Abstract
Background
Asymptomatic variant haemoglobin is increasingly being found in the measurement of glycated haemoglobin (HbA1c) for the management of diabetes mellitus. We compared the HbA1c concentrations measured by high-performance liquid chromatography (HPLC) and immunoassay and glycated albumin (GA) concentrations and calculated the respective ratios in order to classify the variant haemoglobin.
Methods
Twenty different haemoglobin variants from 43 subjects were identified by mass spectrometry and DNA analysis. Since GA accurately reflects glycaemic control in patients with variant haemoglobin, we calculated respective ratios of HbA1c and GA. Haemoglobin variants causing a low ratio of HbA1c measured by HPLC (HPLC-HbA1c) to GA with a normal ratio of HbA1c measured by immunoassay (IA-HbA1c) to GA were classified as C1. A further classification of α and β was used with abnormalities of the α chain or β chain in the haemoglobin gene. Other haemoglobin variants were classified as non-C1. Eight diabetic patients with stable glycaemic control were used as controls.
Results
Twenty forms of variant haemoglobins were classified as C1α (2 variants; I-Interlaken and Hb J-Meerut), C1β (15 variants) and non-C1 (3 variants; Hb Himeji, Hb Woolwich, Hb Peterborough). Positive correlations between GA and HPLC-HbA1c or IA-HbA1c were seen in the C1β patients with diabetes mellitus. The regression line between GA and HPLC-HbA1c, but not that between GA and IA-HbA1c, showed a downward shift in comparison with the data obtained from the diabetic controls.
Conclusions
Variant haemoglobin could be classified by calculating the ratios of HPLC-HbA1c, IA-HbA1c and GA.
Introduction
Variant haemoglobin results from mutations of amino acids in haemoglobin. A total of 1128 variant forms have been reported worldwide as of December 2011. 1 About 20% of the variants produce clinical phenotypes such as haemolytic anaemia, polycythaemia and metohaemoglobinaemia, while the remaining 80% demonstrate no abnormal phenotype. Although variant forms of haemoglobin have traditionally been found in patients with haematological diseases, asymptomatic forms are often found in the measurement of glycated haemoglobin (HbA1c) for the management of diabetes mellitus. 2–4 Currently, high-performance liquid chromatography (HPLC) is most used to measure HbA1c, since the technique is more accurate and reliable than other methods. In patients with variant haemoglobin, their HbA1c concentrations measured by HPLC (HPLC-HbA1c) are higher or lower than the actual levels corresponding to glycaemia, leading to misjudgement of the glycaemic control status. In contrast, HbA1c measured by immunoassay (IA-HbA1c) usually is not influenced by variant forms of haemoglobin, and this method is recommended for distinguishing the majority of variant haemoglobin and making decisions about their glycaemic control state. 5 However, variant forms of haemoglobin in which the lifespan of red blood cells is shortened or glycation is increased or decreased have been reported. 6 With these variant haemoglobins, even IA-HbA1c sometimes yields abnormal results.
Glycated albumin (GA), another glycaemic control marker, is considered to accurately reflect glycaemic control in patients with variant haemoglobin. 7 Through calculation of the ratio of HPLC-HbA1c or IA-HbA1c to GA (HPLC-HbA1c/GA or IA-HbA1c/GA), it is possible to judge the discrepancy between HbA1c levels and glycaemic control state because the HbA1c/GA ratio is not influenced by glycaemic control state. 8,9 Furthermore, no classification schemes have been set for variant haemoglobin based on the reasons by which HbA1c is incorrectly measured. We therefore measured HPLC-HbA1c, IA-HbA1c and GA values in patients with variant haemoglobin and aimed to classify variant forms of haemoglobin using the HbA1c/GA results.
