Abstract
Background
Serum plasmalogens (Pls) have gained interest in several clinical symptoms such as metabolic syndrome/atherosclerosis or Alzheimer's disease possibly because of their antioxidant properties. We have developed a highly sensitive and simple method to determine plasmenylcholine (PlsCho; choline plasmalogen) and plasmenylethanolamine (PlsEtn; ethanolamine plasmalogen) separately, using a radioactive iodine and high-performance liquid chromatography (125I-HPLC method). The present study reports the improvement and validation of 125I-HPLC method by introducing a quantitative standard (QS) and online detection with a flow γ-counter.
Methods
1-Alkenyl 2,3-cyclic glycerophosphate was prepared as QS from l-α-lyso plasmenylcholine by enzymatic treatment with phospholipase D. Online detection with a flow γ-counter was investigated to be available to quantify Pls. The method validation was carried out in terms of selectivity, sensitivity, linearity, precision, accuracy and recovery.
Results
Linearity was established over the concentration range 5–300 μmol/L for Pls and QS with regression coefficients >0.99. The accuracy and reliability were satisfactory. The method has been applied to the determination of human serum Pls from healthy subjects and the elderly with dementia or artery stenoses.
Conclusions
The improved 125I-HPLC method is useful as an autoanalytical system for a routine diagnostic test of human serum Pls.
Introduction
Plasmalogens (Pls) are glycerophospholipids containing a vinyl-ether linkage (–O–CH=CH–) at the sn-1 position of the glycerol backbone, and are widely distributed in human and animal tissues. 1 Although the physiological roles of Pls are not fully understood, the clinical significances of these phospholipids are recognized in relation to peroxisomal disorders. These diseases are often substantially deficient in Pls, because part of Pls' biosynthetic enzymes are localized in this organelle. 2 Recently, serum (or plasma) Pls have gained interest in several clinical symptoms such as metabolic syndrome/atherosclerosis 3,4 or Alzheimer's disease 5 other than peroxisomal disorders.
Serum Pls are mainly synthesized in and secreted by liver as a structural component of lipoproteins, 6 and potentially prevent lipoprotein oxidation relevant to atherosclerosis, probably due to the radical scavenging ability of the vinyl-ether double bond. 7,8 Thus serum (or plasma) Pls are considered as potentially useful biomarkers such as oxidative stress markers. Therefore, a simple and reliable method for the quantification of serum (or plasma) Pls is required for a routine diagnostic test.
The analytical methods for Pls have been developed principally based on the acid labile property of the vinyl-ether linkage of Pls, which leads to the sequential decomposition into fatty aldehydes and 1-lysophospholipids. The resultant fatty aldehydes are usually measured as dimethyl acetal derivatives using gas chromatography/mass spectrometry (GC/MS), 9 and 1-lysophospholipids are measured by two-dimensional thin-layer chromatography (TLC). 10 However, these methods are laborious and insufficient to accurately quantify Pls in a small amount of the serum (or plasma). More recently, liquid chromatography/mass spectrometry (LC/MS) has been applied to the analysis of Pls at the molecular species levels. 11 LC/MS analysis is a powerful method to provide substantial useful information, but the excess data are unlikely to be suitable for a routine diagnostic test, except for specific diagnostic molecular species for particular disorders such as Alzheimer's disease. 5
Our method has been developed based on the binding specificity of iodine to Pls. Although iodine molecule (I2) is well known to reversibly bind to the double bonds of unsaturated lipids, the principle of our method differs from it, and is based on the covalent binding of an iodine atom (I) to Pls. Triiodide (1-) ion (I3 −), which is a reactive iodine molecular form, reacts with a vinyl-ether double bond of Pls in methanol solution specifically and quantitatively. Although the precise reaction mechanism has still remained obscure, we speculate based on the result of MS analysis of the iodine-binding Pls, that an activated iodine molecule (I2) attacks on the double bond of the vinyl-ether moiety of Pls, and a single iodine atom (I) covalently binds to the double bond. On the other hand, other activated halogen species such as hypochlorous acid or hypobromous acid also preferentially react with Pls, but their halogenated products rapidly decompose into chloro- or bromine-fatty aldehydes and 1-lysophospholipids. 12 The difference in the reaction products between iodine and other halogen species may be derived from the size of the binding atom; iodine atom may induce the steric hindrance, and contribute to the structural stability of iodine-binding Pls.
