Abstract
Background
There are significant differences in plasma parathyroid hormone (PTH) results obtained by current immunoassay methods. However, many clinical guidelines relevant to patients with chronic kidney disease (CKD) that recommend PTH target values do not take account of these differences. This raises major questions about the validity of the evidence underpinning current use of PTH measurements in the management of CKD as well as of published relevant audit data.
Methods
PTH was measured in plasma from patients with CKD in six commercially available immunoassays. The initial pilot study included 19 patients while 98 patients were included in a second extended study. Data from the second study were analysed by regression analysis to obtain assay-specific targets for each immunoassay.
Results
Although similar PTH reference intervals are provided by most manufacturers, both studies confirmed substantial between-method differences in observed PTH for all patients, with results varying by as much as 4.2-fold between the lowest and highest reading methods. These differences were sufficient to have treatment implications for 79% of the patients in the pilot study. Applying the assay-specific targets derived here to results from the extended study decreased treatment misclassifications from 53% to 12%.
Conclusions
Existing between-method differences in PTH measurements clearly have treatment implications. International initiatives to address these differences are in progress and will require support and input from all stakeholders. Adoption of assay-specific target values such as those reported here provides a convenient and practical interim solution that should lead to significant improvement in patient management, while also enabling meaningful audit.
Introduction
Since 2000, guidelines used in the management of chronic kidney disease-mineral and bone disorders (CKD-MBD) have made reference to target ranges for plasma parathyroid hormone (PTH) concentration; yet there is compelling evidence to suggest that variation between current immunoassays for the measurement of PTH leads to widely disparate PTH results. Here we present data on the variability of PTH measurements and consider the clinical implications for patients with CKD, including the potential for misclassification. We propose the adoption of target values for PTH concentrations that are specific to the immunoassay used to derive them. This will enable meaningful and consistent interpretation of PTH results in relation to existing guidelines. This work was conducted by the Scottish Clinical Biochemistry Managed Diagnostic Network in association with the Scottish Renal Registry, but should have universal application.
Current guidelines
As the glomerular filtration rate (GFR) falls below 50 mL/min, reduced activation of vitamin D leads to hypocalcaemia, a problem that is exacerbated by rising phosphate concentrations caused by falling renal clearance. The resulting increase in PTH is an appropriate physiological response aimed at restoring normal serum calcium and phosphate concentrations. Moreover, in the presence of uraemia, skeletal resistance to PTH develops, such that higher levels are required to maintain normal bone turnover. 1 However, the homeostatic mechanisms invoked by the rising PTH include increased tubular calcium re-absorption, enhanced renal phosphate excretion and increased vitamin D activation – all of which rely on functioning renal tissue. Thus as the GFR falls further, these systems also fail, leaving PTH-mediated osteoclastic resorption of bone as the only mechanism by which calcium concentrations can be maintained.
Persistent stimulation of the parathyroid glands over time causes structural changes resulting in reduced sensitivity to the normal feedback loops, unregulated release of PTH and hence hypercalcaemia. Untreated, this causes bone pain and fractures, while the uncontrolled release of calcium into the circulation may lead to soft tissue and vascular calcification – a cause of considerable morbidity and mortality. Once this stage of tertiary hyperparathyroidism is reached, the only effective treatment is parathyroidectomy. Guidelines have been devised to improve treatment of these biochemical abnormalities from an early stage in order to minimize the risk of such outcomes. 2–4
Guidelines from the Kidney Disease Outcomes Quality Initiative (K/DOQI) published in 20032 recommended that PTH concentrations in dialysis patients should be maintained within the target range of 150–300 ng/L (approximately 15.8–36.8 pmol/L using a conversion factor of 9.5 derived using the molecular weight of PTH). These guidelines formed the basis for diagnosis, treatment and monitoring of CKD-MBD until 2009, when they were superseded by recommendations made through the Kidney Disease: Improving Global Outcomes (KDIGO) initiative. 3 The KDIGO guidelines expressed target ranges as multiples of the upper limit of normal (ULN) rather than in terms of concentration. The latest edition of the European Best Practice Guidelines 4 is essentially in accord with the KDIGO recommendations.
