Abstract
We report a case of spuriously ‘normal’ haemoglobin A1c (HbA1c) result due to misidentification of HbG Taipei as HbAo by the Variant II built-in retention time algorithm. The defect was circumvented effectively by the implementation of a chromatographic system specific internal quality control mechanism for peak identity verification. HbA1c and estimated average glucose results were corrected from 4.7% to 8.2%, and 4.9 to 10.5 mmol/L, respectively. The results were consistent with the patient's concurrent and previous fasting blood glucose concentrations and existence of diabetes mellitus dermopathy, indicating poor glycaemic control. A review of currently available analytical systems showed that other than mass spectrometry, HbA1c measurements by these systems were generally affected by the presence of haemoglobin variants. The same haemoglobin variant may affect different analytical systems differently, resulting in the deviation of HbA1c results from the true value to different extents. Including the analytical principle of HbA1c measurement in the laboratory report can avoid inappropriate comparison of results obtained by different analytical systems. Moreover, since individual haemoglobinopathy may affect the degree of glucose binding to haemoglobin in a different way, this uncertainty limits the general application of same decision cut-off of established guidelines for glycaemic control monitoring. Adoption of an individualized monitoring system based on the critical difference or reference change value of HbA1c can be considered.
Introduction
The American Diabetes Association has recently recommended the use of haemoglobin A1c (HbA1c) not only as an indicator of glycaemic control in patients with diabetes mellitus, but also as a tool for screening and diagnosis of diabetes mellitus. The presence of haemoglobin variants can affect HbA1c results, depending on analytical methodology and associated technological shortcomings. Here, we report a case of a patient with diabetes mellitus with a spuriously ‘normal’ HbA1c result that resulted in inappropriate clinical management due to the presence of a haemoglobin variant and technological issues affecting the analytical system.
Case
A 75-year-old man with a history of type II diabetes mellitus (DM) presented to the cardiology centre on 4 March 2011 for regular follow-up. Laboratory tests revealed a fasting blood glucose of 9.6 mmol/L but apparently good glycaemic control in terms of a normal haemoglobin A1c (HbA1c) concentration of 4.7% (decision cut-off: <6.5%) and a normal estimated average glucose (eAG) of 4.9 mmol/L (reference range: 3.9–7.8 mmol/L). The patient showed skin changes in the lower limbs consistent with DM dermopathy despite repeatedly ‘normal’ HbA1c results as tested elsewhere. Owing to privacy issues, methods adopted for the HbA1c measurement elsewhere were not known. Review of our past laboratory records showed elevated fasting blood glucose concentrations of 6.7 and 10.6 mmol/L in July and November 2010, respectively, but without accompanying HbA1c results. The only previous HbA1c result determined by our laboratory was back in 2009 when the fasting blood glucose was 7.0 mmol/L with an agreeable concurrent HbA1c concentration of 6.1% and an eAG of 7.1 mmol/L.
Chromatogram assessment of the current 2011 sample (sample A) uncovered HbAo peak misidentification by the built-in retention time algorithm
1
of the VARIANT-II Hemoglobin Testing System (Bio-Rad Laboratories, Hercules, CA, USA) (Figure 1). This resulted in overestimation of the total peak area and consequent underestimation of the HbA1c in %. Manual recalculation after correct peak assignment allowed the HbA1c result to be revised from 4.7% to 8.2%, and eAG from 4.9 to 10.5 mmol/L. This was more consistent with the concurrent fasting blood glucose concentration of 9.6 mmol/L. Chromatogram assessment of the 2009 sample (sample B) showed correct peak identification, which explained the agreeable results of HbA1c in %, eAG and fasting blood glucose (Figure 1).