Pls assay based on this principle has already been established. 13 Quantification of Pls is carried out by measuring the decrease in the absorbance at 355–360 nm, which reflects the extent of eliminated triiodide (1-) ion (I3 −) due to the reaction with a vinyl-ether double bond of Pls. We confirmed that the reaction molar ratio of I3 − to vinyl-ether bond is exactly 1:1, using several chemical compounds possessing a vinyl-ether bond as well as Pls. 14 However, the ‘cold’ iodine method has some problems of low detectable Pls concentrations and interfering substances in the extracted lipid samples from biomaterials like serum. The 125I-HPLC (radioactive iodine and high-performance liquid chromatography) method permitted the highly sensitive and accurate determination of serum (or plasma) Pls by using a radioactive iodine (125I) and removing interfering substances with HPLC. The primary advantage of this method enabled measurement of choline (PlsCho) and ethanolamine plasmalogens (PlsEtn) separately. 14
In the current study, the 125I-HPLC method has been markedly improved by introducing a quantitative standard (QS) and online detection with flow γ-counter. 1-Alkenyl 2,3-cyclic glycerophosphate was decided as a QS in view of both the same quantitative property of Pls and the proper retention time in the HPLC elution profile. Unfortunately, 1-alkenyl 2,3-cyclic glycerophosphate is poorly recovered in the lipid extraction from the serum (or plasma) when it is added in the serum as an internal standard. Therefore, 1-alkenyl 2,3-cyclic glycerophosphate was added following lipid extraction for the purpose of correcting the efficacies of the treatment process subsequent to lipid extraction and measurement. Next, before introducing a flow γ-counter into the HPLC system, the radioactivity of iodine-binding Pls had been measured by collecting HPLC elution fractions. Online detection with a flow γ-counter permitted a more precise and safe measurement of Pls-related radioactivity. The improved method has been fully validated in terms of selectivity, sensitivity, linearity, precision, accuracy, and has applied to the determination of human serum Pls.
Materials and methods
Materials and chemicals
Natural plasmalogens: plasmenylcholine from bovine heart (PlsCho), plasmenylethanolamine from bovine brain (PlsEtn) and l-α-lyso plasmenylcholine from bovine heart (LyPlsCho) were purchased from Doosan Serdary Research Laboratories (Toronto, Canada). Synthetic 18:0p/18:1- and 18:0p/22:6-PlsCho and PlsEtn (‘p’ indicates the carbon chain of sn-1 in plasmalogen with a vinyl-ether linkage) were obtained from Avanti Polar Lipids Inc (Alabaster, AL, USA). Other phospholipids were from Sigma-Aldrich Co (St Louis, MO, USA). Radioactive iodine (Na125I, 37 MBq/10 μL) was purchased from PerkinElmer Life and Analytical Sciences (Boston, MA, USA). Other chemicals were of the highest purity available from commercial sources.
Human serum was obtained from 120 healthy subjects (age-matched each 60 male and female) and 60 elderly subjects participating in a routine health examination. Serum was also provided by the elderly with dementia or artery stenoses. Written informed consent was obtained from all participants, and the studies were approved by several ethics committees. Serum was separated by centrifugation at 1500
‘Cold’ iodine method for determination of plasmalogens
The concentrations of QS and Pls standards were determined by the ‘cold’ iodine method 13 with slight modifications. Lipid samples were dissolved in alkali solution (pH 10.0) because of insufficient solubility of PlsEtn in neutral solution. Lipid samples (100 μL) were mixed with 0.6 mmol/L I-3% KI-50 mmol/L glycine/NaOH (pH 10.0, 100 μL), and placed in a dark room for 15–30 min at room temperature. After addition of 2.2 mL ethanol to the mixture, the absorbance of the mixture was measured with a spectrophotometer (V-530; Jasco Co Ltd, Tokyo, Japan) at wavelength 355–360 nm. A decrease in the absorbance of Pls samples compared with that of blank samples accurately reflects the amounts of Pls because of the equivalent reaction molar ratio of I3 − to vinyl-ether double bond of Pls.