The Renal Association, however, has always expressed the target range as multiples of the ULN for the immunoassay used. The initial recommendation of two to four times the ULN, based on previous research, 5,6 was revised in January 2011 to 2–9 times the ULN. 7 The National Institute for Health and Clinical Excellence (NICE) has also published guidance for the prescription of cinacalcet, an expensive drug used in the treatment of refractory secondary hyperparathyroidism. 8 NICE advises use of the drug ‘only in patients unfit for surgery whose PTH is >85 pmol/L’.
Neither the K/DOQI nor the NICE guidance take into account the well-described variation in PTH concentrations observed in different immunoassays. 9 The use of concentrations expressed as multiples of the ULN, as recommended by KDIGO and the Renal Association, appears to represent an improvement in practice, as it should allow for these differences. However, this approach requires accurate reference ranges for PTH appropriate to each immunoassay and obtained by measuring PTH in a reference population of healthy subjects. Establishing a reference population of healthy subjects is in itself problematic since PTH concentrations are affected by vitamin D status, and vitamin D insufficiency is common in the general population. Vitamin D insufficient subjects should therefore be excluded from the reference population. 10 Given these difficulties, clinicians and laboratories frequently rely on data provided by immunoassay manufacturers which may also be unreliable. It has, for example, recently been reported that two immunoassays yield PTH concentrations for the same patient specimens that differ by approximately 30%; yet the manufacturers recommend similar upper limits of normal (68 and 65 ng/L [7.2 and 6.8 pmol/L], respectively). 11
External quality assessment (EQA) data also reveal the extent of variability in PTH concentrations obtained in different immunoassays. The United Kingdom National External Quality Assessment Service (UK NEQAS) provides EQA for PTH. Lyophilized samples of pools of human plasma are circulated bi-monthly to 310 participating laboratories worldwide, the samples are analysed for PTH using the laboratory's routine immunoassay and results then returned to UK NEQAS [Edinburgh] for analysis. Significant differences in results are evident, 12 but are not reflected in the manufacturers' quoted reference intervals, which are very similar. In collaboration with the Scottish Renal Registry, the Scottish Clinical Biochemistry Managed Diagnostic Network undertook a pilot study to investigate the variability between different immunoassay measurements of PTH, with a view to establishing a nationwide audit of bone mineral metabolism in the Scottish CKD population.
Methods
Pilot study
Twenty-one haemodialysis patients regularly attending the Renal Unit at Dumfries and Galloway Royal Infirmary (DGRI) consented to give 5 mL of blood for the study. Samples were collected into tubes containing ethylenediaminetetraacetic acid 48 h after the previous dialysis. Samples were taken immediately to the DGRI biochemistry laboratory, where each was centrifuged. The plasma was collected, divided into five separate 1-mL aliquots and frozen at −80°C. The DGRI laboratory retained one set of samples while the others were distributed on dry ice to four other laboratories to enable PTH measurement by the five PTH immunoassays used at the time of the study by Scottish laboratories serving renal units. These immunoassays were the Beckman Access DxI (Beckman Coulter UK Ltd, High Wycombe, Buckinghamshire, UK), DiaSorin Liaison (DiaSorin Ltd, Bracknell, Berkshire, UK), Roche Modular E170 (Roche Diagnostics, Burgess Hill, West Sussex, UK) and Siemens ADVIA Centaur and Siemens Immulite 2000 (Siemens Healthcare Diagnostics, Camberley, Surrey, UK). All samples were thawed immediately prior to analysis and PTH results subsequently returned to UK NEQAS [Edinburgh] for statistical evaluation. Results for two patients who had previously undergone parathyroidectomy and had undetectable PTH concentrations were excluded from the analysis.