Chromatogram of 2011 sample (sample A) with wrongly identified Ao and (unknown) peaks and 2009 sample (sample B) with correctly identified Ao and E, D peaks by the VARIANT-II Hemoglobin Testing System. RT, retention time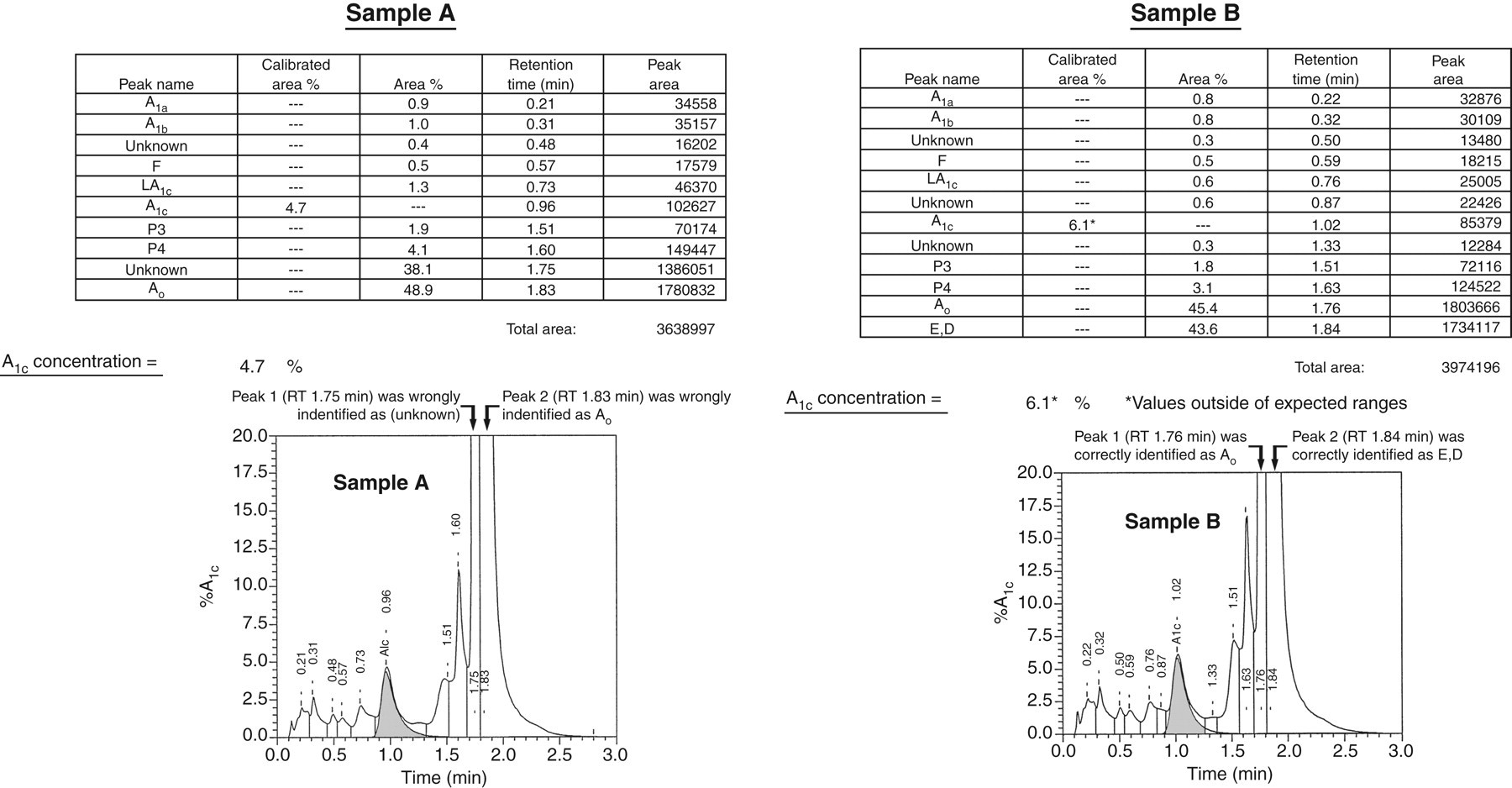
The (unknown) peak indicated the presence of haemoglobinopathy. The red cells of the patient were normochromic normocytic and peripheral blood smear examination was unremarkable. High-performance liquid chromatography (HPLC) by the β-Thalassemia Short Program of the VARIANT-II System showed a slow eluting haemoglobin variant at the HbA2 window with a retention time of 3.52 min that accounted for 37.8% of the total haemoglobin fraction (Figure 2). Direct sequencing of the β-globin gene was performed, showing that the patient was heterozygous for HbG Taipei (β-globin gene codon 22 GAA → GGA; Glu → Gly mutation).