Preparation of 1-alkenyl 2,3-cyclic glycerophosphate as QS
Five milligrams LyPlsCho from bovine heart was dispersed in equivalent volume (each 1.5 mL) of diethyl ether and sodium acetate buffer (100 mmol/L, pH 5.6) containing 10 U phospholipase D from Actinomadura sp. (Meito Sangyo Co Ltd, Nagoya, Japan) and 40 mmol/L CaCl2, and the mixture was stirred during incubation at 40°C for 3–6 h, according to the procedure as described in the literature
15
with slight modifications. The reaction products were checked by TLC: a disappearance of original spot corresponding to LyPlsCho (Rf; 0.28–0.36) and an appearance of new spot corresponding to 1-alkenyl 2,3-cyclic glycerophosphate (Rf; 0.62–0.72). The TLC plate (Silica gel 60 F254; Merck, Darmstadt, Germany) was developed by CHCl3/MeOH/NH3/H2O (65:35:5:5, by vol.), and the spots were detected with a primuline reagent. Subsequent to evaporation of diethyl ether in the reaction mixture by blowing N2 gas, the residual solvent was vigorously mixed with diethyl ether (1.2 mL) and ethanol (3.6 mL) for 10 min, followed by mixing intensely with additional 2 mol/L NaCl (9.0 mL) and diethyl ether (4.8 mL) for five minutes. Ether fraction including 1-alkenyl 2,3-cyclic glycerophosphate (upper layer) was collected by centrifugation at 1000 Scheme for preparation of 1-alkenyl 2,3-cyclic glycerophosphate as the quantitative standard (QS). LyPlsCho, l-α-lyso plasmenylcholine from bovine heart; PLD, phospholipase D from Actinomadura sp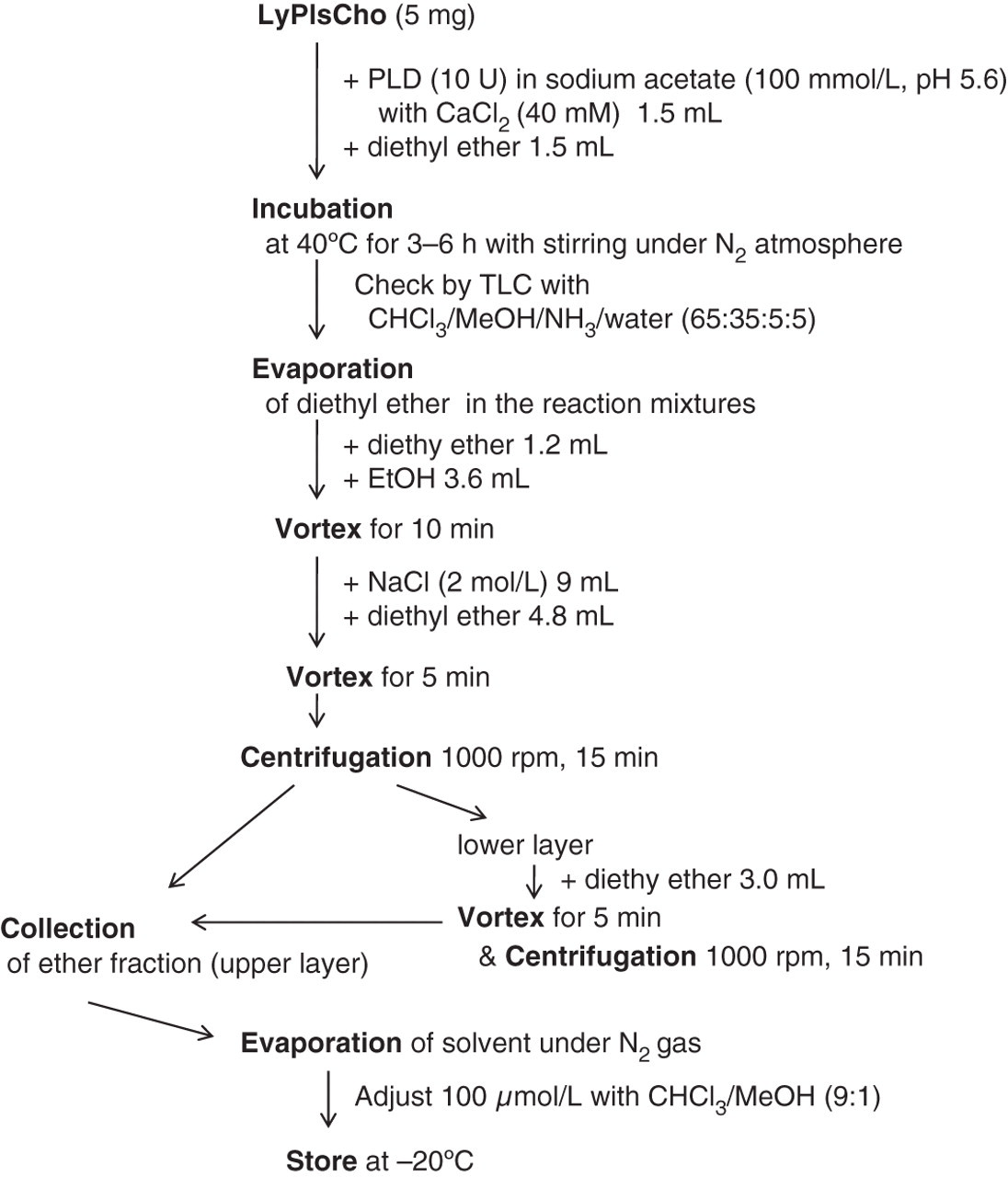
Sample preparation from human serum
Human serum (0.2 mL) was intensely mixed with diethyl ether (0.2 mL) and ethanol (0.6 mL) in a glass tube for 10 min. Following vigorous stirring of the mixture with additional ethanol (1.0 mL) and distilled water (1.6 mL), the ether fraction (upper layer) was collected by centrifugation (4000
