Abstract
Background
Phlebotomy is a potential cause of preanalytical errors. We have observed phlebotomy in routine practice in a busy Emergency Department, to see how current practice compares with optimal blood sampling.
Methods
Phlebotomy episodes were audited and compared with standard procedures. A computer-based search of the number of haemolysed samples from Emergency Medicine and hospital inpatients was reviewed.
Results
Four different ways of taking blood were observed: cannulation and a syringe (38%), cannula with evacuated tube and adaptor (42%), syringe and needle into vein (14%) and evacuated tube system used conventionally (6%). Where a syringe was used, two methods of transfer into the sample tube were observed; needle kept on with cap piercing (77%) and needle and evacuated cap both removed (23%). On 20 out of 50 phlebotomy episodes (40%), the potassium-EDTA tube was filled prior to the biochemistry serum gel tube. A search of the laboratory computer records for ward-based phlebotomy found 30 of 1034 samples were haemolysed (2.9%). In the 50 phlebotomy episodes in the Majors area of the Emergency Department, 24% produced a haemolysed sample (
Conclusions
This study has shown that phlebotomy techniques in the Emergency Department deviate from standard practice significantly. This may well be a reason for the much higher frequency of haemolysed samples and with the wrong order of collection the possibility of potassium-EDTA-contaminated samples.
Introduction
Phlebotomy techniques are an important preanalytical factor that should be controlled to ensure that results most closely reflect patient status. Emergency Medicine is an environment where many different staff work for short periods of time and inherently this is a difficult area to ensure that optimal phlebotomy techniques are practised. Junior medical staff take many of the blood samples and their phlebotomy techniques reflect variation in their training which includes teaching components during the medical degree, together with ‘on the job’ practice in later medical student years.
Previous studies have shown that contamination of biochemistry samples with potassium-EDTA is relatively common. 1–4 It is suggested that this is linked to poor phlebotomy practice, with incorrect order of draw of samples, where the potassium-EDTA tube is filled prior to the biochemistry serum gel tube. Such contamination can lead to erroneous results for analytes with raised potassium due to introduction into the serum tube, and reductions in calcium and magnesium and other less commonly measured metal ions due to chelation effects of EDTA. 2 However, these studies lack direct observational evidence that incorrect order of draw is commonly practised in clinical areas. Studies instead rely on measuring the presence of EDTA in samples, where high potassium or low calcium indicates the possibility of contamination. 1–4 Indeed, one study looked at the incidence in a whole week's worth of samples by measuring potassium-EDTA in every sample received. 1
We have looked at the variation in phlebotomy practice in the Majors area of our Emergency Department. We have studied the basic techniques and have also considered the order of draw of samples. The incidence of haemolysed samples from the Majors area of Emergency Medicine as well as the whole department was compared with the frequency seen in hospital inpatients.
Methods
The phlebotomy system in place throughout SWBH NHS Trust is the Vacuette evacuated tube system (Greiner Bio-One, Stonehouse, UK). Only evacuated tubes are available for use in clinical areas, although syringes are also freely available. Hospital phlebotomy protocols clearly state the techniques that should be used and in particular the order that tubes should be filled. The Vacuette system comprises blood collection tubes, double-ended needles and adaptors and if using a cannula, a single-needle cannula adaptor should be used as provided and not combined with the use of alternative syringes and needles.
The conventional way of using the Vacuette system according to the Greiner Bio-One customer information leaflet 5 is as follows: For taking blood directly from a vein a multisample double-ended needle and tube holder are used. After one end of the needle is screwed into the holder the other end of the needle is inserted into the vein. Tubes are then inserted into the holder by puncturing the rubber diaphragm over the needle and the tube fills itself by vacuum suction. For drawing blood from a venous cannula a single-ended needle is provided with the other end fitting onto the cannula. Once this adaptor is in place on the cannula, tubes are filled in the same manner as the conventional venous approach. After filling, tubes are mixed and additional tubes placed in position as required by the repertoire of tests requested. The order of tube filling is described in the SWBH NHS Trust Phlebotomy Guide and relevant to this study the order of drawing blood should be: serum gel tube prior to potassium-EDTA tubes.
