Abstract
Background
Aminoterminal propeptide of type I procollagen (PINP) reflects bone formation. Two different antigens exist in human serum: intact PINP and a monomeric form. The intact PINP assay measures trimeric form and the total assay measures both forms. The structure and origin of the monomeric form is still unclear.
Methods
Automated intact and total PINP assays were compared in breast cancer patients with (n = 60) or without bone metastases (n = 226). In addition, cross-linked carboxyterminal telopeptide of type I collagen (ICTP) was measured from the same patients and compared with the concentration of PINP monomer (difference between intact and total PINP). Monomeric PINP was purified from human ascitic fluid and characterized by sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) and matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS).
Results
The correlations were good (r > 0.948) between intact PINP and total P1NP in all patient groups. The correlation between the monomeric form and ICTP was lower in patients without bone metastases (r = 0.507) than in patients with bone metastases (r = 0.894). This indicates that the monomeric form reflects the degradation of type I collagen because bone metastases are osteolytic in nature. After several steps in the purification of the monomer form there was a single peak. Only the single band was visible in the SDS-PAGE gel. The alpha1-chain of intact PINP consists of 161 amino acids with a molecular weight of 14224.02. The purified monomer peptide in MALDI-TOF MS was smaller, 10576.41, and most likely cleaved after the arginine residue (amino acid number 120) with a trypsin-like protease.
Conclusions
Intact and total PINP assays give similar results in many conditions, but there are differences, for example in breast carcinoma, which should be recognized.
Introduction
Type I collagen is a product of two genes. COL1A1, 1 which is located in chromosome 17, encodes proα 1(I) protein chain and COL1A2, 2 located in chromosome 7, encodes proα 2(I) chain. Its precursor form, type I procollagen, is synthesized by fibroblasts in soft tissues and by osteoblasts in bone tissue, and it contains aminoterminal and carboxyterminal propeptides. 3 During the collagen maturation, these propeptides are cleaved off by specific proteases to yield type I collagen proper. Type I collagen molecule is a heterotrimer comprised of two α 1(I) chains and one α 2(I) chain, which forms a unique triple helical structure. 3 Recently, a variant of type I collagen, a homotrimer of three α 1(I) chains, was found. This variant can be found in, for example, breast carcinoma. 4
The aminoterminal propeptide of type I collagen is usually abbreviated as PINP. 5 There are two different forms of PINP antigen in human serum. 6 The larger antigen called intact PINP is a trimeric peptide which is cleaved en bloc from the procollagen. 3 The smaller antigen is called the monomeric form, of which molecular weight, fine structure and origin still remain unclear. It is thought that the monomer form could consist of a single proα 1-chain.
There are two automated assays available to measure PINP antigens. The total P1NP assay 7 measures both intact and monomeric forms of the antigen, whereas the intact PINP assay 6 measures only the trimeric form. We have previously evaluated the automated assay for intact PINP and found that this assay is very specific for intact PINP. 8
In human serum, PINP antigens contain almost only the trimeric form and hardly any monomeric form. 3,5 In certain conditions, such as chronic kidney disease or even in bedridden geriatric patients, the concentration of the monomeric form is increased. 9 In this study we are going to expand our knowledge by analysing both total and intact PINP in patients with breast cancer with or without bone metastases. Usually bone metastases in breast carcinoma are osteolytic by nature 10 and this gives us a possibility to further study the concentration of the monomeric form. This was correlated with the concentration of the ICTP (cross-linked carboxyterminal telopeptide of type I collagen), a true degradation marker of type I collagen. 9 In addition, we have purified the monomeric form of PINP antigen from ascitic fluid and measured its molecular weight.
Materials and methods
Subjects
The material in this study was obtained from patients with breast cancer with (n = 60) or without bone metastases (n = 286). The presence or absence of bone metastasis was detected with native X-ray images and verified with bone scans. Serum samples from all patients were collected during January–April 2002 and were kept frozen in −20°C. Intact PINP, total P1NP and ICTP were measured from all the samples. This study was approved by the Ethical Committee of Oulu University Hospital and conformed to the Declaration of Helsinki.