125I-HPLC
Lipid samples (30 μL) were mixed with 125I reagent (5 μL), prepared as described previously, 14 in borosilicate glass, vial-attached, rubber covers for an autosample injector, and stood overnight at 4°C. It was confirmed that the reaction was completed till 12 h, and that the iodine-binding products were satisfactorily stable at least till 48 h at 4°C. Liquid chromatography separation was performed using a Prominence UFLC system (LC-20AD; Shimadzu, Kyoto, Japan) with a LiChrospher 100 Diol column (5 μm, 250 mm×4 mm i.d.; Merck) at 25°C and a flow rate of 1 mL/min. The auto sampler (SIL-20AC; Shimadzu, Kyoto, Japan) was kept at 10°C. The sample injection volume was 20 μL. The sample was eluted with isocratic mobile phase A/B (65:35, by vol.); A consisted of acetonitrile, and B consisted of acetonitrile/H2O/acetic acid/NH3 (80:19.7:0.2:0.1, by vol.). The total elution time was 15 min. Peak areas on LC were detected with a flow γ-counter (FC-3300; Bioscan Inc, Washington, DC, USA), and calculated by personal computer using SmartChrom software (KYA Tech Co, Tokyo, Japan).
Results and discussion
Extraction of Pls from human serum
Comparisons of Pls extraction efficiencies from human serum in different extraction methods
Pls, plasmalogens; PlsCho, plasmenylcholine (choline plasmalogen); PlsEtn, plasmenylethanolamine (ethanolamine plasmalogen)
Values are expressed as % compared with max value obtained in the examined methods
Dry&C/M (1:2): serum lyophilization followed by extraction with chloroform/methanol (1:2, by vol.)
Hex/EtOH (3:2): extraction with hexane/ethanol (3:2, by vol.)
