Abstract
Alcohol is associated with significant morbidity and mortality. Subjects abusing alcohol can be identified through clinical history, examination or self-report questionnaires. A range of biomarkers is available for detecting alcohol misuse, but there is still a need for a marker that can detect alcohol consumption in the time window between one day (ethanol) and one week (gamma-glutamyl transpeptidase and carbohydrate-deficient transferrin). Ethyl glucuronide is a direct metabolite that can be detected in urine for up to 90 h and has the potential to become a useful marker of ‘binge’ drinking. As a non-invasive marker, it could have a role in a variety of clinical and forensic settings.
Introduction
The 2004 Global Status Report on Alcohol from the World Health Organization (WHO) estimated that more than 76 million people worldwide had recognizable alcohol misuse. 1 The morbidity and mortality associated with alcohol misuse is significant; the WHO estimated that it causes 1.8 million deaths worldwide each year, one-third of which were due to accidents. In the UK, it is estimated that alcohol misuse costs the National Health Service between £1.4 and £1.8 billion each year. 2
Many subjects misusing alcohol can be identified by the clinical history and examination or with self-report questionnaires, but sensitivity is generally poor with deliberate under-reporting being common. A range of biomarkers for detecting alcohol misuse have been described, including ethanol itself in breath, blood or urine, gamma-glutamyl transpeptidase (GGT), carbohydrate-deficient transferrin (CDT) and erythrocyte mean corpuscular volume (MCV).
3
Ethanol measurements have high specificity for excessive alcohol intake, but the time window for positivity is short (breath 4–6 h, blood 10–12 h, urine 18–24 h). MCV and GGT require significant alcohol intake over a prolonged period of time (>1000
Metabolism of alcohol
The majority of alcohol ingested is metabolized in the liver in a two-stage enzymatically catalysed oxidation process. Alcohol is first converted to acetaldehyde by alcohol dehydrogenase and then further metabolized to acetate by aldehyde dehydrogenase. A small amount is excreted unchanged in urine, sweat and expired air.
Ethyl glucuronide (ethyl β- Alcohol metabolism and formation of ethyl glucuronide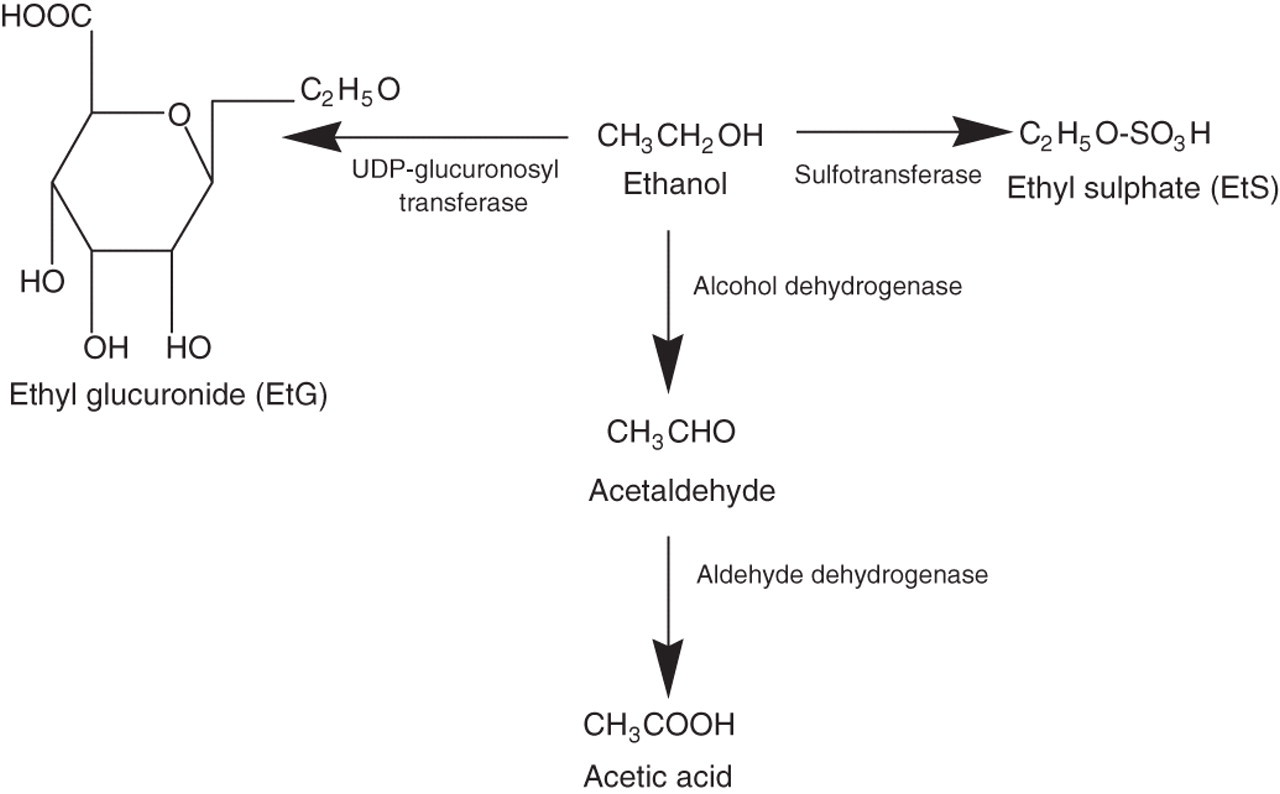
The average time difference after ingestion of alcohol between reaching maximum blood ethanol concentrations and maximum plasma metabolite concentrations is approximately 2–3 h for EtG and 1–2 h for EtS. 7,8 There appear to be wide interindividual variations in the maximum plasma EtG and EtS concentrations and there is no correlation between the metabolites and blood ethanol concentration. 7 Studies have found that metabolite elimination occurs exponentially with a median half-life of between 2 and 4 h. 8–10 EtG can usually be detected in the urine for between 72 and 90 h.
The elimination rate of EtG and EtS does not appear to differ between a healthy population and heavy drinkers during alcohol detoxification. 9 However, a decreased elimination rate and higher blood concentrations have been reported in patients with renal disease, 9 which would delay excretion of these metabolites. A study by Wurst et al. 11 found that EtG concentrations were influenced by age, gender, cannabis use, kidney disease and the total grams of ethanol ingested in the previous month. Race, smoking, body mass index, liver cirrhosis, the age at which subjects began drinking regularly and total body water had no significant influence on EtG concentrations in urine. 11 While no apparent interference with the formation of EtG or EtS from other ingested drugs has been found, it is possible that competition from drugs conjugated by the same enzyme system might decrease EtG formation, and drugs resulting in enzyme induction might increase EtG.
