Abstract
Urine is recognized as the prime matrix for drug test screening with well-established methods and testing protocols. Its major limitation is with regard to the inconvenience of sample collection and lack of integrity due to adulteration, dilution, drug spiking or sample exchange. The question is whether oral fluid, with its apparent better sample integrity, can replace urine for drug screening. This review examines the sample integrity problems and the advantages and limitations of oral fluid and urine in drug screening programmes. The variety of sample collection devices for oral fluid is shown to be a problem with recovery and detection for some drugs. This is examined in relation to the pharmacokinetics of drug metabolism and excretion in this matrix. Buccal contamination with drugs in oral fluid may also cause problems with interpretation. The clinical advantages of oral fluid analysis compared with urine testing are highlighted. Parent drugs are often found in oral fluid where only their metabolites may be found in urine, for example the benzodiazepines. 6-Monoacetylmorphine, an indicative marker of heroin, has a high prevalence in oral fluid from users of this drug but its detection in urine is limited due to its short half-life. Advances in analytical techniques, particularly chromatography linked to tandem mass spectrometry, are helping to promote oral fluid analysis. However, the lack of concordance studies examining both urine and oral fluid drug levels and kinetics in the clinical setting is of some concern.
Introduction
Drug testing in society is becoming an increasingly important issue with toxicology laboratories having to consider the wide spectrum of areas requiring this service and their individual needs. Whereas substance abuse clinics provide a significant workload for drug testing, there is increasing interest outside the clinical addiction arena. Workplace testing (employment and pre-employment screening), although controversial within the UK, is particularly prevalent within the USA and gaining interest in some European countries. Other areas requiring drug testing include hospitals, general practice surgeries, sports organizations and the criminal justice system. Drug testing trials have also been conducted with regard to roadside testing of drivers 1 and in some schools. 2 In the UK, the control of drugs and harmful substances comes under the direction of the Misuse of Drugs Act 1971 and its following subsequent amendments. 3 The medicinal use of such controlled substances is then guided by the Misuse of Drugs Regulations 2001. The list of compounds governed by this legislation is extensive, with approximately 500 being listed on the home office website 4 and a more extensive list held by the Laboratory of the Government Chemist. New compounds arise in society which may then come under such legislation, placing further challenges for the toxicologist. Traditionally, urine has been the matrix for testing, particularly in drug screening programmes where there are well-established laboratory protocols. However, oral fluid or saliva drug testing is gaining interest, helped by the advances in chromatography linked to tandem mass spectrometry (LC/MS/MS). 5,6 The immediate attraction of oral fluid over urine is that it offers a non-invasive collection matrix, which can be easily supervised, thus reducing the problems associated with sample integrity. Oral fluid drug analysis has been extensively reviewed in the literature, 7–9 but questions still arise as to the relative benefits of this matrix over urine. This review examines more closely the advantages and disadvantages of these two matrices, initially looking at how drugs and their metabolites enter these fluids, sample collection procedures, sample integrity and stability. Their relative limitations with regard to detection limits and time windows are explored and the main drug groups examined with regard to their detection and monitoring treatment compliance.
Pharmacokinetics of drugs in oral fluid and urine
Drugs may be ‘used’ or administered therapeutically either through the enteral or through the parenteral route. The parenteral routes, i.e. intravascular injection and inhalation and smoking, are favoured by users of heroin and cocaine as they avoid the first-pass effect and provide an immediate ‘high’. The enteral route is used in therapeutic management of opiate addiction, e.g. oral methadone. Once absorbed, most drugs undergo some degree of metabolism mainly to products that have no biological activity, although some metabolites may exhibit various degrees of activity or toxicity. The initial stage of drug metabolism ‘phase 1 reactions’ involves hydrolysis, oxidation or reduction of the drug. Heroin and cocaine are both rapidly hydrolysed by the same enzymes, butyrylcholinesterase and the liver carboxylesterases hCE1 and hCE2. 10,11 Other drugs, for example, methadone, codeine and some of the benzodiazepines, are metabolized by one of the major forms of enzyme groups involved in drug oxidation, i.e. the hepatic microsomal cytochrome P450 enzyme system (CYP enzymes). The latter is a family of enzymes characterized according to genetic nomenclature and showing variable distribution within the liver. The intraindividual variation in CYP enzymes, together with both induction and suppression of their activity by drugs and their metabolites, has become a major area of interest. Knowledge in this field of pharmacogenomics 12 is important in the interpretation of drugs and their metabolites in oral fluid and urine.
Phase 1 drug metabolism is useful for preparing lipophilic drugs with reactive species such as hydroxyl groups to undergo ‘phase II’ reactions in which drugs/metabolites are rendered more polar by the addition of glucuronide, acetate or sulphate. This enables the final products of drug metabolism to be excreted via the kidneys or the biliary system and unlike phase I metabolism is generally not rate limiting. Consequently, drug metabolites accumulate within the urine and may be detected for several days following initial drug use or administration.
In oral fluid, drugs and their metabolites appear due to passive diffusion from the blood into the saliva from the highly perfused salivary glands. Hence, drugs may be detectable in oral fluid very soon following oral administration and absorption into the blood stream. For example, controlled trials 13 using oral codeine (0.86 mg/kg body weight) demonstrated the presence of the drug in an average time of 30 min in 19 volunteers with peak codeine concentrations at an average time of 1.3 h. The half-life of codeine in this study compared very well with that of the plasma, t 1/2 ∼2.2 h, with the concentration of codeine in oral fluid paralleling that of plasma. The levels of codeine in oral fluid were overall much greater than plasma with an average saliva:plasma ratio of ∼4.0. In general, drugs in oral fluid have a greater concentration than corresponding levels in the plasma. 13–15 This saliva:plasma ratio is dependent on the pK a, degree of protein binding and lipophilicity of the drug. Hence, basic drugs, which include many of the illicit substances of interest (opiates, amphetamines and cocaine), will be ionized at the lower slightly acidic pH of ‘normal’ saliva (pH = 6.7), leading to ‘ion-trapping’ within the buccal cavity. The saliva:plasma ratio may also be influenced by the way a drug is being used. Inhalation or smoking of drugs such as cocaine and heroin will lead to buccal contamination and much higher saliva:plasma drug ratios. In many cases, saliva does account for the bulk of secreted drug within the buccal cavity. However, many collection procedures are based on oral fluid, which is a mixture of saliva, mucosal and buccal transudates, and gingival crevicular fluid together with cell debris and bacterial flora. The saliva fraction contains only the ‘free’ drug present by passive diffusion from the blood circulation. Therefore, drugs which are heavily protein bound may not be detectable or be of a very low concentration within saliva. The presence of transudates in oral fluid, which contain low concentrations of plasma proteins, will aid in the detection of drugs bound to these proteins.