Extended study
Essentially the same protocol was used for a second study in which plasma was obtained from 106 patients attending the Renal Unit at Glasgow Royal Infirmary. Material remaining after routine testing was frozen and PTH was subsequently measured in six immunoassays, including the five represented in the pilot study and the Abbott Architect (Abbott Diagnostics, Maidenhead, Berkshire, UK). Eight specimens were excluded as some results were not available due to insufficient specimen or some results had been reported as either ‘less than’ or ‘greater than’. Statistical analysis was undertaken using the method of Passing and Bablok. 13
Results and discussion
The observed PTH concentration varied widely between immunoassays. Between-method differences observed for a typical single patient specimen are shown in Figure 1. The differences between the lowest and highest reading immunoassays ranged from 1.4- to 4.2-fold (mean 2.8-fold) although the manufacturer's reference intervals for the lowest and highest reading immunoassays were similar (1–6.5 and 1.2–7.6 pmol/L, respectively). The proposed Scottish audit of PTH use in clinical practice would consequently be meaningless without taking account of the method used. Even more importantly, the study suggested that individual patients might be treated differently purely as a result of the magnitude of the discrepancies between PTH immunoassays.
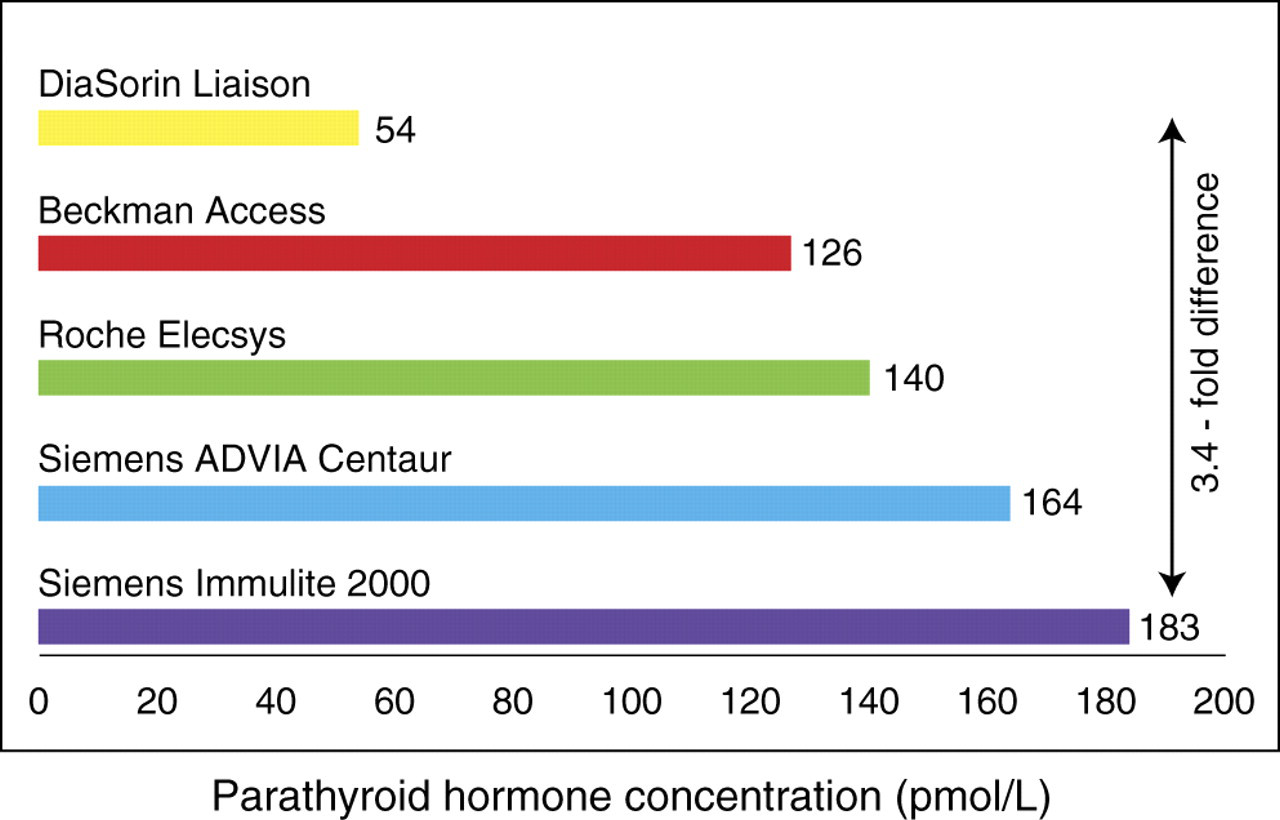
Between-method differences in the concentration of parathyroid hormone (PTH) observed in a typical single patient specimen
Pilot study – treatment implications
The Renal Association guidelines in place at the time of the study recommended a target range for PTH of 2–4 times the ULN of the immunoassay used. A PTH concentration less than twice the ULN is associated with adynamic bone disease, 5 a condition resulting in bone pain, fractures, deformity and a tendency to hypercalcaemia. In such patients, giving active vitamin D analogues should be avoided to prevent further suppression of PTH and low bone turnover. Two study patients, whose PTH according to the lowest reading immunoassay was less than twice the ULN, would not have been given vitamin D by this criterion had the lowest reading immunoassay been used. However, using the highest reading immunoassay, these same two patients would almost certainly have received vitamin D treatment to suppress an apparently excessively high PTH.