The effects of water diuresis have been published. While it is possible to lower the EtG concentration by drinking large volumes of water, expressing EtG as a ratio to creatinine overcomes this problem of urinary dilution. 5,12 However, Helander et al. 13 reported wide interindividual variations in detection times even after adjusting concentrations for urine dilution.
Stability
EtG has been shown to be a stable marker. Studies have shown that urine samples stored at 4°C for five weeks were found to have no change in EtG concentration. 14 When stored at room temperature in ventilated vials, EtG concentration increased due to water evaporation. During the study, there was no evidence of analyte decomposition in urine. 14 However, EtG-positive tissue material allowed to slowly decompose at room temperature exhibited a decrease in EtG concentration over time but no postmortem formation was found indicating that a positive result proves alcohol consumption prior to death. 14 Further studies on EtG in postmortem samples is warranted as there are well-known problems in interpreting ethanol measurements in this setting.
Cut-off values
Studies in healthy volunteers given alcohol at doses between 0.1 and 0.8 g/kg body weight have consistently shown that applying cut-off values of 100–200 μg/L for urine EtG and 100–110 μg/L for EtS successfully detected the intake of alcohol. 13 In routine clinical use, cut-offs as high as 500 μg/L have been used to reduce the risk of false-positive results. 13 To date, there have been insufficient clinical studies on plasma EtG to allow the reliable determination of the most appropriate cut-off values. In a volunteer study, a maximum plasma EtG concentration of 360 μg/L (range 280–410 μg/L) was found in samples taken 1.5–24 h after a 0.5 g/kg dose of alcohol and 1060 μg/L (range 800–1220 μg/L) after a dose of 1.0 g/kg dose of alcohol. 15
Detection times
There have been a number of studies characterizing the time window during which EtG remains detectable in urine following alcohol intake in healthy volunteers. 5,7,8,16–19 These studies involved doses of alcohol from 0.1 to 0.85 g/kg body weight and the detection window was 24–48 h for both EtG and EtS. One study using a larger dose of alcohol (>1 g/kg) found that EtG remained above the limit of detection (100 μg/L) for 39–102 h. 20 This wider time window is in agreement with those reported in two studies of alcohol-intoxicated subjects where EtG was detectable for 40–130 h. 13,21
Causes of false-positive or -negative results
Despite the stability EtG exhibits in urine, it has been documented that both false-positive and -negative results can arise from bacterial contamination of urine. Glucuronide and sulphate conjugates are cleaved by β-glucuronidase and sulphatase enzymes, respectively. Studies have shown that EtG, but not EtS, is sensitive to bacterial hydrolysis when samples are infected with Escherichia coli and Clostridium sordelli. 22,23 Since E. coli is the most common pathogen in urinary tract infections, there is a risk of falsely low EtG results. It is, therefore, recommended that EtG measurement is combined with EtS analysis using liquid chromatography-mass spectrometry (LC-MS), since EtS is unaffected by contamination. In addition, samples can be taken into containers with fluoride preservatives and frozen to prevent bacterial growth. 22 Other preservatives, e.g. boric acid, have not been investigated with respect to EtG.