Sample integrity
One of the main advantages of oral fluid over urine for drug testing is related to the practicality and integrity of collecting the matrix. Urine collections do offer a large bulk of matrix, highly suitable for analysis being an aqueous media and generally containing high concentrations of drugs and their metabolites. However, many of the problems associated with urine collections centre around adulteration, dilution or falsification of the sample. In practice, a balance has to be struck between trusting clients and ensuring specimens are not contaminated, replaced or diluted in any way. In some cases, clients may have to be supervised during the urine collection procedure, which will pose resource implications on the clinic. Unfortunately, there is a wealth of information and products available on the Internet to help and advise addicts on ‘how to beat the drugs test’. The laboratory has to be aware of such practices and take measures to examine sample integrity where possible. Procedures for sample validity testing have been revised and published in the American Federal register 16 and endorsed by the Substance Abuse and Mental Health Services Administration (SAMHSA) in the USA. 17
Attempts to dilute the urine to decrease concentrations of drugs below their cut-off levels is probably one of the most common ways of trying to produce a ‘negative’ urine drug test. 18 Urine creatinine is generally measured as a marker of dilution and any urine sample with a creatinine of less than or equal to 1.8 mmol/L is considered as being ‘dilute’. Urine may be diluted ‘in vitro’ by the addition of a variety of diluents or ‘in vivo’ by excessive consumption of water or other fluids or the use of diuretics. 19 In some cases, samples may be ‘substituted’ with fluids that are not human urine or exchanged with samples from drug-free associates. Drug-free ‘human urine’ substitutes may be purchased through the Internet.
Adulterants are added to urine in an attempt to interfere with the screening and/or confirmatory assays themselves or reduce the drug level by chemical destruction. This is achieved in many cases by the adulterant either altering the pH of the urine sample or by its action as an oxidizing agent. There are several common household products that have been used to cause false-negative results in the initial immunoassay drug screen. These include bleach, concentrated lemon juice, vinegar, table salt and eye drops. More recent adulteration products available on the Internet include ‘Urine Luck’, ‘Klear’, ‘Instant Clean’ and Stealth. The active ingredients within these products, which are, respectively, pyridinium chlorochromate, 20 nitrite, 21–23 glutaraldehyde 24 and peroxidase with peroxide, 25 have been investigated with regard to their effects on drug assays. Guidelines with regard to the levels of adulterants and acceptable pH range for urine have been published. 16 Both laboratory-based and on-site adulteration tests are available with regard to detecting adulteration. 26 The simplest testing procedures make use of test strips which have various degrees of effectiveness in detecting adulteration. Regardless of any tests for adulteration, visual inspection of urines should always be carried out with regard to their colour or any evidence of turbidity or unusual odour.
There is no doubt that more novel ways of beating the urine drug test will be pursued and hence the attraction of drug testing moving towards oral fluid. The much easier supervision of sample collection for oral fluid renders any tampering of the sample difficult. There are several commercial oral fluid adulterants available but their mode of action is very similar to that of any common mouth wash. They do not destroy any drugs or change the pH of the oral fluid. 27 In our experience, we have received oral fluid samples from substance abuse clinics where clients have resorted to sucking on sterilizing tablets (active ingredient sodium dichloroisocyanurate) immediately prior to sample collection. This practice does destroy any drugs present in the oral fluid.
Samples may also be adulterated with drugs in an attempt to conceal non-compliance with medication. Spiking urine samples with methadone or buprenorphine may be carried out to mimic compliance. Subjects may also resort to taking oral medication such as methadone or benzodiazepines immediately prior to oral fluid collection. This can lead to extremely high levels of these drugs and may lead to carryover problems with the drug assays, bearing in mind the very low cut-off limits applied to oral fluid testing. Rinsing the mouth prior to sample collection may help to reduce any residual drug contamination.
Collection procedures and devices
Collection of urine certainly presents more issues with regard to sample collection supervision and also washroom facilities, which ideally should be designed to prevent any possible sample dilution. In cases where there are legal implications relating to the drug test, results may be challenged in court and chain of custody procedures for sample collection must be applied. This makes the whole process of urine collection both complex and time-consuming. Subjects may present with ‘shy bladder syndrome’ and if urine collection cannot be properly supervised, then collected samples must be temperature monitored to ensure there has been no substitution of the sample.
From this perspective, the ease of collection and supervision of oral fluid has significant advantages. Oral fluid may be obtained by spitting or dribbling or by the use of special collection devices usually obtained commercially. Unfortunately, the procedure and device used for collection can have very significant effects on the drug concentration and recovery. 28,29 Some sample devices contain a buffer system in which the collection pad is placed following sampling of the saliva/transudates from the oral cavity. This buffer system may impose restrictions on the type of analysis that can be done. The collected and diluted fluid may be applicable to an immunoassay screen but may produce problems with confirmatory analysis by gas chromatography-mass spectrometry (GC-MS) or LC/MS/MS. The presence of surfactants such as Tween 20 in some buffer solutions may be tedious or difficult to remove and presents a problem with ion-suppression when using LC/MS/MS. Surfactants may be included in the buffer system to help recover particularly ‘sticky’ drugs such as cannabis, i.e. Δ 9 tetrahydrocannabinol (Δ9THC). In practice, laboratories can consider altering the collection fluid in the device to suit their particular analytical systems or take extra steps to elute ‘sticky’ drugs from the collection device. 30 Any alterations in the collection device should of course be fully validated.
