Abstract
Background
When screening for macroprolactin, many laboratories use precipitation by polyethylene glycol (PEG) with molecular weight 6000 (PEG6000) or 8000 (PEG8000), and report the percentage prolactin recovery. It has been proposed that reporting of percentage prolactin recovery should be replaced by absolute post-PEG prolactin; however, the post-PEG prolactin reference interval has been established using PEG6000 only. We sought to determine whether the use of PEG8000, instead of PEG6000, changed post-PEG prolactin concentrations.
Methods
We compared the post-PEG6000 and post-PEG8000 prolactin concentrations in hyperprolactinaemic serum samples referred for macroprolactin screening (n = 40), using Passing–Bablok regression analysis and Bland–Altman difference plot.
Results
The median (25th–75th percentile, range) total prolactin, post-PEG6000 and post-PEG8000 prolactin concentrations were, respectively, 36 (31–46, 23–83) μg/L, 27 (20–38, 18–72) μg/L and 24 (18–35, 16–64) μg/L for male serum samples (n = 5); and 56 (39–83, 24–596) μg/L, 45 (31–67, 8–503) μg/L and 41 (28–62, 6–457) μg/L for female serum samples (n = 35) (mIU/L conversion factor: 21.2). The Passing–Bablok analysis demonstrated a significant constant bias of −1.27 and a non-significant proportional bias of 0.96. The Bland–Altman plot showed a bias of −8.2% (95% limits of agreement −19.3–2.9%).
Conclusions
There is a significant constant bias between the two macroprolactin precipitation methods. We changed our PEG precipitation to a PEG6000 method. Laboratories that use PEG8000 should consider the transference of the reference interval established with PEG6000 carefully.
Introduction
Prolactin is measured in the diagnosis and management of infertility and hypothalamic–pituitary disorders associated with hyperprolactinaemia. Current prolactin immunoassays detect all circulating forms of prolactin, including active monomeric prolactin (23 kDa, 60–90%), dimeric prolactin of unknown function (40–60 kDa, 15–30%) and macroprolactin – a biologically inactive protein complex consisting of various isoforms of prolactin and immunoglobulins (>100 kDa, 0–10%). 1 The extent of the macroprolactin detection is assay-dependent, varying from minimal detection in some immunoassays, to much higher levels of interference with other immunoassays. 1 Macroprolactin is detected in up to 20% of hyperprolactinaemic patients, resulting in falsely higher total prolactin concentrations that can lead to misdiagnosis and unnecessary interventions. 1,2 Many clinical laboratories have adopted macroprolactin screening procedures using precipitation with polyethylene glycol (PEG), an inert polymer that removes solvent from a solution, increasing the effective protein concentration until it exceeds protein solubility. 1 This procedure precipitates high molecular weight (MW) forms of prolactin (>50 kDa). Traditionally, the percent of the total prolactin recovered in the PEG-treated serum samples has been used as an estimate of the macroprolactin concentration. 1,3 PEG precipitation has several limitations, including co-precipitation of various amounts of monomeric prolactin, 3 and interference in several prolactin immunoassay methods. 4 The limitations of PEG precipitation, together with a common co-detection of excess monomeric prolactin and macroprolactin in the same serum samples, has led to recommendations to use method-specific prolactin reference intervals for PEG-treated serum samples. 2,5 Recently, post-PEG monomeric prolactin reference intervals have been established for six prolactin immunoassay platforms using PEG with the MW of 6000 (PEG6000). 4 However, our laboratory had used PEG with the MW of 8000 (PEG8000) for macroprolactin precipitation, as previously described. 2,3,6 The authors advocating the use of the post-PEG monomeric prolactin reference interval had used PEG6000 for the PEG precipitation. 4 We sought to determine whether the use of PEG8000, instead of PEG6000, changed post-PEG monomeric prolactin concentrations in our laboratory.
Methods
We compared the post-PEG6000 and post-PEG8000 monomeric prolactin concentrations in serum samples referred for macroprolactin screening to our laboratory reference centre between October 2010 and June 2011 (n = 40). These samples are derived by direct request from the physician, and are not generated by reflex testing of high prolactin results. Each specimen was split and simultaneously precipitated using 25% w/v PEG6000 or 25% w/v PEG8000. Equal volumes of the patient’s sample and PEG solution were vortex-mixed and incubated at room temperature for 10 minutes before being vortex-mixed again and centrifuged at 17,000
Results
PEG precipitation imprecision, as %CV, was 5.3% at 39 μg/L for PEG6000 and 8.0% at 37 μg/L for PEG8000. The median (25th–75th percentile, range) total prolactin, post-PEG6000 and post-PEG8000 monomeric prolactin concentrations were, respectively, 36 (31–46, 23–83) μg/L, 27 (20–38, 18–72) μg/L and 24 (18–35, 16–64) μg/L for male serum samples (n = 5); and 56 (39–83, 24–596) μg/L, 45 (31–67, 8–503) μg/L and 41 (28–62, 6–457) μg/L for female serum samples (n = 35). The Passing–Bablok regression analysis between PEG6000 and PEG8000 monomeric prolactin demonstrated a significant constant bias of −1.27 (−2.50 to −0.26) and a non-significant proportional bias of 0.96 (0.93–1.00) (both mean [95% confidence interval]) (Figure 1). The Bland–Altman difference plot showed a bias of −8.2 (−10.0 to −6.4)% (mean [95% confidence interval]) (Figure 1) with 95% limits of agreement −19.3 to 2.9% (not shown).