Further work by Helander et al. 24 identified the potential for postcollection synthesis of ethyl glucuronide if the sample is infected with E. coli in the presence of ethanol. The formation of ethanol in unpreserved specimens can occur from microbial contamination and fermentation. This risk is highest in subjects with diabetes due to their glycosuria. Formation of EtG postcollection is not always prevented by fluoride preservatives or storage at 4°C 24 and, therefore, caution is advised when interpreting results. Corresponding EtS concentrations were shown to be stable, suggesting a lack of EtS formation in these contaminated samples, indicating that EtS should accompany or be used to verify EtG results.
As the utilization of EtG becomes more common for clinical and medicolegal purposes, it has become more important to determine if sources of ethanol other than consumption can be responsible for the presence of EtG. Studies have shown that ethanol absorbed into the body from the use of alcohol-based mouthwash may result in the presence of EtG in the urine, although normal routine use is not likely to cause high values. 25 In the study by Costantino, 25 routine use, three times a day after meals, resulted in 29% of subjects showing urine EtG concentrations >50 μg/L. In two further small studies involving 14 subjects, only one had a urine sample that was positive for EtG, although seven had detectable EtS (maximum concentration 104 μg/L). 26,27 It has also been demonstrated that detectable concentrations of EtG can be generated using alcohol-containing hand sanitizer gels. 28 These products can contain 60–65% ethanol by weight, and when used eight times over an eight-hour period, subjects achieved urine EtG and EtS concentrations up to 103 and 51 μg/L, respectively. 28 A more intensive regimen in which 11 volunteers cleansed their hands with a sanitizer (62% ethanol) every five minutes for 10 h on three consecutive days showed that EtG and EtS could be detected (maximum concentration 2001 and 84 μg/L, respectively) at the end of the study day, but only two specimens had detectable EtG the next morning (96 and 139 μg/L) and only one detectable EtS (64 μg/L). 29
The ‘alcohol-free’ beers sold in parts of the world may have up to 0.5% alcohol and still be deemed a non-alcoholic beverage. In four volunteers who consumed 2.5 L of these beers, EtG concentrations ranging from 300 to 14,100 μg/L were found in the next morning's urine sample. 30 Positive EtG results were also found in another study 13 h after consumption of non-alcoholic beers, five hours after eating sauerkraut and 3.5 h after consuming matured bananas. 31 Similarly, in vivo fermentation of baker's yeast to ethanol with subsequent formation of EtG and EtS has been reported, although, paradoxically, ingestion of brewer's yeast did not result in any positive EtG or EtS results. 32
Methods for measurement of EtG and EtS
Various methods for measuring EtG have been published. The most commonly used is LC-MS since it is highly sensitive, specific and is able to determine both EtG and EtS simultaneously. 33–36 This technology has been used to determine EtG in urine, whole blood, serum, meconium, 37 saliva 38 and hair. 39
LC-MS detection of EtG can be carried out using selected ion monitoring of the precursor ion (m/z 221) and the principal daughter ion (m/z 75). Penta-deuterated EtG (ETG-D5, m/z 226) is used as the internal standard. 33 The transition m/z 221→85 has also been used. 34,38 Corresponding transitions for EtS are m/z 125→97 and m/z 125→80. 40 Penta-deuterated EtS is also available as an internal standard. Most LC-MS methods have a limit of quantitation for EtG and EtS of 50–100 μg/L. Urine can be injected directly without extraction following centrifugation and dilution with water and internal standard. Serum samples can be analysed after deproteinization with methanol or acetonitrile, centrifugation and addition of the supernatant to an aliquot of the mobile phase. A comparison of five LC-MS methods for measurement of urinary EtG and EtS recommended that solid-phase extraction followed by liquid chromatography-tandem mass spectrometry (LC-MS/MS) be adopted as the reference method as it had the best selectivity and sensitivity. 41
Other methods developed include reversed-phase liquid chromatography with pulsed electrochemical detection, 42 microwave-assisted extraction followed by gas chromatography-mass spectrometry (GC-MS), 43,44 GC-MS with solid-phase extraction for sweat samples 45 and GC-MS of silylated derivatives, 46 capillary electrophoresis, 47 capillary zone electrophoresis-mass spectrometry, 48 capillary isotachophoresis and zone electrophoresis 49 and an enzyme-linked immunosorbent assay based on polyclonal antibodies. 50