A particular problem with oral fluid is collecting adequate material for analysis or in the case of some collection devices, employing diluents, not knowing the actual collection volume. The ‘dry mouth’ syndrome may also occur with cigarette smoking and as a result of certain medications or drug use which restricts salivary flow. The latter is under the control of both the parasympathetic nervous system which increases flow rate and sympathetic nerve control which produces a more viscous saliva. Therefore, any drugs that block parasympathetic or increase sympathetic activity may reduce saliva flow and hence total drug excretion. This will affect the measured drug concentration, particularly with collection devices employing a diluent buffer. Possible drugs/medications that may reduce salivary flow include cannabis, amphetamines, antipsychotics, antidepressants and antihistamines. Endogenous markers present in oral fluid, e.g. IgG have been measured and used to correct any dilution effects. 31 Drugs or compounds that increase salivary flow may also reduce oral fluid drug concentrations. As saliva flow increases, the concentration of bicarbonate is increased, rendering the oral fluid more pH neutral. This results in decreased ionization of basic drugs within the oral cavity and hence a reduction in the ‘ion-trapping’ effect. The practice of using stimulated oral fluid collection by chewing or with citrate (sour candy) may result in a reduction in drug levels in oral fluid 32,33 and could lead to concentrations falling below recommended cut-off limits.
Sample stability
The stability of drugs in the collected matrix is extremely important as some samples may take several days to get to a laboratory if sent via the postal system or by courier. Also, samples may have to be stored for possible re-analysis at some later date, particularly for medicolegal work. The stability of some drugs in urine may be a problem in the presence of bactiuria 34 whereas drug stability in oral fluid is very much collection device dependent. 35 Drug instability in both matrices may occur due to the spontaneous hydrolysis of cocaine and 6-monoacetylmorphine (6MAM) 34,36 and conversion of the 7 nitro-benzodiazepines to their respective 7-amino metabolites. 34,37 The aseptic collection of urine or use of preservatives such as sodium azide may help to reduce the problems of urine drug instability. Although, the latter has been suggested to accelerate the hydrolysis of the ester type drugs, i.e. cocaine and 6MAM. 34 The addition of fluoride to oral fluid helps to reduce the degradation of the nitrobenzodiazpines. 37 Further stability problems with oral fluid have been noted for THC 38 and the methadone metabolite, 2-ethylidene-1,5-dimethyl-3,3-diphenylpyrrolidine (EDDP). 39
The amphetamine drug group has been shown to be stable in urine for up to one week at room temperature with or without aseptic collection. 40 Spontaneous hydrolysis of cocaine may still occur in urine even when stored at −20°C. 41
Drug cut-off concentrations and time windows
The time window for the detection of any drug depends upon several factors, including the dose of drug, frequency of dosing, the individual's metabolism, the cut-off used to define the presence of the drug and the matrix tested. Defined cut-off levels for drugs are particularly important in situations where drugs assays have to be legally defensible. Guidelines for cut-off levels for drug testing, i.e. the level above which the drug test is deemed positive, have been well established for urine with respect to both initial screening tests and confirmatory investigations. In the USA, there are mandatory guidelines for drug testing of Federal Employees, defined by SAMHSA and published in the Federal Register. 42 There are also published European 43 and UK 44 guidelines for ‘Legally Defensible Workplace Drug Testing’ in urine samples. With regard to oral fluid drug testing, guidelines have been proposed by SAMHSA and are expected to be produced in the near future in Europe. The SAMHSA cut-off levels for oral fluid are very much lower than those for urine and hence more sensitive screening and confirmatory assays are required for oral fluid analysis.
Overall, the detection windows for drugs in urine are very much longer than for oral fluid. This is not surprising given that oral fluid drug levels have a similar pharmacokinetic profile to blood. A review 45 of single drug dose studies with urine and oral fluid collections demonstrated detection times of 1.5–4 d and 5–48 h, respectively. The latter being measured at the low microgram per litre level. In clinical practice, where more chronic drug use is likely, a more extended window of detection is found. This occurs particularly with cocaine in both oral fluid and urine samples and cannabis in urine. 45
Drug interpretation in oral fluid compared with urine
The growth of interest in oral fluid testing has prompted much method development in this area with published work examining its role in a clinical setting. However, there is a lack of studies looking at the concordance of oral fluid versus urine drug testing in terms of clinical sensitivity and specificity. The relative merits of oral fluid testing versus urine with regard to their clinical effectiveness are discussed below with regard to both drug abuse and treatment.
Detection of opiate abuse
The primary interest in this area is in the detection of heroin produced illicitly by acetylation of morphine present within opium extracted from the poppy plant, Papaver somniferum. Although, any opiate pain medication, including morphine and codeine and the synthetic opiates, oxycodone, hydrocodone, hydromorphone and dihydrocodeine, may be subject to abuse.