Passing–Bablok regression and Bland–Altman difference plot for the post-polyethylene glycol (PEG) prolactin concentrations. The percentage difference was calculated as post-PEG8000 prolactin subtract post-PEG6000 prolactin divided by the average of the post-PEG prolactin concentrations (mIU/L conversion factor: 21.2)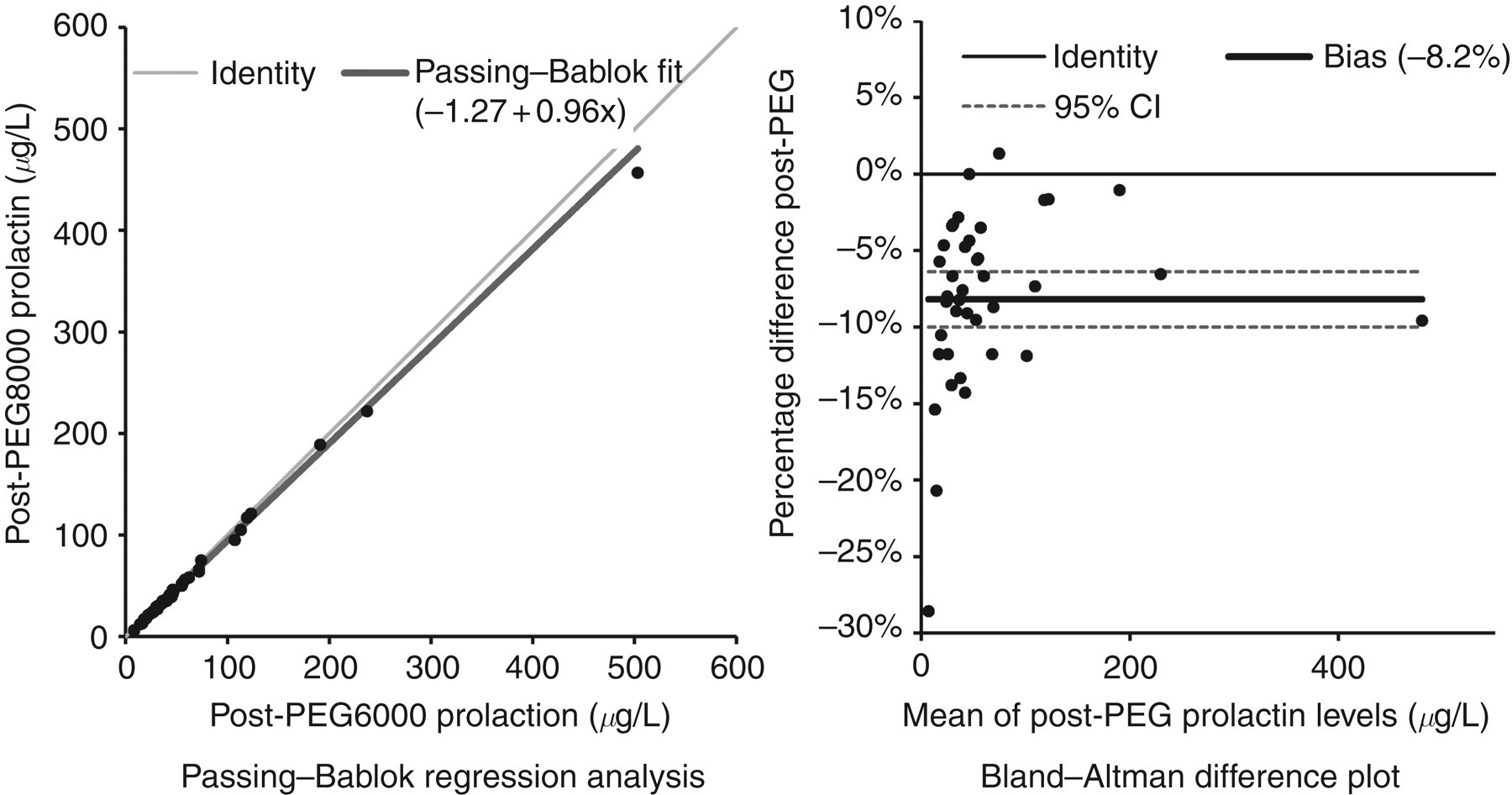
Conclusion
Our data suggest that there is a significant constant bias between post-PEG6000 and post-PEG8000 monomeric prolactin concentrations, which will affect the post-PEG monomeric prolactin reference interval. These findings have resulted in our laboratory changing to PEG6000 precipitation. There is no apparent advantage of the PEG8000 method over the PEG6000 method. We recommend that laboratories that use PEG8000 either adopt the PEG6000 precipitation method or carefully examine the transference of the reference interval established with PEG6000, 4 when reporting monomeric prolactin concentrations using post-PEG reference interval. This is an important clinical consideration if the presence of macroprolactin is based on absolute amounts of monomeric prolactin. 2,5
DECLARATIONS