Chromatogram of slow eluting haemoglobin variant peak at the haemoglobin A2 (HbA2) retention time window by the β-Thalassemia Short Program of the VARIANT-II System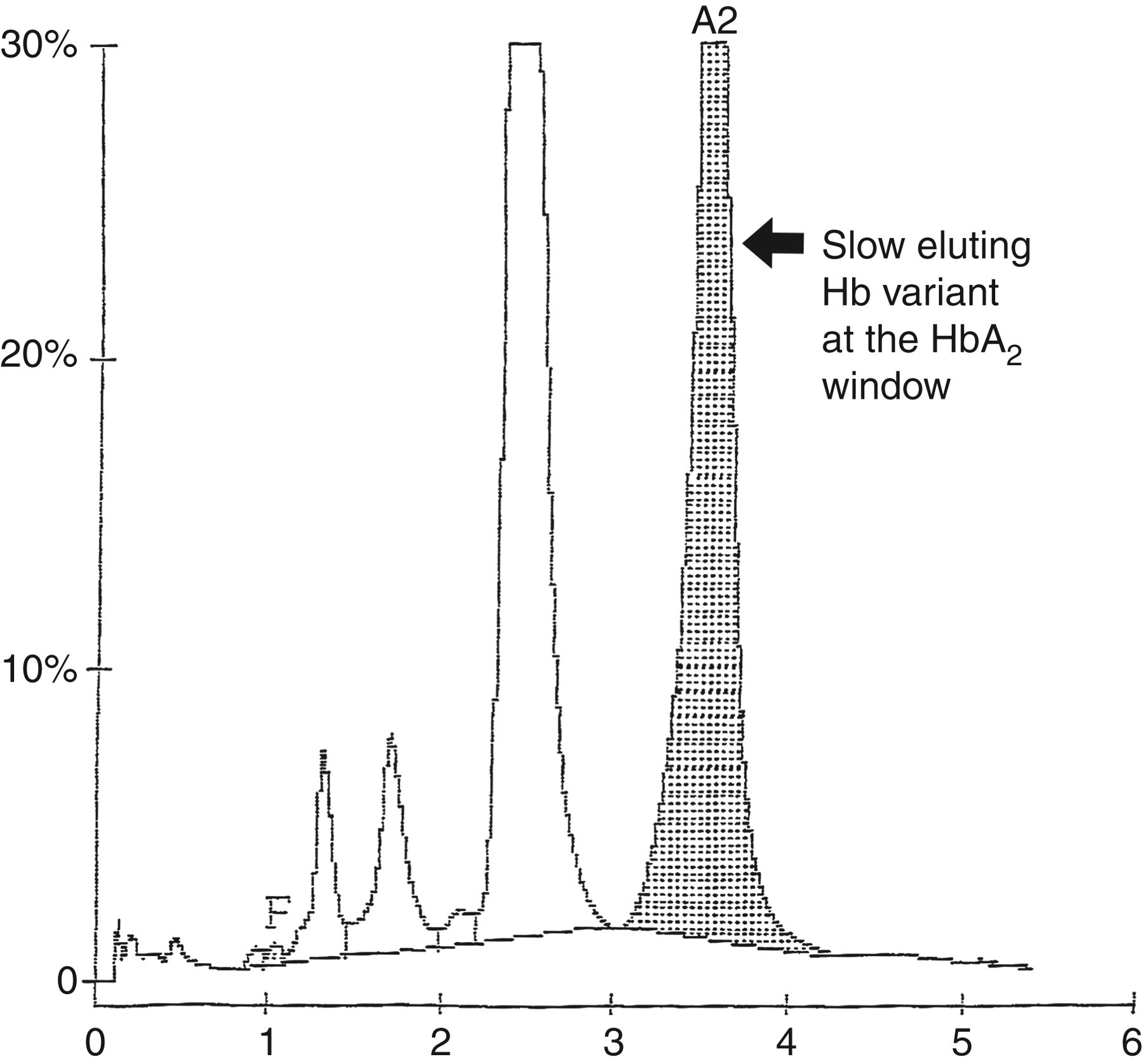
Discussion
The American Diabetes Association has recommended HbA1c as both an indicator of long-term glycaemic control in patients with DM, and a screening and diagnostic tool of the disease. 2 Appropriate clinical application of the HbA1c result relies on how accurately this analyte reflects the glycaemic status of the patient, which can be upset by the presence of haemoglobin variant(s) and technological shortcomings. Studies regarding interference of the HbA1c result by haemoglobin variants 3–6 and various methodologies to tackle this issue 7,8 have been extensively reported. Nevertheless, to the best of our knowledge, none reported shortcomings of the proprietary peak identification algorithms, including the wide and significantly overlapped retention time windows, which may result in wrong peak identification and erroneous HbA1c measurement.
Our laboratory adopts a fully automated NGSP-certified cation-exchange HPLC system, the VARIANT-II, for HbA1c analysis. A proprietary exponentially modified Gaussian (EMG) algorithm 1 is applied to quantify HbA1c concentration based on relative peak area, which is obtained by dividing area of peak HbA1c by area summation of all peaks up to HbAo, against two levels of calibration. All peaks eluting after HbAo, labile HbA1c and carbamylated peaks are excluded.
HbA1c and HbAo retention time comparison between the VARIANT-II built-in algorithm and in-house established system
HbA1c, haemoglobin A1c; CV, coefficient of variation; RT, retention time
CV (%)* = 1/4 RT window/central value, with RT window =± 2 SD
CV (%)† = 1/6 RT window/central value, with RT window =± 3 SD
Our stringent in-house retention time system enabled significant narrowing of retention time windows of both HbA1c and HbAo peaks and complete separation of HbAo and HbE/D peaks. This control mechanism guaranteed correct peak identification in our case and thus revising area summation of all peaks up to HbAo from 3,638,997 to 1,858,165. This resulted in upgrading HbA1c results from 4.7% to 8.2%.