Street heroin contains the highly lipophilic drug diacetylmorphine which is rapidly hydrolysed to 6MAM and then to morphine. The plasma half lives of diacetylmorphine and 6MAM are approximately 3 min and 6–25 min, respectively. 46 Morphine persists for longer (plasma half-life 2–3 h) and undergoes conjugation with glucuronic acid to morphine-3-glucuronide and morphine-6-glucuronide (Figure 1). The metabolites 6MAM and morphine are detectable in both urine 47 and oral fluid 48 of subjects abusing heroin. Both free and conjugated morphine are found in urine but the later have been reported as undetectable in oral fluid. 49 The parent drug heroin has been detected both in urine and in oral fluid using GC-MS 50 and LC/MS/MS, 51 respectively. However, heroin is rarely measured routinely as the metabolite 6MAM, an indicative marker of heroin use, is more prevalent in both urine 50 and oral fluid samples 51 in subjects taking heroin. Oral fluid may provide improved identification for 6MAM over urine due to its increased frequency of detection in heroin use, whereas detection of 6MAM in urine is limited by its short half-life. 52 Morphine, although the most prevalent of the heroin metabolites in opiate-positive urine 50 and oral fluid samples, 51 may arise from sources other than heroin metabolism. In urine, morphine may occur from either morphine administration, poppy seed containing foods or codeine metabolism. Poppy seeds contain a variable amount of morphine (2–251 μg/L) and codeine (0.4–57.1 μg/L), 53 and consumption of food products containing these seeds has resulted in positive screening tests for opiates with confirmation of morphine in urine. 54 This became a particular problem in the USA with subjects having a positive urine test for morphine claiming the ‘poppy seed defence’. Hence, the US Government increased the urine screening cut-off for morphine to a level that would prevent this defence from being used. Morphine greater than a cut-off level of 40 μg/L has been detected in oral fluid following consumption of poppy seed containing foods but for a shorter period (1 h) compared with urine samples from the same subjects (8 h). 54
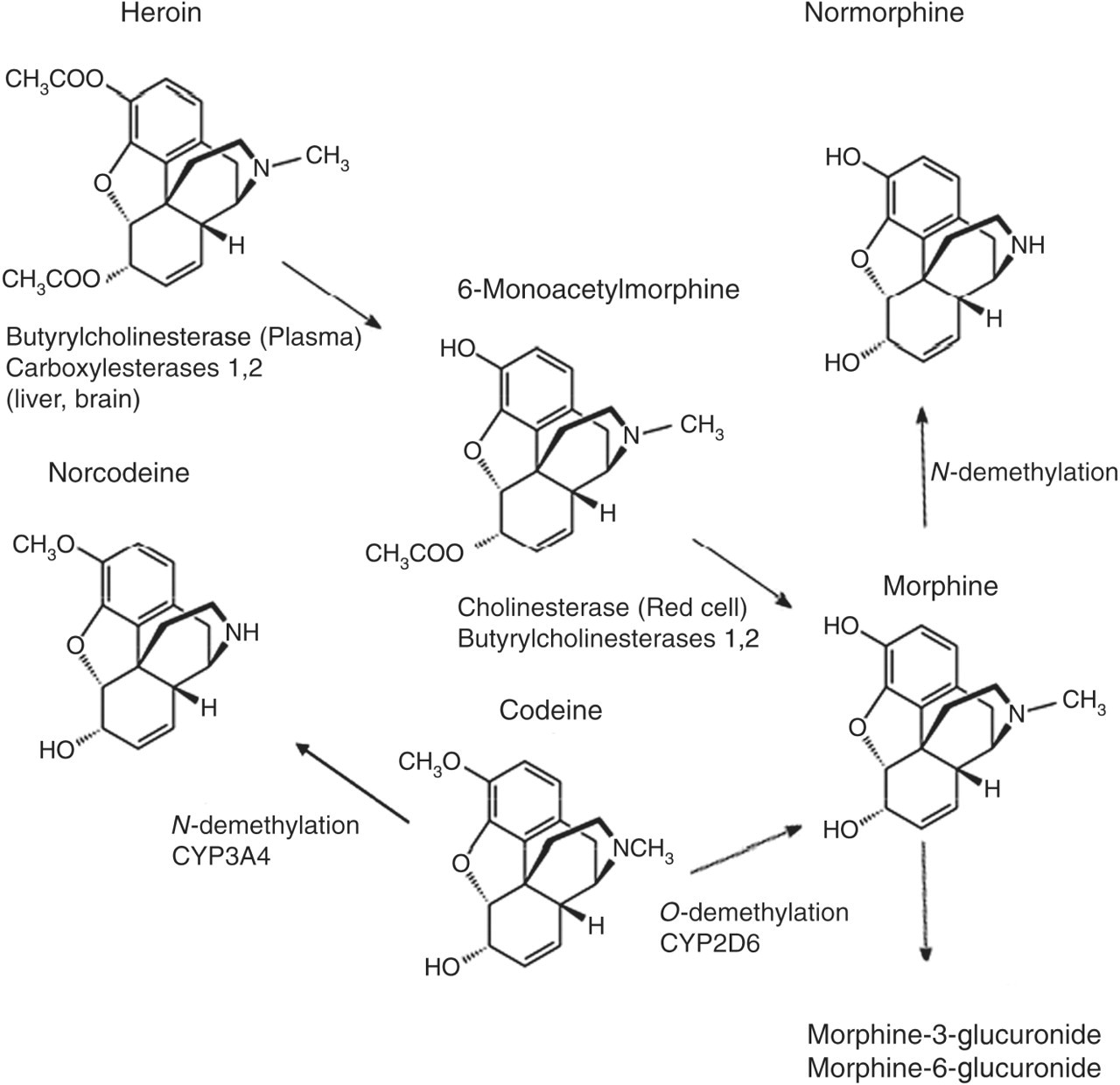
Metabolism of heroin
Codeine which may be present in over-the-counter medications or present within street heroin may also give positive results for morphine in urine. 55 Codeine, although primarily metabolized to codeine-6-glucuronide, is also converted to morphine by the CYP2D6 and norcodeine by CYP3A4. This is with the exception of approximately 10% of the Caucasian population who are deficient in CYP2D6 activity and hence little or no morphine is formed from codeine. 56 The ratio of codeine to morphine in urine if >1 may give some indication that the morphine present may have arisen due to codeine metabolism. 55 One advantage of oral fluid over urine is that following codeine administration, both codeine and norcodeine may be detected but not morphine. 13 Hence, the presence of morphine in oral fluid is more likely to raise suspicion of heroin use.