Recently, a commercially available enzyme immunoassay (EIA) method based on a new monoclonal antibody (DRI Ethyl Glucuronide Enzyme Immunoassay; Thermo Fisher Scientific Diagnostics, Hemel Hempstead, UK) came onto the market for the analysis of EtG in urine. Comparison with an established LC-MS method showed good agreement (r 2 = 0.931), indicating a low cross-reactivity of the EtG antibody to other urinary constituents. 51 The method evaluation showed that the EIA is sensitive, specific and offers a low, but clinically relevant, measuring range (0–500 μg/L) with a detection limit of 100 μg/L. Samples with high EtG concentrations require dilution. Despite the good correlation to LC-MS techniques, the EIA method is considered a screening test and EtG-positive samples should always be confirmed by LC-MS/MS with EtS to rule out false-positives due to contamination or possible cross-reactivity with other drug glucuronides, although the latter is unlikely. 51
Applications
The measurement of EtG has been carried out in a variety of clinical and forensic settings. Alcohol misuse can be implicated in a significant proportion of subjects admitted to a hospital emergency department with gastrointestinal symptoms or following minor injury. Self-report of alcohol intake using the Alcohol Use Disorders Identification Test (AUDIT) questionnaire can be unreliable due to underestimation of alcohol consumption by the subjects. Two studies of the use of EtG measurements in the emergency room setting have been carried out; one using urine 52 and one plasma samples. 53 While most subjects either tested negative for blood ethanol or had only low blood alcohol concentrations (BAC) in the range 0.01–0.07 g/L, 25–38% tested positive for EtG regardless of whether the AUDIT score was positive (≥8 points) or negative.
Monitoring abstinence in subjects undergoing alcohol detoxification programmes is important, but can be difficult using blood or urine ethanol measurements due to the relatively short period of time these markers remain positive following alcohol ingestion. In a group of 139 detoxified alcohol-dependent patients followed up for 12 weeks after discharge from inpatient treatment, 28% of subjects denying relapse tested positive for EtG and EtS measured by LC-MS/MS. 54 Similarly, four out of 30 patients, in whom neither clinical assessment nor routine laboratory testing suggested relapse, tested positive for EtG in urine with concentrations ranging from 4200 to 196600 μg/L. 55 However, only the subject with the highest urine EtG concentrations had detectable EtG in serum. In a double-blind placebo-controlled study of oral acamprosate, 56 alcohol-dependent subjects (30 men) gave urine samples at baseline and at weekly intervals for measurement of EtG and EtS. On the first day, 72% of subjects tested positive, which reduced to 31% after three weeks with no difference between the acamprosate and placebo groups. Significantly, 28% of samples from subjects who denied alcohol consumption in the day prior to testing were positive for EtG and EtS. 56 In a similar study of 24 outpatients undergoing treatment for alcohol or drug dependency, urine EtG/EtS was compared with self-reporting. For 211 cases with both self-report and EtG/EtS results, there was 87% concordance. In one patient, samples were only positive for EtS. 57
Liver transplantation for treatment of end-stage liver disease resulting from alcohol misuse has often been considered to be controversial as 20–25% of subjects return to harmful drinking after surgery. Abstinence is a requirement to remain on the transplant list, but is difficult to assess as GGT and CDT are affected by the liver disease per se and patients know to refrain from drinking in the 24–36-h period prior to testing by breath, blood or urine alcohol measurements. In a study of 18 liver transplant candidates who denied alcohol consumption, nine had positive EtG results in 24 (49%) of 49 urine samples whereas only one of 127 breath alcohol tests was positive. 58 In a cross-sectional study of 109 adult patients undergoing assessment for liver transplantation, 20% of subjects with alcohol-related liver disease had positive urine EtG and EtS results, but only 4% self-reported alcohol use. 59 In a larger study of 141 liver transplant candidates in Germany, urinary EtG had a positive predictive value of 89.3% and a negative predictive value of 98.9% for detecting alcohol consumption and was superior to CDT, MCV and GGT. 60 Theoretically, end-stage liver disease could result in reduced production of EtG and EtS, although as these are minor pathways of alcohol metabolism, there is no evidence that such a reduction occurs.
