the sentinel event, as well as in laboratories that service cardiac surgery wards. What is also concerning is that prolonged periods following contamination of the reagent pack will result in significantly greater elevations of measured cTnI. In smaller laboratories where a reagent pack of 50 tests may last a few days, this problem may become more evident than in larger laboratories where multiple reagent packs are used daily.
Potential solutions to the problem are complex. Obviously discarding the reagent pack once contaminated is mandatory, but prevention of the contamination in the first place is desirable. This may be achieved by using a more intensive pipettor wash (with ultrasonics) after each patient sample has been aspirated; consequently, a prolonged time to obtain an urgently required result would ensue. Alternatively, it could be specified that a different pipettor is used to aspirate patient sample and reagent; this may be feasible for the DxI600 and DxI800 with a resultant reduction in sample throughput capability, but the Access2 only has a single pipettor. Moreover, none of these strategies can be implemented by anyone other than the IVD manufacturer themselves. In the interim, what can be done? The measuring range of the analysers is 0.010–100 μg/L; thus, samples with results over 100 μg/L are reported as ‘>100 μg/L’. Not all laboratories routinely perform dilutions to obtain a final numerical result. Exactly at what level this contamination becomes significant needs to be determined, and we are currently investigating this. Although analysis of a high sample may cause contamination of the reagent pack, this will be more noticeable with potential false-positive results being reported when this contamination occurs near the end of a reagent pack, with little reagent remaining, than when a reagent pack is full. Furthermore, when the reagent pack is allowed to stand following contamination, a further increase in measured cTnI is evident. Should one always obtain a final result, and discard the remaining reagent once a result exceeds x μg/L? This may be feasible for a smaller laboratory, but for laboratories servicing cardiac hospitals, which may see multiple extremely high cTnI results daily, this becomes a tedious and inconvenient workaround. Troponin results are usually analysed urgently, with the results autovalidated and released to clinicians as soon as available. Should we hold results until manually validated, with all the potential pitfalls that this entails? Discussions with the manufacturer towards potential solutions are currently underway.
Conclusions
Due to the enormous analytical range of cTnI (0.01–100 μg/L), and the exquisite analytical sensitivity required at the 99th percentile cut-off to aid in the diagnosis of ACS, carryover from extremely elevated cTnI in patient samples may have dire consequences for patient samples that are analysed thereafter. Our study demonstrates a very concerning elevation of measured cTnI concentrations of a negative patient pool when patient samples with a very high cTnI are analysed, resulting in elevations above the 99th percentile cut-off and false-positive results. This was reproducable on all of the analysers we performed our experiments on. The pipettor, which aspirates sample as well as reagent, demonstrated carryover and as a result the reagent pack was contaminated. Viable solutions remain to be found, but in the interim, one should remain cognizant of this when samples with very high cTnI concentrations are analysed.
Declarations