Detection of cocaine abuse
Cocaine used illicitly may be injected or taken intranasally (snorted) as its hydrochloride or smoked as free base (crack). 57 The drug is rapidly hydrolysed in the blood to its major inactive metabolites, i.e. benzoylecgonine by human carboxylesterase-1 and to ecgonine methyl ester (EME) by action of butyrylcholinesterase and human carboxylesterase-2 58 (Figure 2). A controlled dosage study demonstrated benzoylecgonine to have the highest concentration of cocaine metabolites in urine followed by EME and six minor metabolites, including norcocaine, benzoylnorecgonine, m- and p-hydroxycocaine, and m- and p-hydroxybenzoylecgonine. 59 Of these, only norcocaine has any biological activity but the psychoactive compound cocaethylene is produced if alcohol is taken with cocaine. Smoking crack cocaine produces the specific metabolite anhydroecgonine methyl ester. Variable amounts of unmetabolized cocaine may also be found in urine following use of the drug and can account for 1–9% of the total level of cocaine products within the urine. 60
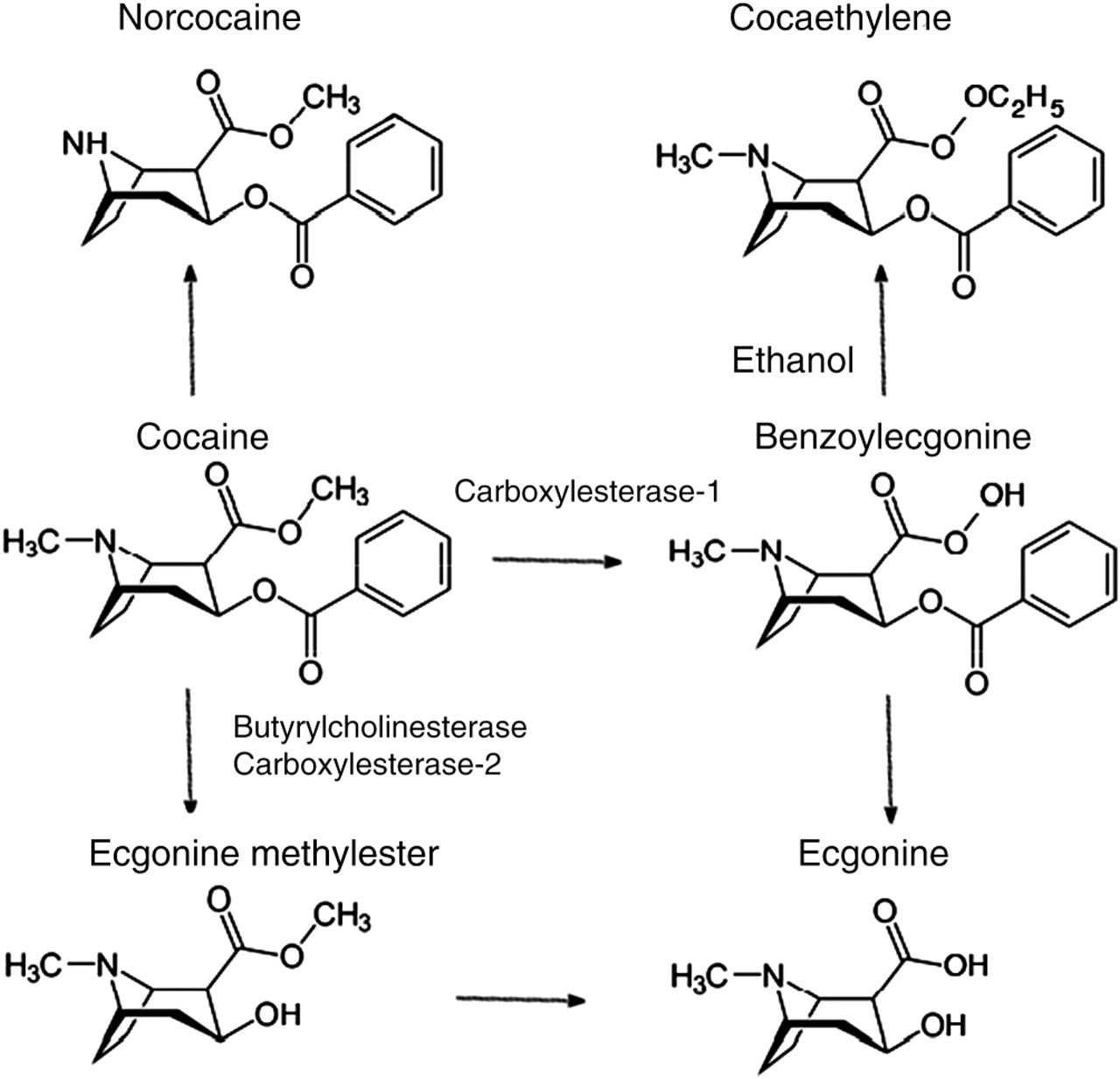
Metabolism of cocaine
In oral fluid, both cocaine and benzoylecgonine as well as EME and norcocaine and trace amounts of the minor metabolites m- and p-hydroxycocaine are found following cocaine use. 61 Studies comparing urine versus oral fluid with regard to the detection of cocaine use may be dependent on methods used to measure cocaine and its metabolites and decision cut-off levels used. In particular, single dose studies of cocaine in volunteer subjects may have underestimated the utility of oral fluid for detection of cocaine in a realistic setting. 62 A recent study by Dams et al. 61 comparing cocaine in urine and oral fluid at their respective SAMSHA cut-offs demonstrated oral fluid to be more sensitive than urine in detecting cocaine use.