Subjects undergoing opioid maintenance therapy often abuse alcohol, but often deny it with negative AUDIT scores. Urine and hair EtG measurements identified cases of excess alcohol intake in subjects on a methadone maintenance programme that would have been missed using self-report alone. 61–63 In health-care professionals recovering from substance-related disorders, complete abstinence from drugs, including alcohol, is required before they can return to work. Random urine testing is usually incorporated into such programmes, and in one study of 100 urine samples collected, none tested positive for alcohol but seven were positive for EtG (range 500–196,000 μg/L). 64 EtG has also been measured in oral fluid in an employee recruitment scheme in Norway with 2.1% testing positive (EtG > 2.2 µg/L). 65
Fetal alcohol syndrome (FAS) and fetal alcohol spectrum disorder (FASD) are recognized as causes of congenital abnormalities, cognitive dysfunction and developmental delay. It is estimated that FAS affects about 2/1000 and FASD 9/1000 live births in the developed world; however, diagnosis after birth can be difficult. EtG and EtS have been measured by LC-MS/MS in meconium samples from the infants of 177 randomly selected women from Italy and Spain. EtG was detectable in over 80% of samples while EtS was only found in 50%. A cut-off of 2 nmol/g was found to have 100% sensitivity and specificity to distinguish heavy maternal ethanol consumption during pregnancy from occasional or no use (defined by questionnaire and meconium fatty acid ethyl ester concentration). 66 This cut-off was validated in a study from the same group using a subset of mothers who self-reported alcohol consumption during pregnancy. These workers also showed that neonatal hair EtG was a poor predictor of maternal alcohol intake. 67 Fatty acid ethyl esters (FAEEs) are esterification products of ethanol and fatty acids that have been implicated as mediators of alcohol-induced organ damage. They have been measured in blood and tissues as markers of alcohol intake. 68 A similar study of 602 meconium samples from a maternal health evaluation in Germany found that only 97 (16.3%) of cases had detectable EtG. In none of the 602 cases did the mothers report serious alcohol consumption and no evidence of FAS or FASD were found in the medical examination of the newborn infants. EtG was compared with FAEEs and a cut-off of 274 ng/g provided the best agreement between the two markers. Two outliers (EtG 10,200 and 82,000 ng/g) suggested heavy alcohol consumption that was not reported by the mothers. The authors concluded that the combined use of EtG and FAEE in meconium samples as markers of fetal alcohol exposure helps avoid false-positive and false-negative results. 69
Only one study has been published on the measurement of EtG and EtS in urine and hair in pregnant women. 70 One hundred and three Swedish women gave urine and hair samples for EtG, EtS and FAEE, and completed the AUDIT questionnaire. Twenty-six women (25.2%) were identified as possible alcohol consumers and seven subjects had hair EtG or FAEE concentrations highly suspicious of heavy drinking, although only one was positive according to the AUDIT questionnaire. This appears to be another example of under-reporting of alcohol consumption when self-report questionnaires are used.
Urinary EtG compared with 5-hydroxytryptophol/5-hydroxyindoleacetic acid ratio
Acute alcohol intake alters the normal metabolism of serotonin (5-hydroxy-tryptamine) to 5-hydroxyindoleacetic acid (5-HIAA), resulting in the formation of 5-hydroxytryptophol (5-HTOL), albeit it at only 1% of the 5-HIAA concentration. The ratio of 5-HTOL to 5-HIAA in urine was shown to be a more sensitive and specific marker of alcohol ingestion than urine ethanol or methanol, remaining positive for 6–15 h after the BAC had returned to baseline values. 71 A number of groups have compared urine EtG with the 5-HTOL/5-HIAA ratio for the detection of acute alcohol intake. 20,72,73 In all cases, both tests had a high specificity for alcohol intake, but EtG had a longer time window of positivity compared with the 5-HTOL/5-HIAA ratio of up to 48 h.