The Emergency Department is split into ‘Minors’ and ‘Majors’ areas, with more serious presentations being triaged to the Majors side of the department. Fifty phlebotomy episodes were observed at random over a two-week period in the Majors area of the Emergency Department as part of an agreed clinical audit. At the time of observation, the patient's registration number was collected and an observation form used to detail precise ways that the phlebotomy exercise was undertaken. Variables recorded included the basic techniques used and the order of draw/fill of the blood bottles, with particular reference to the order of the biochemistry serum gel tube in relation to potassium-EDTA tubes. Where the sample was not collected directly into the evacuated tubes, the time taken between sampling and final depositing in the tube was recorded.
Once all 50 phlebotomy episodes had been studied, sample details were tracked in the laboratory. The Majors area unit staff were given no indication of which of the aspects of their phlebotomy techniques were being considered. Samples were sent to the laboratory in the normal way for analysis and reporting. At a later date, results from the 50 phlebotomy episodes were reviewed on the laboratory computer to determine which ones were haemolysed at the time of analysis. Haemolysis was determined in the laboratory using the ‘H’ index on the Roche Modular P800 system (Roche Diagnostics Ltd, Sussex, UK) with an index cut-off above 70, being the level that a potassium result is reported as unavailable.
In the audit of phlebotomy practice in the Emergency Department, we assessed observed practice against the following standards:
Phlebotomy technique complied with the manufacturer's guidance and also the local phlebotomy instructions given in the phlebotomy operating procedure; Order of draw of the blood tubes complied with the guidance.
A laboratory computer search was performed of all blood samples taken in the Emergency Department and also on inpatient wards over a seven-day period within the two-week period of the phlebotomy observational audit and the number of haemolysed samples compared in these groups.
Statistical analysis was performed using the chi-squared test using GraphPad Software (GraphPad Software, Inc, San Francisco, CA, USA).
Results