EtOH/Ether (1:2): extraction with ethanol/diethyl ether (1:2, by vol)
Detection and quantification of125I-bound Pls with a flow γ-counter
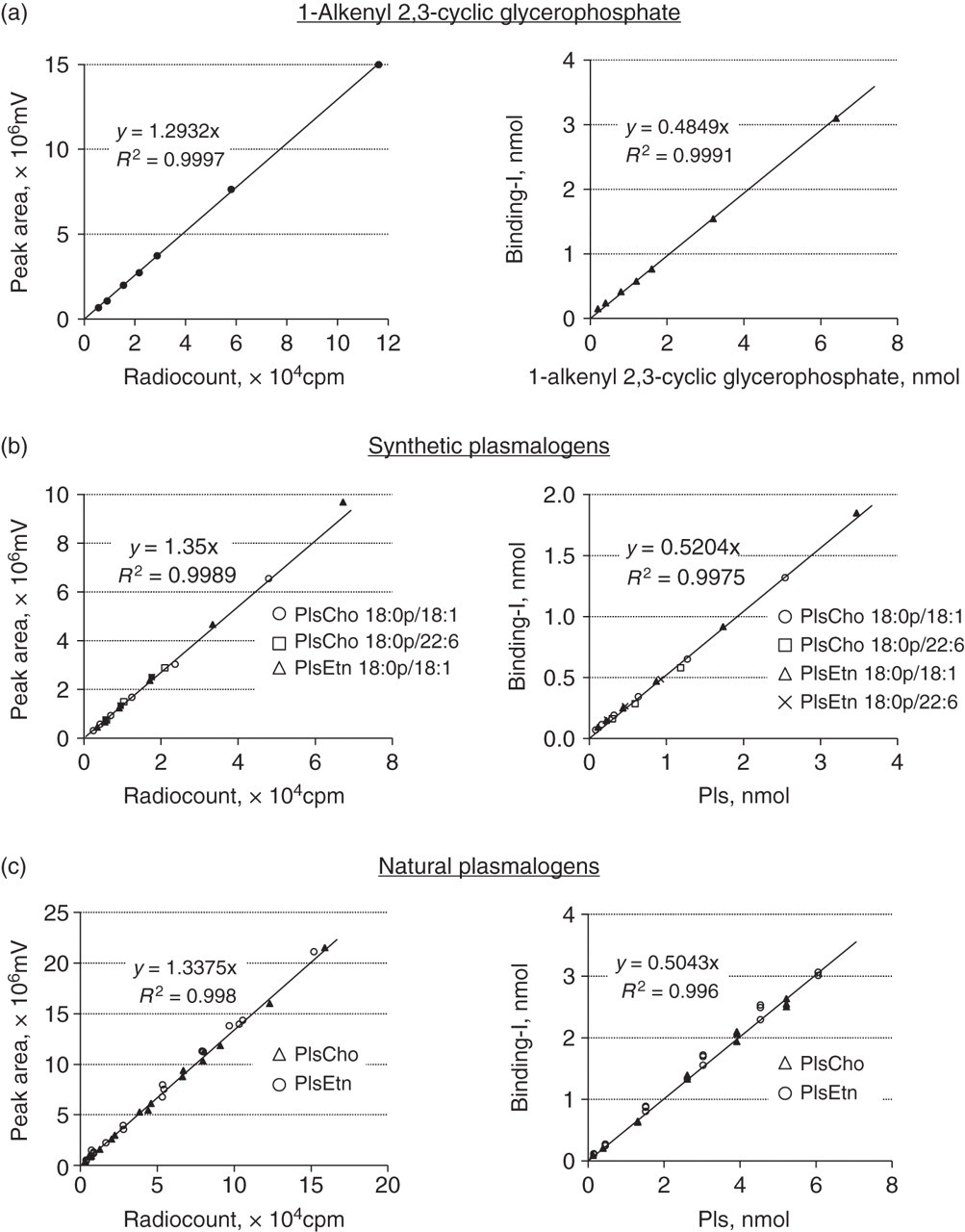
A typical HPLC elution profile of serum Pls detected with a flow γ-counter is shown in Figure 2. Subsequent to the elution of non-binding iodine (void fraction), the QS, PlsCho and PlsEtn were eluted as separate single peaks in the order. These radioactive peak fractions were collected in test tubes, and their radiocounts (cpm) were measured with a well type γ-counter (ARC-380CL; Aloka Co Ltd, Tokyo, Japan). The relationship between the radioactive peak area detected with a flow γ-counter (mV) and the radiocounts (cpm) measured with a well type γ-counter were examined for QS and Pls standards. The linear relationships, having almost the same slope, between the peak areas and radio counts were obtained in all the examined analytes (Figure 3). This indicates that the detection with a flow γ-counter is available to measure radioactivity regardless of peak shape and elution time on LC. The plots of the amounts of binding-iodine, which were estimated based on the measured radioactivity, against the actual amounts of Pls or QS show that the binding ratio of iodine to Pls or QS is exactly 1:2, as described previously.
14
This apparent binding ratio seems to be due to a radioisotopic effect. In the case of making a pair of a radiolabelling atom with a non-radiolabelling atom, the radiolabelling effect would reduce by half when only either atom in a pair combines to Pls. Therefore, the actual binding ratio of iodine (I) to Pls or QS should be regarded as 1:1, which is consistent with the result of MS analysis of iodine-binding Pls (data not shown).