The largest study incorporating EtG measurements was the World Health Organization/International Society for Biomedical Research on Alcoholism (WHO/ISBRA) Study of State and Trait Markers of Alcohol Use and Dependence that recruited 304 subjects from Brazil and Australia of whom 158 were diagnosed as lifetime alcohol abusers. 74 The study also measured the 5-HTOL/5-HIAA ratio in urine, blood ethanol and serum CDT and GGT. The method used for urine EtG was LC-MS with a limit of detection of 100 μg/L. The authors concluded that EtG was a good candidate for a sensitive, specific and reliable marker of recent alcohol intake.
Hair EtG
Analysis of drugs of abuse in hair samples has long been used to attempt to identify chronic misuse over a period of weeks to months. It was, therefore, understandable that there has been considerable interest in whether hair EtG could play a similar role in the identification of alcohol misuse over similar time periods, i.e. longer than that for which CDT is useful. Initial methods developed using GC-MS were hampered by limits of quantitation in the range 0.5–2.0 ng/mg hair. 46,75,76 Over the past decade, the LC-MS methods developed for measurement of urine EtG have lowered this to 2–10 pg/mg hair. 77–84 Cut-off values for EtG of between 4 and 30 pg/mg hair have been proposed to distinguish between social (<20 g ethanol a day) and heavy drinkers (>40 g ethanol a day), achieving sensitivity and specificity values between 90% and 95%. In subjects with low-to-moderate alcohol intake, defined as daily consumption of 16–32 g alcohol over a three-month period, the maximum hair EtG concentration was 11 pg/mg. These workers propose an abstinence threshold of <7 pg/mg and an excessive consumption threshold of >30 pg/mg. 85 The latter cut-off has been applied to a fitness to drive following previous alcohol problems programme and produced a higher positive rate than CDT. 86 Because of concerns about false-positive hair EtG results in cases that could have legal implications (fitness to drive, workplace testing, child custody, etc.), several groups have recommended a combination of hair EtG and FAEE measurements. 87,88
In most studies, hair samples were prepared for analysis by organic solvent treatment and sonication. A concern in hair analysis is the effect that hair treatments might have on the concentration of the target analyte. One study that treated hair sample in vitro with a commercially available bleaching solution showed that EtG in hair completely disappeared after treatment. 89 EtG has also been detected in postmortem hair samples together with tissue samples (gluteal and abdominal fat, liver and brain) from intoxicated subjects. 90 It is unclear, however, as to why EtG was not detected in the liver and gluteal fat of one subject who died intoxicated.
Conclusions
Although EtG was first described as a metabolite of ethanol in man in 1967, 91 it required the significant advances in analytical techniques, particularly mass spectrometry, that have taken place over the past 10–15 y for accurate and reliable methods for the measurement of EtG and EtS in biological samples to be achieved. Published data now include measurement of EtG in more than 5000 samples of urine, hair and serum from over 2500 individuals. These data suggest that EtG has potential as a marker of high sensitivity and specificity for the detection of alcohol misuse in a variety of settings, both in clinical and forensic medicine.
As a non-invasive marker, EtG in urine or hair could have a role in screening, diagnosis and monitoring treatment in selected groups of subjects or in general population studies. Urine EtG remains positive for periods of up to 48–72 h following heavy alcohol consumption; therefore, providing a marker with an intermediate timeframe between ethanol measurements and GGT/CDT. This may prove particularly useful in workplace monitoring schemes, prior to and following liver transplantation, alcohol detoxification programmes, and in the identification of neonates at risk of FAS/FASD.
The development of an immunoassay for EtG which can be automated could provide the opportunity for larger studies which would contribute to our understanding of the biology of these markers. However, the concern that the presence of certain bacteria in urine could lead to both false-positive and false-negative results from an immunoassay for EtG, but not for EtS, means that positive results from an immunoassay for EtG should be confirmed using chromatographic methods with consideration of the EtG:EtS ratio. Further work is going to be needed before the full potential of these direct ethanol biomarkers can be realized. In particular, there are currently few clinical studies on EtS compared with EtG due to the requirement for the use of LC-MS/MS methods for determination of EtS. It may transpire that EtS on its own would be a suitable marker of alcohol misuse, but this will need to be demonstrated in well-designed and adequately powered clinical studies.
DECLARATIONS