Subjects and methods
Subjects and controls
We investigated 20 different variant forms of haemoglobin from 43 subjects (Table 1). As previously reported, identification of variant haemoglobin was performed by electrospray ionization/mass spectrometry, tandem mass spectrometry and DNA analysis. 8 Eight patients with type 2 diabetes mellitus with stable glycaemic control were used as diabetic controls. All steps of this study, including DNA analysis, were approved by the Ethics Committees at Osaka Medical College and the study complied with the ethical guidelines of the Helsinki Declaration as revised in 2000. All subjects provided their written informed consent.
Variant haemoglobin specimens in the present study
*Number of patients with diabetes mellitus
Laboratory measurements
HbA1c was measured by HPLC (HPLC-HbA1c) using HA-8150 (Arkray Inc., Kyoto, Japan) 3 and by immunoassay (IA-HbA1c) using DCA-2000 (Bayer Diagnostics, Boston, MA, USA) 10 in these subjects. All these methods for HbA1c were corrected as a National Glycohemoglobin Standardization Program (NGSP) equivalent value (%) calculated using the formula HbA1c (%) = HbA1c (Japan Diabetes Society: JDS) (%) + 0.4%, considering the relational expression of HbA1c (JDS) (%) measured using previous Japanese standard substances and measurement methods and HbA1c (NGSP). 11 Serum GA was measured by HPLC. 12
Since GA accurately reflects glycaemic control in patients with variant haemoglobin, we calculated the respective ratios of GA and HbA1c and aimed to classify the variant forms of haemoglobin according to HbA1c/GA. Variant forms of haemoglobin demonstrating low HPLC-HbA1c/GA (<0.35) with normal IA-HbA1c/GA (0.3–0.5) were classified as C1. A further classification of α and β was used in order to differentiate between variant haemoglobin with abnormalities of the α chain and β chain in the haemoglobin gene. Other haemoglobin variants were classified as non-C1.
Statistical analysis
All data are shown as mean ± SD. For statistical analysis, unpaired Student's t-test was used to compare two groups. P values of <0.05 were considered to be statistically significant.
Results
According to the levels of HPLC-HbA1c, IA-HbA1c and GA, 20 variant forms of haemoglobin identified from 43 subjects in this study were classified as C1α (2 variant forms; Hb I-Interlaken and Hb J-Meerut), C1β (15 variant forms, n = 38) and non-C1 (3 variant forms; Hb Himeji, Hb K-Woolwich and Hb Peterborough) (Table 1). The majority of variant forms of haemoglobin were classified as C1β type. Among the 38 C1β patients, 10 patients (3 species) had diabetes mellitus while the other 28 patients (14 species) did not have diabetes mellitus.
We identified a positive correlation between GA and HPLC-HbA1c (R = 0.692, P = 0.057) or IA-HbA1c (R = 0.770, P = 0.025) in the diabetic controls (Figure 1). A positive correlation between GA and HPLC-HbA1c (R = 0.625, P = 0.053) or LA-HbA1c (R = 0.660, P = 0.038) was also seen in the C1β patients with diabetes mellitus. The regression line between GA and HPLC-HbA1c, but not GA and IA-HbA1c, showed a downward shift in comparison with the data obtained from the diabetic controls. A significant positive correlation between HPLC-HbA1c and IA-HbA1c was also seen in the diabetic controls (R = 0.972, P < 0.001) and the C1β patients with diabetes mellitus (R = 0.741, P = 0.014). The regression line between HPLC-HbA1c and IA-HbA1c also showed a downward shift in comparison with the data obtained from the diabetic controls.