In addition to the cases of these two patients there were other potential treatment anomalies:
Five patients, who were within the Renal Association target range for PTH by the lowest reading immunoassay – and were therefore deemed to be receiving the correct treatment – would have had their treatment intensified (i.e. received an increased dose of alfacalcidol) if PTH had been measured by the highest reading immunoassay. For some of these patients, this might increase the risk of adynamic bone disease, while patients with hyperparathyroid bone disease may be under-treated; Eight patients almost certainly would have been maintained on medical treatment if PTH had been measured by the lowest reading immunoassay but may have been considered for cinacalcet or even parathyroidectomy according to the highest.
In summary, using the Renal Association guidelines, 7/19 (37%) of the study patients would have been considered to have a different category of bone turnover by the highest reading PTH immunoassay than by the lowest reading immunoassay. On applying the guidelines for cinacalcet and parathyroidectomy as well, treatment could have varied in up to 15/19 (79%) of the patients studied (Figure 2).
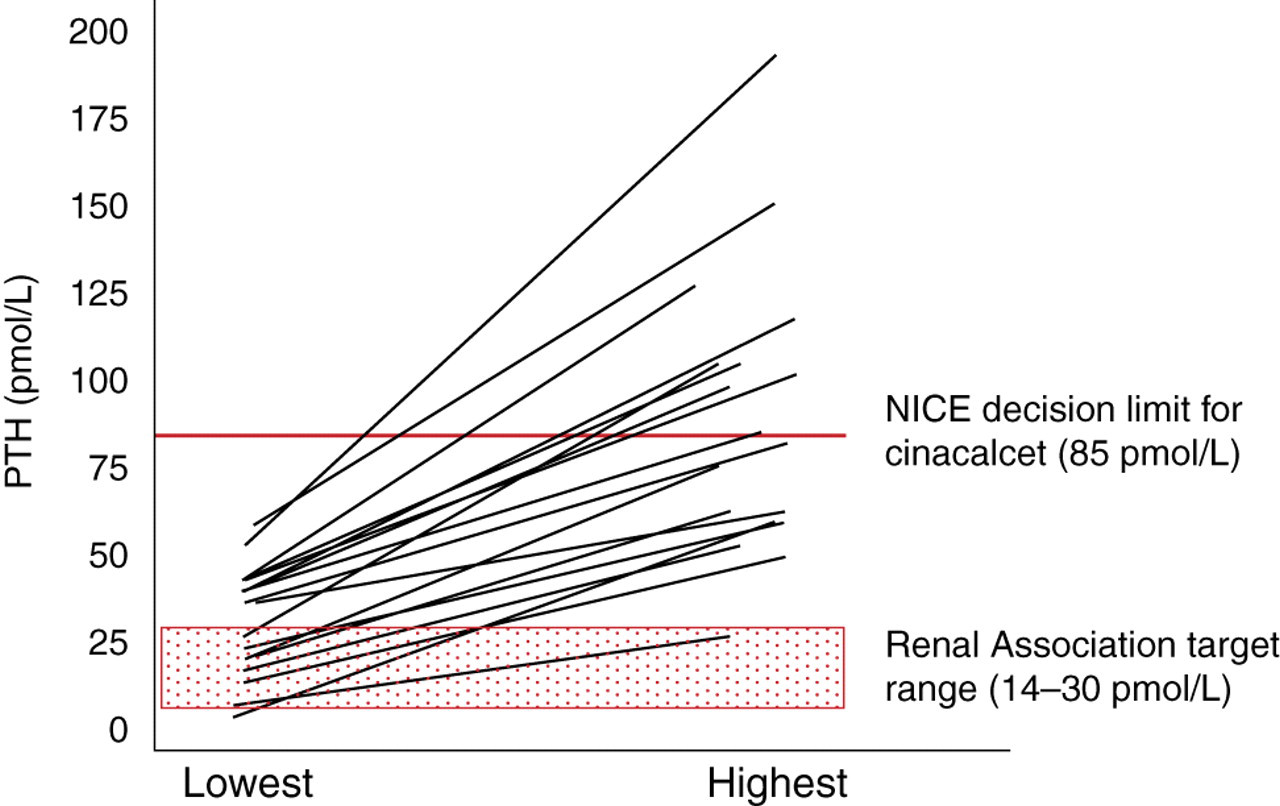
Treatment variation caused by comparing highest and lowest PTH concentrations in 18 patients. Each line represents a single study patient (shaded area, Renal Association target range for PTH in CKD patients; red line, NICE decision limit for eligibility for cinacalcet). NICE, National Institute for Health and Clinical Excellence; CKD, chronic kidney disease; PTH, parathyroid hormone
Extended study – rationale, protocol and results
The pilot study confirmed significant between-immunoassay differences in PTH that are not reflected in relevant reference intervals, and which could affect treatment decisions and invalidate renal audit data. A further study was therefore conducted with a larger cohort of patients in order to define better the differences between immunoassays. The full data for this study are shown in Supplemental Table 1 (this can be found at