A typical high-performance liquid chromatography elution profile of human serum PlsCho and PlsEtn as well as QS detected with a flow γ-counter. Column: LiChrospher 100 Diol (5 μm, 250 mm×4 mm i.d.); elute: isocratic A/B (65:35, by vol.); (A) acetonitrile; (B) acetonitrile/H2O/acetic acid/NH3 (80:19.7:0.2:0.1, by vol.); flow rate: 1 mL/min; sample injection volume: 20 μL; detection: flow γ-counter; temperature: 25°C. PlsCho, plasmenylcholine (choline plasmalogen); PlsEtn, plasmenylethanolamine (ethanolamine plasmalogen); QS, quantitative standard Linear regression analysis between peak areas on liquid chromatography (LC) and radio counts, and the analysis between the amounts of binding-iodine and plasmalogens (Pls) or quantitative standard (QS). Peak areas of each Pls standards and QS were measured by LC with a flow γ-counter, and the radio counts of the collected fractions corresponding to peak areas were determined with well-type γ-counter, and the amounts of binding-iodine were calculated based on the radioactivity of 125I, described in the Materials and methods section. Samples are as follows: (a) QS, seven different concentrations covered from 12.5 to 400 μmol/L. (b) Synthetic 18:0p/18:1- PlsCho and PlsEtn, six different concentrations covered from 5 to 160 and 7 to 217 μmol/L, respectively; 18:0p/22:6- PlsCho and PlsEtn, three different concentrations covered from 18.5 to 75 and 14.5 to 58 μmol/L, respectively. (c) Natural PlsCho from bovine heart and PlsEtn from bovine brain, six different concentrations covered from 5 to 200 and 6 to 232 μmol/L, respectively, triplicate determinations at each concentration. PlsCho, plasmenylcholine (choline plasmalogen); PlsEtn, plasmenylethanolamine (ethanolamine plasmalogen)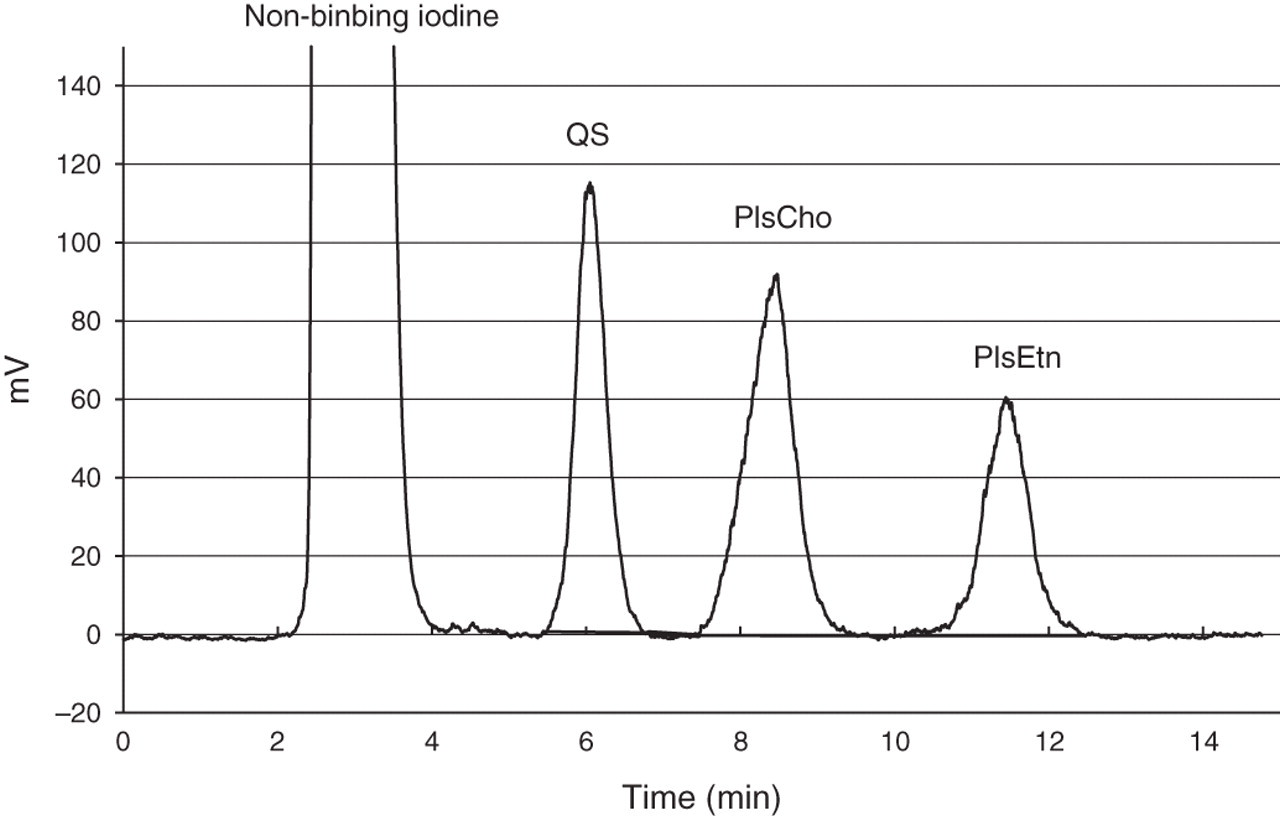
Quantification of Pls by 125I-HPLC method