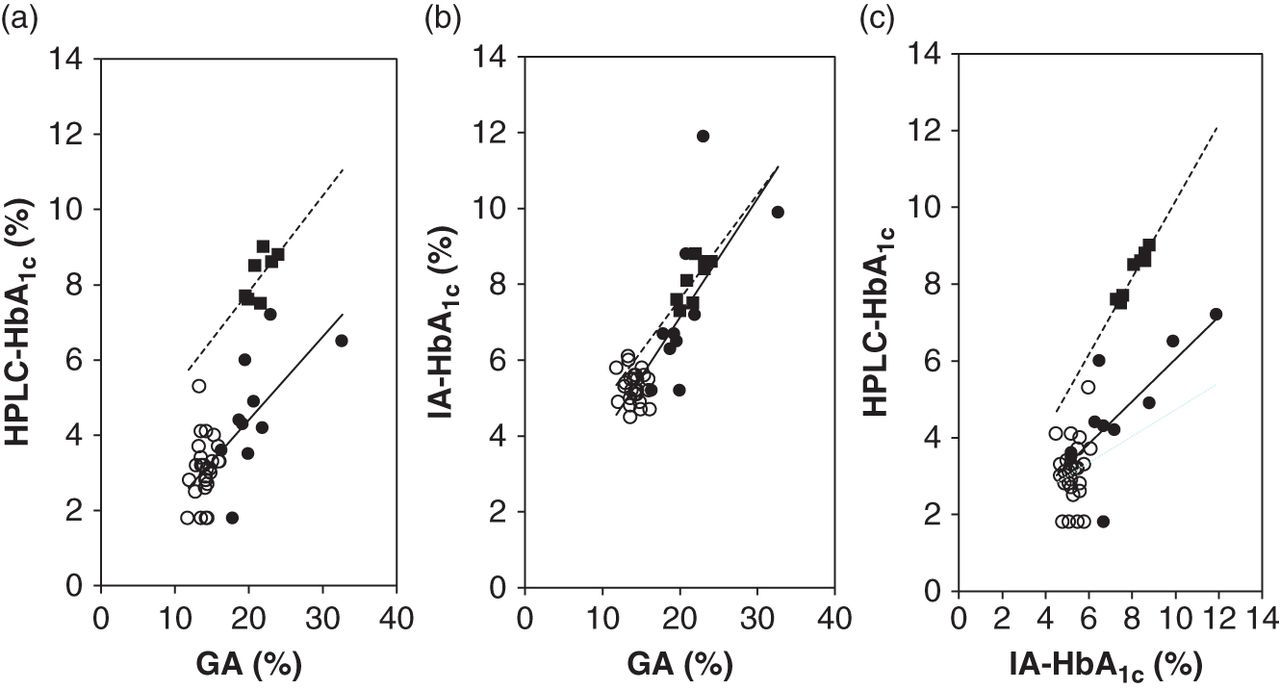
Correlation between GA and HbA1c in the C1β patients with variant haemoglobin with or without diabetes mellitus. The correlations between GA and HPLC-HbA1c (a), GA and IA-HbA1c (b), and IA-HbA1c and HPLC-HbA1c (c) in the C1β patients with variant haemoglobin with (closed circles) or without diabetes mellitus (open circles) and the diabetic controls (closed squares). The regression lines for the C1β patients with diabetes mellitus (straight line) and the diabetic controls (dotted line) are also shown. GA, glycated albumin; HbA1c, glycated haemoglobin; HPLC, high-performance liquid chromatography; HPLC-HbA1c, HbA1c measured by HPLC; IA-HbA1c, HbA1c measured by immunoassay
Table 2 presents the results obtained in the present study. Although GA and IA-HbA1c in the C1β patients with diabetes mellitus were not significantly different from the diabetic controls, their HPLC-HbA1c, HPLC-HbA1c/GA and HPLC-HbA1c/IA-HbA1c were significantly lower than the diabetic controls. GA, HPLC-HbA1c and IA-HbA1c concentrations in the C1β patients with diabetes mellitus were significantly higher than the C1β patients without diabetes mellitus, but the HPLC-HbA1c/GA and the HPLC-HbA1c/IA-HbA1c were not significantly different between both groups. The IA-HbA1c/GA in the C1β patients with or without diabetes mellitus was not significantly different from that in diabetic controls. The HPLC-HbA1c/GA and the HPLC-HbA1c/IA-HbA1c in the C1α patients were higher than that in the C1β patients. In the non-C1 patients, the HPLC-HbA1c/GA and the HPLC-HbA1c/IA-HbA1c were different from those in the controls. Furthermore, the IA-HbA1c/GA in the patient with Hb Himeji and in the patient with Hb Peterborough was also different from that in the controls.