Detection of amphetamine abuse
The amphetamine class of drugs include the powerful central nerve stimulants, amphetamine and methamphetamine, as well as the designer drugs including 3,4 methylenedioxymethamphetamine (MDMA), 3,4 methylenedioxyamphetamine and 3,4 methylenedioxy-N-ethylamphetamine. Other drugs in this class that may be of interest include ephedrine, pseudoephedrine and phentermine. Amphetamine and methamphetamine may be prescribed or be subject to abuse. Complications can arise from the fact that this class of drugs exist as both S-(+) and R-(−) enantiomers with the former having five times the psychostimulant activity than the latter enantiomer. Street amphetamines may present as a racemic mixture of these enantiomers whereas prescribed amphetamine is in the S-(+) form. The R-(−) enantiomer of methamphetamine is available in the Vicks inhaler available in the USA. Amphetamine and methamphetamine may also arise from the metabolism of other substances. 63
Amphetamine and methamphetamine are metabolized primarily by aromatic hydroxylation by dopamine-β-hydroxylase, the hydroxylated metabolites undergoing a phase II reaction by conjugation with sulphate or glucuronic acid. In addition, methamphetamine is N-demethylated to amphetamine. Significant amounts of unmetabolized amphetamine and methamphetamine are excreted and both these parent drugs have been measured in urine 64 and oral fluid 65 following their administration. However, the urinary excretion of these drugs is heavily influenced by the pH of the urine. Under normal physiological conditions, the acidic nature of urine results in the relatively basic amphetamines (pK as ∼9.9) remaining in an ionized state reducing any reabsorption by the kidneys. This can result in 57–66% of amphetamine being excreted in the free form. If the urine is alkalinized by excessive intake of bicarbonate, then this free fraction can fall to less than 5%, which may potentially produce a negative urine screening test. 66 Oral fluid appears to be unaffected with regard to systemic intake of bicarbonate. Hence this matrix has advantages over urine testing in being less susceptible to adulteration. Although, controlled administration studies have shown lower oral fluid concentrations of amphetamine and a reduced window of detection compared with urine. 65
Detection of cannabis abuse
Cannabis comes in three major forms, including marijuana, hashish and hash oil, and is usually smoked but may also be eaten with hash baked into food products. The active constituent THC is hydroxylated in the liver by CYP2C9 to 11-hydroxy-THC. This is further oxidized to the carboxylic acid metabolite, 11-nor-Δ9-tetrahydrocannabinol-9-carboxylic acid (THC-COOH), which undergoes a phase II conjugation reaction with glucuronic acid and is excreted in the urine 58 (Figure 3). Both screening and confirmation of cannabis use is determined by measurement of the carboxylic acid metabolite in urine. In oral fluid, assays have targeted the measurement of the parent compound THC as the metabolite THC-COOH has been reported as being undetectable. 67 However, more recently, GC-MS assays with a quantitation limit down to 2 pg/mL have detected THC-COOH in 76.2% of oral specimens that have screened positive for cannabinoids. 68 The detection of THC in oral fluid may have advantages over the detection of THC-COOH in urine in indicating more recent exposure to cannabis. The detection window for THC-COOH in urine is dependent on several pharmacological and physiological factors, including the amount, frequency and strength of the parent drug, THC, being used. Heavy users of the drug may show positive urine tests for several weeks or months even following abstinence from using cannabis. 69,70 This may be explained by the fact that THC, being a very lipohilic drug, is deposited within the adipose tissue and slowly released back into the blood stream and metabolized to THC-COOH. The release of THC from adipose tissue is highly variable and to some extent dependent on the body mass index of the individual.
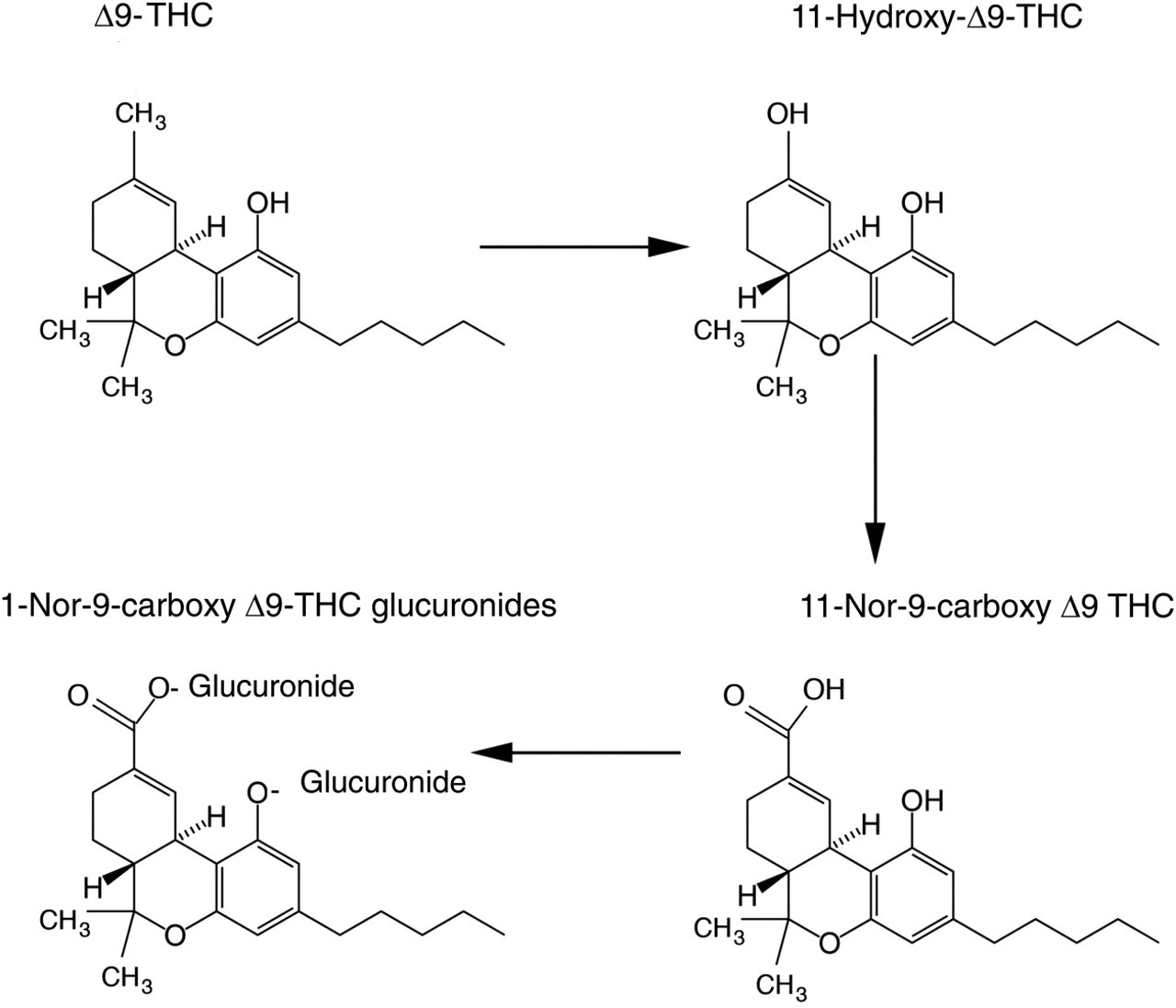
Metabolism of tetrahydrocannabinol (THC)
In oral fluid, the pharmacokinetics of THC appears to compare very favourably with that of blood plasma but with a high saliva:plasma ratio. 71 However, the bulk of THC measured in oral fluid is more likely to come from the depot effect following smoking of the drug. A comparison of oral fluid collection to urine following the smoking of a single marijuana cigarette by 10 individuals demonstrated a high degree of variability in detection times for the measurement of THC and THC-COOH, respectively. Overall, the degree of positivity for cannabis detection in oral fluid was higher in the early phase following marijuana exposure but the average window of detection was much longer for urine than oral fluid (58 h compared with 34 h using GC-MS). 71 The depot effect of cannabis in the oral cavity could potentially result in positive screening results due to passive inhalation. Exposure of volunteers to cannabis smoke in a controlled environment has demonstrated positive oral fluid and urine results with regard to their respective cut-off limits. 72 The risk of a positive oral fluid test for cannabis due to passive inhalation was limited to a period of approximately 30 min following exposure. The measurement of conjugated THC-COOH in oral fluid has been suggested to minimize concerns associated with passive inhalation. 68
Detection of alcohol abuse
Although alcohol can be measured in both urine and oral fluid, its clinical usefulness in the management of addiction is limited by its narrow time window of detection. 73 A small fraction of any alcohol consumed (<0.1%) undergoes a phase II conjugation to produce ethyl glucuronide (EtG). EtG has a longer half-life than alcohol and can be measured in both oral fluid and urine with respective time windows of 14 74 and 25 h 75 following a moderate dose of alcohol.