Thirteen different staff were observed undertaking phlebotomy in the Majors area of the Emergency Department. These included a mixture of junior doctors, Emergency Medicine registrars, nurses and physicians' assistants as shown in Figure 1. The phlebotomy techniques used by these operators are summarized in Table 1. Despite the ready availability of an evacuated tube system, including the manufacturer's cannula adaptors, 52% of samples were taken using a syringe.
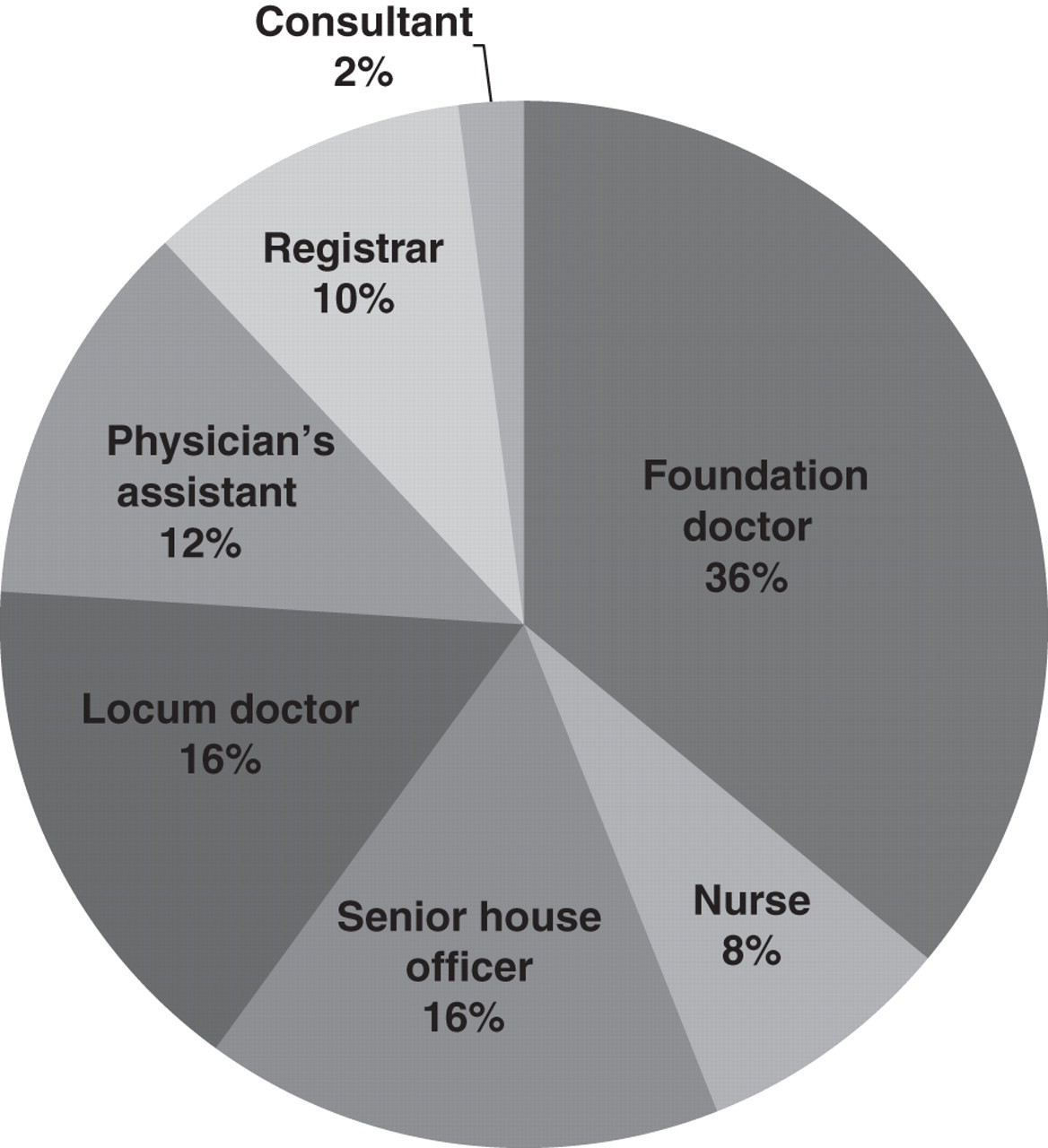
Analysis of the different types of medical staff that undertook phlebotomy in the Majors section of the Emergency Department
Variation in phlebotomy technique practised in the Majors area of the Emergency Medicine Department
Table 2 summarizes how the syringe-taken blood samples are transferred to sample tubes. Where a cannula and syringe were used, the majority of users added a needle after drawing the blood and then either pierced the tube cap or alternatively removed the cap to squirt the blood into the tube. Where a needle and syringe was used, then on all but for one occasion the blood was added to the tubes by cap piercing.
How blood tubes are filled when they are not the primary receiver of samples
Non-standard techniques involving the transfer of samples between syringes and tubes were undertaken away from the patient in 44% of the 50 observations. This added in a time delay before all the blood samples were in the correct tubes which ranged from 30 to 480 s, with an average time of 204 s (SE = 20.4).
In Table 3, the number of haemolysed samples in the Emergency Medicine Department is seen to be over three times greater than inpatient samples, which are mainly collected by hospital phlebotomists. Furthermore, in the study of the 50 phlebotomy episodes in the Majors area, the proportion of haemolysed samples was much greater at 24% (
Comparison of haemolysed samples in the Emergency Medicine Department and hospital inpatients
*
The analysis of the order of tube filling is restricted to consideration of the biochemistry and haematology samples. In Table 4, it can be seen that of the 49 episodes, where both a serum and potassium-EDTA tube was filled, the correct order of fill was practiced in only 51%. In two cases (4%), the biochemistry serum gel tube was topped up using a syringe and needle after filling the potassium-EDTA tube, with operators explaining that this was because they were worried that the tube did not contain enough blood. Either due to incorrect order of draw or revisiting the serum sample tube, a total of 49% of samples taken in the Majors department risked potassium-EDTA contamination.