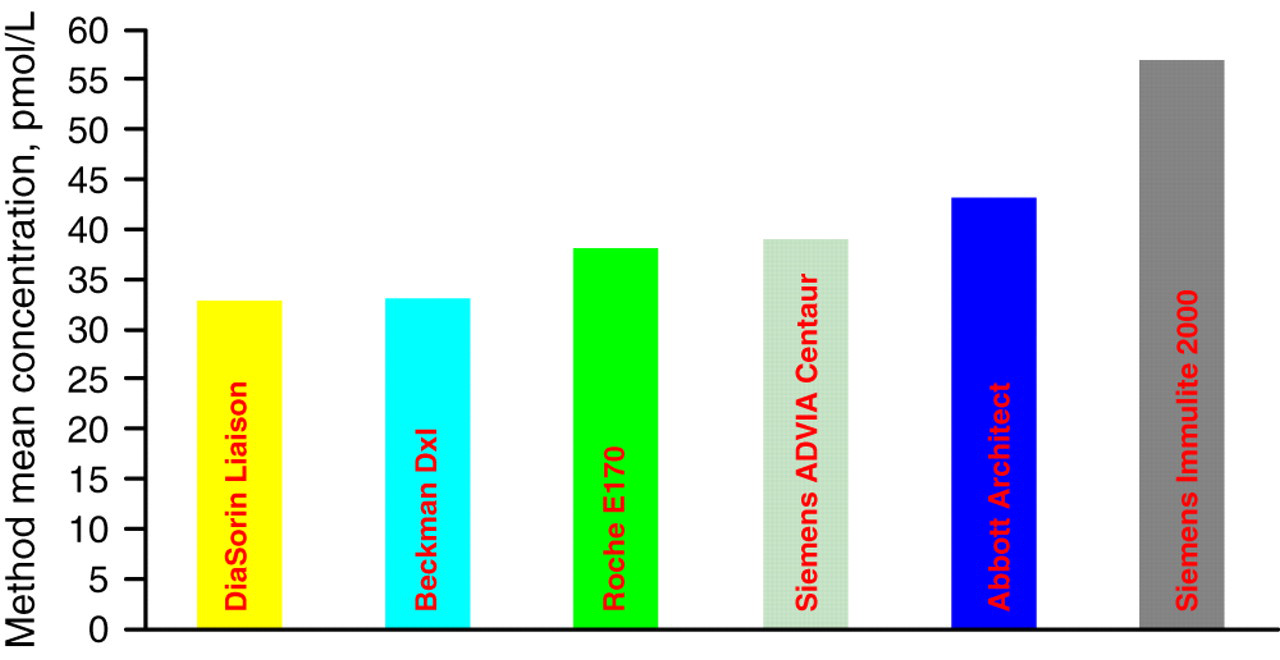
Method mean parathyroid hormone (PTH) concentrations observed in the individual patient specimens in the extended study
Method-specific target values for parathyroid hormone (pmol/L) proposed for interim use in the management of patients with chronic kidney disease
The values are computed from regression analysis and have been rounded to the nearest whole number as appropriate (see text for details)
*Baseline immunoassay for comparative purposes. See text for details
Extended study – treatment implications
While the variation between the lowest and highest PTH measurements was less marked in this larger cohort (mean difference, 1.8-fold; range, 1.2–2.7-fold), the number of misclassifications due to the variations was greater, with 53% (52/98) of patients being classified differently according to the highest and lowest reading immunoassays when manufacturers' reference interval data are used. However, on applying the assay-specific targets, the number of misclassifications was reduced to 12% (12/98). Of the original 19 patients in the pilot study, the misclassifications observed for seven (37%) patients when using the original PTH targets decreased to five (26%) using the assay-specific targets.
Discussion and interim recommendations
Only a bone biopsy can give a true picture of bone turnover and guide optimal treatment, 15 but undertaking regular bone biopsies for all patients is not practicable. PTH therefore remains the most widely used indicator of bone turnover in renal patients. Existing guidelines for the management of bone mineral metabolism in CKD patients are based on studies that show that very high PTH results are associated with high turnover bone disease and normal or low PTH results are associated with low bone turnover. Our data, however, demonstrate that, using current PTH target values, these classifications cannot be made reliably, due to the large discrepancies between PTH measurements, which can lead to inconsistencies in the management of CKD patients unless account is taken of method-related differences in PTH results.