Monitoring treatment compliance
The role of the laboratory here is primarily in compliance monitoring of opioid substitution therapy with methadone, buprenorphine or dihydrocodeine and in some cases monitoring opiate antagonist therapy with naltrexone. Benzodiazepines may also be prescribed to help alleviate symptoms in the management of opiate and alcohol abuse and are also monitored for compliance. The laboratory has to be aware that all these drugs, with the exception of naltrexone, may be subjected to abuse or may be used to adulterate samples to mimic compliance. There is also increasing interest in treating heroin addiction by offering diacetylmorphine to subjects and this is now an established treatment in some European countries. This poses a problem for the laboratory in the detection of subjects on such pharmaceutical heroin treatment programmes returning to street heroin use.
Methadone
Methadone is metabolized primarily by N-demethylation to EDDP by CYP3A and to a minor extent by CYP2D6 and CYP1A2. 76 CYP3A exhibits large intraindividual variation which was thought to account for the differences in methadone metabolism between subjects. 77 This proposal has been disputed more recently with CYP3A activity being a contributing factor to the clearance of unbound rather than total methadone. 76 However, the influence of pharmacological interactions 78 and pregnancy on CYP3A activity may be significant factors in methadone metabolism. The latter has been demonstrated to enhance CYP3A activity 79 and may also affect methadone absorption. Drugs may act as inducers of CYP3A activity, e.g. carbamazepine and rifampacin or inhibitors, e.g. ritobavir, omeprazole, cimetidine and itraconazole. A host of others drugs, metabolized by CYP3A4, may act as competitors. 80 Although it has been suggested that CYP3A4 activity has only a modest influence on methadone disposition, 76 any influence on its activity should be considered in patients taking this drug. In fact, any variation in the pharmacokinetics of methadone metabolism may influence the interpretation of laboratory screening tests. Compliance testing for methadone by measuring the parent drug above a defined cut-off level could be affected by ‘fast’ or ‘slow’ metabolizers of the drug and in cases where its absorption is decreased. Hence, measuring the metabolite EDDP in urine at a suitable cut-off concentration has proved successful in monitoring methadone compliance, with methadone positive/EDDP negative samples suggesting tampering in some cases. 81 There also appears to be a link between urine methadone and urinary pH, with urinary concentrations decreasing with increasing urinary pH but no such relationship with EDDP. 82
A comparison of urine testing to that of oral fluid for methadone compliance has been carried out in a treatment setting using point-of-care testing. This study demonstrated a sensitivity for methadone in oral fluid and urine as 91% and 94%, respectively, and specificity of 90% and 95%, respectively. 83 A combination of immunoassay screen for methadone followed by GC-MS-MS confirmation demonstrated 100% positivity for oral fluid testing for methadone in subjects undergoing opioid replacement therapy. 84 However, the metabolite EDDP in this latter study was demonstrated in only 30.1% of methadone-positive samples. Other studies using LC-MS-MS have also supported the poor sensitivity of EDDP in confirming methadone compliance in oral fluid. 5 This reduced sensitivity for detection of EDDP in oral fluid compared with urine testing does disadvantage oral fluid as a matrix for monitoring compliance for methadone. A positive oral fluid test for methadone could easily be attributed to oral contamination with the drug, particularly if procedures to rinse the mouth prior to specimen collection are not carried out. If gross contamination of the buccal cavity with methadone occurs, then very high drug levels in oral fluid will be achieved, leading to significant carryover in the analysis.
Buprenorphine
Buprenorphine, a semisynthetic opioid used in the treatment of opiate addiction, is provided in tablet form as Subutex® or in combination with naloxone as Suboxone®; both drugs are administered sublingually. Buprenorphine may be subject to misuse with tablets being crushed, dissolved and injected. The purpose of adding the opiate antagonist naloxone to the tablet is to prevent such misuse as it blocks the action of buprenorphine on the opioid receptors when injected. Naloxone is poorly absorbed orally and hence does not have any pharmacological effect when given sublingually as Suboxone. Following absorption, buprenorphine has a large volume of distribution and is heavily protein bound (96%). Metabolism occurs in the liver by N-dealkylation to norbuprenorphine primarily by CYP3A4. Faecal elimination predominates in removal of the majority of a buprenorphine dose followed by urinary excretion (10–30%). 85 Both the parent drug buprenorphine and the metabolite norbuprenorphine can be found in urine as well as their glucuronide conjugates. 86 Generally, compliance for buprenorphine is determined by measuring the parent drug using immunoassay or both total buprenorphine and norbuprenorphine by GC-MS or LC/MS/MS following enzymatic deconjugation of the glucuronides. 87 Urine samples from patients on buprenorphine maintenance therapy demonstrate a high ratio of norbuprenorphine to buprenorphine. 87
Buprenorphine is present in oral fluid but at low concentrations due to its high protein binding in the blood. Also, its measurement in oral fluid may be compromised due to buccal contamination following sublingual administration of the drug which may lead to very high saliva levels. 7 There is very little work published with regard to the metabolite norbuprenorphine in oral fluid, but the experiences of this laboratory have shown this metabolite to be very challenging to measure due to extremely low levels in this matrix.