Order of fill of evacuated tubes with particular reference to the potassium-EDTA and biochemistry tube (
Discussion
Preanalytical factors are an important potential cause for erroneous laboratory results. This study has seen a number of deviations from the manufacturer's recommended phlebotomy techniques for an evacuated tube system. Some of these alternative practices at the point of blood taking lead on to further issues in the way that the blood samples are then handled. These non-standard approaches seem to reflect personal choice of the operator, perhaps influenced by their original phlebotomy training rather than any difference in patient presentation.
Clearly, the Emergency Department has a much greater proportion of haemolysed samples with the situation even more extreme in the Majors area. This means that potassium cannot be reported and a number of other tests may also not be available, necessitating that the patient is rebled using up valuable time and resources. Hospital inpatient samples are mainly taken by trained phlebotomists using the conventional Vacuette system without a cannula. The percentage of inpatient haemolysed samples was just 2.9%, dramatically less than the 10.7% seen in samples from the Emergency Department. In the Majors area of Emergency Medicine, haemolysed samples were seen in 24% of phlebotomy episodes. The key difference in the Majors area is that the standard practice is to cannulate the patient and then draw the blood samples from the cannula. The Trust Infection Control Policy insists that the smaller diameter pink cannulas (20G) are used and pulling blood through such small diameter tubing may increase the likelihood for haemolysis. In our study, the use of a cannula and the appropriate adaptor with blood going directly into the evacuated tube gave the highest number of haemolysed samples. In laboratory experiments on haemolysis, pulling and pushing blood through fine lines such as needles and cannula are used to produce haemolysis and so it is not that surprising that cannula-drawn blood samples show more haemolysis. Other factors increasing haemolysis in samples, such as tourniquet time, number of attempts at venepuncture and methods of sampling have previously been shown to be related to increased haemolysis rates. 6 In our study, the use of cannula is demonstrated as something that needs to be considered further as a major cause of heamolysis in samples collected in the Emergency Department.
Incorrect order of drawing or filling the blood tubes for biochemistry and haematology is well known by phlebotomy and laboratory staff to lead to a high risk of contaminated samples and erroneous results. While we have not looked for the presence of potassium-EDTA contamination directly in samples in this study, one recent study 1 showed an overall incidence of 0.46% in potassium-EDTA contamination of hospital samples. Potassium-EDTA contamination of the biochemistry serum gel tube can increase the potassium concentration directly and also lead to chelation of metal ions, where, for example, calcium and magnesium concentrations can be erroneously lowered. With the very high level of incorrect order of draw shown by our audit, it would be very relevant to measure EDTA directly in this patient cohort in a future study.
The use of evacuated tubes incorrectly has included tubes being filled from a syringe. In all methods of this technique there is a time delay in filling the sample tube that often includes the transfer of the blood away from the patient. This delay creates the potential for the clotting process to commence in those samples that are intended to ultimately contain an anticoagulant. There is also greater risk of labelling the tubes incorrectly when done away from the patient. Where the evacuated tubes have their caps removed for blood filling, there is an increased risk for the cap to dislodge in transit, especially in an air tube system as the residual vacuum helps keep the cap on.
The busy Emergency Department is a difficult environment to apply uniform protocols for an area such as phlebotomy and this is exacerbated by the four-monthly turnover of junior medical staff. There is little awareness of the potential for seriously misleading results to be produced when simple changes in technique are made. There is a clear preference for using a syringe to take blood samples in Emergency Medicine even among junior staff, which in turn leads to the phlebotomy blood collection system being used incorrectly. In particular, the importance of the order of draw was clearly not appreciated by the staff concerned. When those observed had a feeling that order of drawing (or filling) was one of the key observations being made, they were still as likely to fill the potassium-EDTA sample prior to the biochemistry serum gel tube.
Haemolysed and contaminated samples are a cause of frustration for clinical and laboratory staff alike and are potentially detrimental to patient care. At best, repeat samples must be taken and at worst, incorrect treatment decisions due to erroneous results can occur. Results of this study, including the audit of phlebotomy practice in the Emergency Department, have led us to recommend that straightforward protocols and greater input from the hospital phlebotomy service in training staff are key to improving this situation. However, something more fundamental should also be considered for Emergency Department phlebotomy with perhaps a smaller group of staff dedicated to performing this function.
DECLARATIONS