Although haemoglobin variants including haemoglobin F, E/D, S and C were measurable by the VARIANT-II Hemoglobin Testing System, a chromatographic run of three minutes limited its capability to identify a wide spectrum of haemoglobin variants correctly, particularly when the haemoglobin variant eluted within the retention time window of haemoglobin F, E/D, S and C due to charge similarity. As in this case, HbG Taipei was erroneously identified as haemoglobin E/D due to elution in the respective retention time window. Likewise, while the β-Thalassemia Short Program of the VARIANT-II System covers most of the common and important haemoglobin variants in different populations, for example HbS and HbC in Africa, and HbE in South East Asia, it is notable that Hb New York and Hb Malay escape the detection due to co-elution with HbA. Therefore, direct sequencing of the α or β globin genes was required to confirm the haemoglobin variant identity as reported in this case.
The Chinese are a population with a high prevalence of haemoglobinopathy. One review reported the identification of 24 haemoglobin variants in 271 persons living in the Silk Road region of China, among which HbG Taipei, originating from Han in the Yellow River valley, occurred at the highest frequencies. 9 Apart from that, there is hitherto no report in the literature reporting its occurrence in other Asian populations, or its prevalence locally.
Haemoglobin A1c assays can be divided into methods that apply molecular charge, including cation-exchange high-performance liquid chromatogram and electrophoresis; or molecular structure, comprising immunoassays, boronate affinity chromatography and mass spectrometry. The presence of haemoglobin variants may result in over- or under-determination of HbA1c concentration depending on the analytical principle. Cation-exchange high-performance liquid chromatogram and electrophoresis are affected by haemoglobin variants co-eluting with HbA and/or HbA1c due to charge similarity. Erroneous detection of epitopes of haemoglobin variants with amino acid substitutions occurring within the N-terminal β-globin chain can cause interference in immunoassay by an anti-HbA1c antibody, and the presence of a high percentage of HbF. Boronate affinity chromatography is affected by haemoglobin variants with decreased or increased glycation. While mass spectrometry, the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) reference method, is generally not affected by the presence of genetic or chemical modification of HbA and/or HbA1c molecules, cost and technical expertise requirements do not allow general application in routine medical laboratories. The same haemoglobin variant may affect different analytical systems differently. Stating the analytical principle of HbA1c measurement in the laboratory report can avoid inappropriate comparison of results determined by different analytical systems. This is particularly crucial for glycaemic control monitoring.
Individual haemoglobinopathy may affect the degree of glucose binding to haemoglobin in a different way. A subject with a haemoglobin variant and having a HbA1c concentration <6.5% may not always equate to good glycaemic control due to the possible lower degree of glucose binding. This uncertainty limited the general application of same decision cut-off of established guidelines for glycaemic control monitoring. Although fructosamine is an alternative monitoring tool, 10 it only reflects the glycaemic status on a fortnightly basis.
To serve the glycaemic control monitoring purpose, particularly in populations with a high prevalence of haemoglobinopathy, establishment of an individualized system based on critical difference or reference change value is worth consideration. Reference change value involves calculating an appropriate decision goal in terms of analytical and biological variations with an equation [1.96 × sq root (2) × sq root (sq (CVa)+sq (CVw))], where CVa is analytical between-run imprecision and CVw is intra-individual biological variation. Sequential results being significantly different must vary by more than the reference change value. Therefore, a patient with good glycaemic control should have HbA1c in % not deviating from the previous result by the magnitude of reference change value. As an example, an HbA1c system meeting an optimal analytical goal should have CVa not exceeding 2.2% 10 with a CVw of 1.0%, 11 giving a reference change value of 6.7%. For sequential results to be significantly different, the HbA1c result must differ by more than 0.44% if the previous HbA1c reading was 6.5%. Nevertheless, appropriate application of reference change value relies on the constant effect of the haemoglobin variant over time and two sequential results obtained by the same analytical principle. The latter requirement further reinforces the positive and important implication of including the HbA1c analytical principle in the laboratory report.
Conclusion
We report a case of spuriously normal HbA1c result due to HbAo peak misidentification by the VARIANT-II built-in retention time algorithm, which is circumvented effectively by the implementation of a chromatographic system specific internal quality control mechanism for peak identity verification. We recommend adoption of an individualized glycaemic control monitoring system based on critical difference or reference change value in the face of individualization of haemoglobinopathy, which may affect different analytical systems differently. We suggest including the analytical principle of HbA1c measurement in the laboratory report, not only to avoid inappropriate comparison of results obtained by different analytical systems, but also to maximize the application of an individualized glycaemic control monitoring system, particularly in a population with a high prevalence of haemoglobinopathy.
DECLARATIONS