Benzodiazepines
The benzodiazepines are among the most frequently prescribed drugs in the world and comprise a number of compounds based on a similar chemical structure of a benzene ring fused to a diazepine ring. With regard to the treatment of drug addiction, diazepam is the most commonly prescribed benzodiazepine. However, there are a host of other benzodiazepines which may be prescribed or subject to abuse. Although the benzodiazepines share a similar chemical structure, they do have different physiochemical properties, which can influence their pharmacokinetics. Overall, they are extensively metabolized involving hepatic microsomal oxidation, N-dealkylation or aliphatic hydroxylation with the parent compounds or metabolites undergoing glucuronidation. 88 Consequently, they do present an analytical challenge, particularly as in many cases very little free parent benzodiazepine is found in urine and common metabolites may arise from several benzodiazepines. Methods using general immunoassay screen for benzodiazepines in oral fluid and urine are available. However, immunoassay screening methods require an antibody with broad specificity against a number of benzodiazepines which can lead to other drug interferences in the assay, particularly in urine samples. 89
The benzodiazepines are measurable in oral fluid and in many cases the parent drug is detectable as well as some metabolites (nordiazepam, oxazepam, temazepam, 7-aminonitrazepam). 5 The detection of the parent drug can have a significant advantage over urine in some clinical situations such as distinguishing a prescribed benzodiazepine from a ‘street’ drug. For example, chlordiazepoxide is licensed for prescription to subjects with an addiction to street benzodiazepines or to those with an alcohol addiction. The parent drug, chlordiazepoxide, has been confirmed in oral fluid samples analysed within this laboratory, but its extensive metabolism leads to less than 1% of the dose excreted in urine. 90 The major problem with oral fluid as a matrix for detecting benzodiazepines is that sensitive assays are required to enable their detection and quantitation. These drugs have a high percentage of protein binding within the blood and their oral fluid:plasma ratios are very low. 91,92 Their detection limits in oral fluid may also be very much device dependent with regard to oral fluid collection. Smink et al. 93 using LC/MS/MS demonstrated a poor sensitivity of oral fluid compared with urine for benzodiazepines in a roadside testing. However, this latter study had relatively high limits of quantitation in oral fluid for the benzodiazepines studied. More sensitive LC/MS/MS assays 94,95 for benzodiazepines in oral fluid have been described, but these do require validation against urine using authentic samples. Controlled trials 92 administering oxazepam to volunteers demonstrated that oral fluid measurements of this compound paralleled those of blood, although at much lower concentrations, but were able to demonstrate recent ingestion.
Diacetylmorphine (pharmaceutical heroin)
Pharmaceutical grade heroin is being used in several countries on the European Continent and also being trialled on a smaller scale within the UK for the treatment of heroin addiction. The role of the toxicology laboratory in this case is to exclude the continued use of illicit heroin in subjects receiving pharmaceutical heroin. Hence, attention has been directed at measuring biomarkers that may uniquely determine the use of illicit heroin including noscapine, papaverine and their metabolites 95 and 6-acetylcodeine. 96 As expected, most published work with respect to these markers has been conducted on urine. Acetylcodeine measurements have been done on both oral fluid 51 and urine 96 in subjects using heroin. The variability of acetylcodeine concentrations in illicit heroin does pose problems with the sensitivity of this investigation in detecting street heroin use. Overall, biomarkers for illicit heroin are an area requiring future research and development work.
Discussion
The question remains as to whether or not oral fluid can replace urine as the primary matrix for drug test screening or if it is just an additional matrix that may be useful in some situations. From the clinical perspective, the ease of sample collection of oral fluid and reduced likelihood of sample interference is highly attractive and toxicology laboratories may come under pressure to offer this service. From the toxicology perspective, there are still issues that may cause concern in the introduction of this matrix. There is a lack of concordance studies with patient urine samples and only limited pharmacokinetic evidence based on single or multidose drug studies. Problems highlighted with the detection of cannabis and metabolites of buprenorphine and methadone remain of concern. The choice of oral fluid sampling device and protocol for collection may have a considerable effect with regard to drug excretion, drug recovery and hence final drug concentration. Buccal contamination with drugs may occur through passive inhalation or ingestion immediately prior to sampling. There is also no guidance within the European community with respect to cut-off levels for drugs in oral fluid. Cut-off levels, once decided upon, will no doubt be very much lower than those required for urine testing. This will require more sensitive screening and confirmation assays which together with the cost of a sampling device will have financial implications for both the laboratory and service users. Given these issues, it would appear that the trade-off in moving to oral fluid testing to achieve better sample integrity and convenience of collection is significant.
There are, however, some clinical advantages to oral fluid testing other than that of better sample integrity. Some drugs are extensively metabolized and only very small amounts may be found in urine whereas detection of the parent drug may be more likely in oral fluid. The benzodiazepines are one such example, provided an appropriate validated oral fluid collection device is used. A further clinical advantage of oral fluid over urine analysis is the detection of 6MAM. This indicative marker of heroin use demonstrates a high incidence in oral fluid samples taken from heroin users.
The measurement of drugs in oral fluid which may require very low detection limits has been helped by developments in LC/MS/MS. This technique has improved oral fluid drug testing and with faster chromatography has made this form of analysis more routinely applicable. Oral fluid drug analysis may also be simpler than urine in some respects, e.g. not requiring any preanalytical enzymatic hydrolysis steps to analyse conjugated drugs. Certainly, more concordance studies with respect to these matrices are needed to establish their relative advantages and their limitations in the clinical setting. Also, as oral fluid analysis gains acceptance, the ingenuity of those abusing substances will no doubt produce challenges that may impair the integrity of this matrix.
DECLARATIONS