In the pilot study, 17/19 patients were at risk of high turnover bone disease (PTH >4 times the ULN) according to the highest reading immunoassay, while only 11 were in this category according to the lowest reading assay. Applying the new assay-specific targets, both these immunoassays classified 11 of the 19 patients in this category. Similarly, in the larger study, use of the assay-specific targets improved agreement of the lowest and highest results significantly, with misclassifications decreasing from 53% to 12%.
An alternative approach suggested by Monge et al. 11 involves applying a method-dependent correction factor to each individual PTH result rather than correcting the ULN for method bias as in the present study. However the logistics of the former approach are more complex, with considerably greater risk of error in data handling.
The analytical differences reported here and elsewhere 9,11,14 raise major questions about the validity of the evidence underpinning current use of PTH measurements in the management of CKD patients. They may also help to explain conflicting views in the literature concerning PTH targets in CKD. Critically, they also suggest that unless audit programmes carefully record and take account of the PTH immunoassays used, any conclusions are likely to be unreliable.
Development of decision limits that are clinically fit for purpose will only be fully achievable when there is much better agreement among PTH methods and the methods are well-characterized. Such improvements in the quality and analytical standardization of PTH immunoassays will require coordinated international initiatives and will undoubtedly represent a major step forward. 16 In the interim, we recommend that all laboratories serving renal units in the UK adopt the method-specific targets for PTH reported here (Table 1). These values may require revision in the event of significant changes to assay performance, but their use should lead to more consistent patient care as well as enable meaningful audit, which is currently not feasible.
DECLARATIONS