Quantification was carried out as follows: (1) convert the peak area (mV) on LC detected with a flow γ-counter into the radiocounts (cpm) by using the equation to adjust for correlation between them; (2) calculate the amounts of iodine (I) bound to Pls from the radio counts based on the radioactivity of 125I, which is determined considering the decay rate of 125I (T1/2, 60.14 d); (3) estimate the amount of Pls two times as that of binding iodine because of the apparent binding ratio of iodine to Pls (1:2); and (4) correct the amount of Pls with the measurement efficiency, which is calculated from simultaneous measurement of the QS. Conveniently, it is possible to quantify Pls from the relative ratio of peak area to that of the QS.
Validation
Intra- and interday precision and accuracy for QS, PlsCho and PlsEtn
CV%, coefficient of variation percentage; MRE%, mean relative error percentage; PlsCho, plasmenylcholine (choline plasmalogen); PlsEtn, plasmenylethanolamine (ethanolamine plasmalogen); QS, quantitative standard
Stability of Pls and QS during storage
Pls standards and QS were assessed for the stability during storage in several stock solutions at different temperatures by examining the residual%. It was shown that Pls are labile in methanol regardless of thermal conditions (4, −20, −80°C), whereas they are completely stable in chloroform or diethyl ether as a stock solvent at −20°C till eight weeks' examined period. On examination of the freeze–thawing cycle, PlsEtn was shown to be more labile than PlsCho (Figure 4). QS was more stable than Pls in chloroform, and confirmed to be fully stable for more than three months at −20°C (data not shown). Pls in the serum were stable at least for three months during storage at −80°C, but thereafter they gradually decomposed. Lipid samples extracted from the serum were also sufficiently stable in chloroform as well as Pls standards. These results suggest that it is desirable to use or measure Pls standards, QS, and extracted lipids in chloroform or serum samples stored at −20°C or −80°C within three months.