GA values, HbA1c values measured by HPLC and immunoassay, and ratios of the three results in types of variant haemoglobin
GA, glycated albumin; HbA1c, glycated haemoglobin; HPLC, high-performance liquid chromatography; HPLC-HbA1c, HbA1c measured by HPLC; IA-HbA1c, HbA1c measured by immunoassay
*P < 0.001 versus control
**P < 0.001 versus C1β with diabetes mellitus
Discussion
We aimed to classify variant haemoglobin on the basis of the reasons by which HbA1c is measured as abnormal values. In patients with variant haemoglobin complicated with diabetes mellitus, because HbA1c is also affected by the glycaemic control state, evaluation of the HbA1c value is difficult. We therefore calculated the respective ratios of HPLC-HbA1c or IA-HbA1c and GA and were able to identify a C1 group and a non-C1 group of variant forms of haemoglobin by comparing these ratios.
The majorities of variant haemoglobin measured in the present study were classified as C1β type. Alpha chain variants cause less discrepant results from actual HbA1c values than β chain variants, because there are two genes (α1 and α2) for haemoglobin α chains. Two variant haemoglobins of C1α type were identified as Hb I-Interlaken 13,14 and Hb J-Meerut. 15
We could not subcategorize non-C1 type since there were only three patients identified as non-C1. However, the HbA1c/GA ratios of these patients with non-C1 type were different from each other. The HPLC-HbA1c in patients with Hb K-Woolwich was known to be extremely high because the variant haemoglobin overlaps with HbA1c on HPLC. 16 However, since HbA1c measured by immunoassay reflects their state of glycaemic control, the LA-HbA1c/GA was similar to that of the controls. Hb Himeji is reported to be increased glycation. 17,18 Therefore, IA-HbA1c in Hb Himeji was high, although HPLC-HbA1c was low. Hb Peterbrough is known to be unstable haemoglobin. 19 Therefore, IA-HbA1c in Hb Peterpough showed a low value. As a result, the non-C1 type of variant haemoglobin includes various forms and thus it is necessary to subclassify the non-C1 type in future after collecting data from many non-C1 patients.
Fructosamine has been previously used as a measure of glycaemic control in patients with variant haemoglobin. 5 However, as fructosamine is expressed as the concentration of glycated protein, its measured values are affected by serum protein concentration. With distorted serum protein concentrations, fructosamine cannot accurately reflect glycaemic control. 20 GA is considered superior to fructosamine as a clinical marker of glycaemic control, as this value is expressed as a ratio of glycated albumin to total (glycated and non-glycated) albumin. 7 The present study is the first to examine GA concentrations in patients with variant haemoglobin systematically.
Several diseases showing abnormal HbA1c values are known besides those involving variant haemoglobin. Haemolytic anaemia, liver cirrhosis, renal anaemia and iron deficiency anaemia under treatment show values that appear to be low, 21 while iron deficiency anaemia shows values that appear to be high. 22 Differentiating these diseases from variant haemoglobin is sometimes difficult. However, both HPLC and immunoassay yield similar HbA1c results in the former, whereas both assays yield divergent HbA1c results in all forms of variant haemoglobin (Table 2). As a result, variant haemoglobin and the other diseases can be differentiated in patients showing discrepancies between glycaemic control and HbA1c values by measuring HbA1c with HPLC and immunoassay and comparing both results.
In conclusion, variant forms of haemoglobin could be classified by measuring HbA1c by HPLC and immunoassay and GA, and comparing the respective ratios of the three results. Differential diagnosis can also be made between variant haemoglobin and other conditions yielding abnormal HbA1c values, using this approach.
DECLARATIONS