Freeze–thawing test for the stability of plasmalogens (Pls). Pls standards (PlsCho and PlsEtn, each 0.4 mmol/L in MeOH, 500 μL) were tested for the stability by freezing in liquid nitrogen for one minute and thawing in water at 25°C for five minutes repeatedly. Residual % of Pls was determined by ‘cold’ iodine method (ABS) at each freeze–thawing cycle. Values are expressed as averages of duplicate determinations. PlsCho, plasmenylcholine (choline plasmalogen); PlsEtn, plasmenylethanolamine (ethanolamine plasmalogen)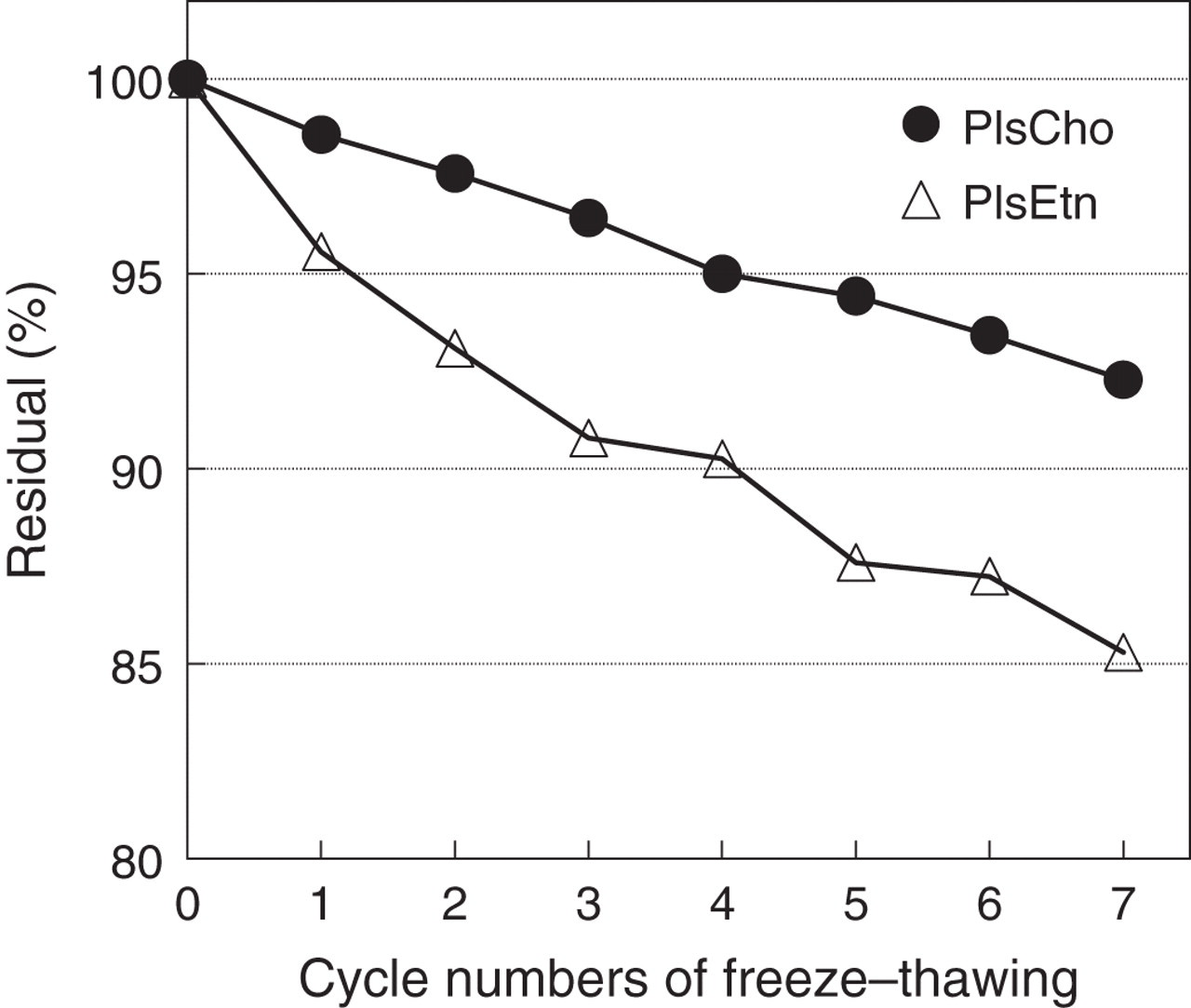
Quantification of human serum Pls
Human serum Pls concentrations from 120 healthy subjects
Mean±SD. PL, total phospholipids; PlsCho, plasmenylcholine (choline plasmalogen); PlsEtn, plasmenylethanolamine (ethanolamine plasmalogen)
Serum Pls concentrations of healthy elderly and elderly with dementia or artery stenoses
Mean±SD PL, total phospholipids; CDR, clinical dementia rating; PlsCho, plasmenylcholine (choline plasmalogen); PlsEtn, plasmenylethanolamine
Analysis of variance, significant differences between any other groups: *<0.005, **<0.001, ***<0.0001
Serum PlsCho concentrations, PlsCho/PlsEtn ratio and PL of elderly with artery stenoses were significantly reduced compared with other elderly groups. On the other hand, PlsEtn/PL% as well as total Pls/PL% of elderly with dementia was significantly decreased compared with any other groups (Table 4). These results demonstrate that the determination of serum Pls with this method is available to diagnose clinical symptoms.
We conclude that the improved 125I-HPLC method is fully validated, and is useful as a continious autoanalytical system for a routine diagnostic test of human serum (or plasma) Pls.
DECLARATIONS