Automated assay of intact PINP
The intact PINP assay is intended for the quantitative determination of intact PINP in human serum or plasma on the IDS-iSYS Multi-Discipline automated analyser (IDS iSYS Intact PINP, Immunodiagnostics Systems, Boldon, UK). 8 The assay is based on chemiluminescence technology (CLIA). Measurements were carried out in a single analysis according to the manufacturer's kit insert instructions during May 2010. The intra- and inter-assay coefficients of variation (CVs) reported by the manufacturer are 2.6–3.0% and 4.2–5.3%, respectively.
Automated assay for total P1NP
Total procollagen type I aminoterminal propeptide (total P1NP) was measured in single analysis by the electrochemiluminescence immunoassay technique using Roche Modular analyser according to the manufacturer's instructions (total P1NP Elecsys and Cobas® analysers; Roche Diagnostics, Mannheim, Germany). 7 Serum samples were analysed during July in 2010. The intra- and inter-assay CVs reported by the manufacturer are 2.3–3.7% and 1.8–2.9%, respectively. Control samples (PreciControl Bone Level 1, 2 and 3) gave inter-assay CVs between 0.7% and 2.4% and an intra-assay variation CV of 0.8% at a level of 35.0 ng/mL (n = 5).
Cross-linked carboxyterminal telopeptide of type I collagen
ICTP antigen was measured in duplicates in the serum samples using the ICTP [125I] radioimmunoassay (RIA) kit from Orion Diagnostica (UniQ ICTP™, Espoo, Finland) according to the manufacturer's instructions. The intra- and inter-assay CVs reported by the manufacture are 4.8–9.4% and 5.6–9.0%, respectively.
Radioimmunoassay for synthetic peptide SP15
A peptide of 18 amino acids (Gln-Glu-Glu-Gly-Gln-Val-Glu-Gly-Glu-Asp-Glu-Asp-Ile-Pro-Pro-Ile-Tyr-Cys) analogous to the aminoterminal part of the α 1 chain of the procollagen of type I aminoterminal propeptide of the human type I collagen (PINP) was synthesized commercially (NeoMPS S.A., Strasbourg, France) and an extra tyrosine residue was added to facilitate radioiodination. The peptide was named SP15 and it was used to develop a competitive radioimmunoassay to measure trimeric and monomeric forms of PINP. The chloramine T method 11 was used to radioiodinate three micrograms of SP15 peptide used as a tracer antigen. Serial dilutions of SP15 were used as the reference inhibitor to construct a calibration curve. A polyclonal antiserum obtained from rabbits was previously raised to a highly purified human PINP, and it was selected for use in the immunoassay. 9
Purification of the monomeric form of the PINP from the human ascitic fluid
Ascitic fluid (700 mL) was first precipitated by 20% (NH4)2SO4 with constant stirring at +4°C overnight following centrifugation by 10,000
Statistics
A statistical analysis was performed with Analyse-it for Microsoft Excel (Analyse-It Software Ltd, Leeds, UK). Correlations were tested with the Pearson's correlation coefficient. Differences were considered significant at P < 0.05. Linear correlations and Passing–Bablok regressions 12 were used to calculate slopes and intercepts. If two methods are compared and they give similar results, the 95% confidential interval (CI) of slope should include value 1 and the 95% CI of intercept value 0.
Results
The concentrations of PINP were measured by two different automated methods from breast cancer patients with or without bone metastases. The concentration of the intact PINP (mean [SD]) was 40.1 (20.8) μg/L in patients without bone metastases and the mean concentration increased to 62.8 (57.4) μg/L in patients with bone metastases. The corresponding values measured by total P1NP were 53.4 (28.8) and 87.9 (84.2) μg/L, respectively. Thus, the total PINP assay gives in patients with breast cancer significantly higher concentrations than intact PINP.
The correlations of intact PINP and total P1NP were highly significant in all patient groups. All the correlations were above 0.9476 (Table 1). The difference plot (Figure 1) shows a few individual observations which did not fit within the 95% CI, and it seems that the other observations were not evenly spread between 95% CI bands. Passing–Bablok regressions show that the 95% CI of the slope was much below 1, and also the 95% CI of the intercept was higher than value 0 (Table 1).
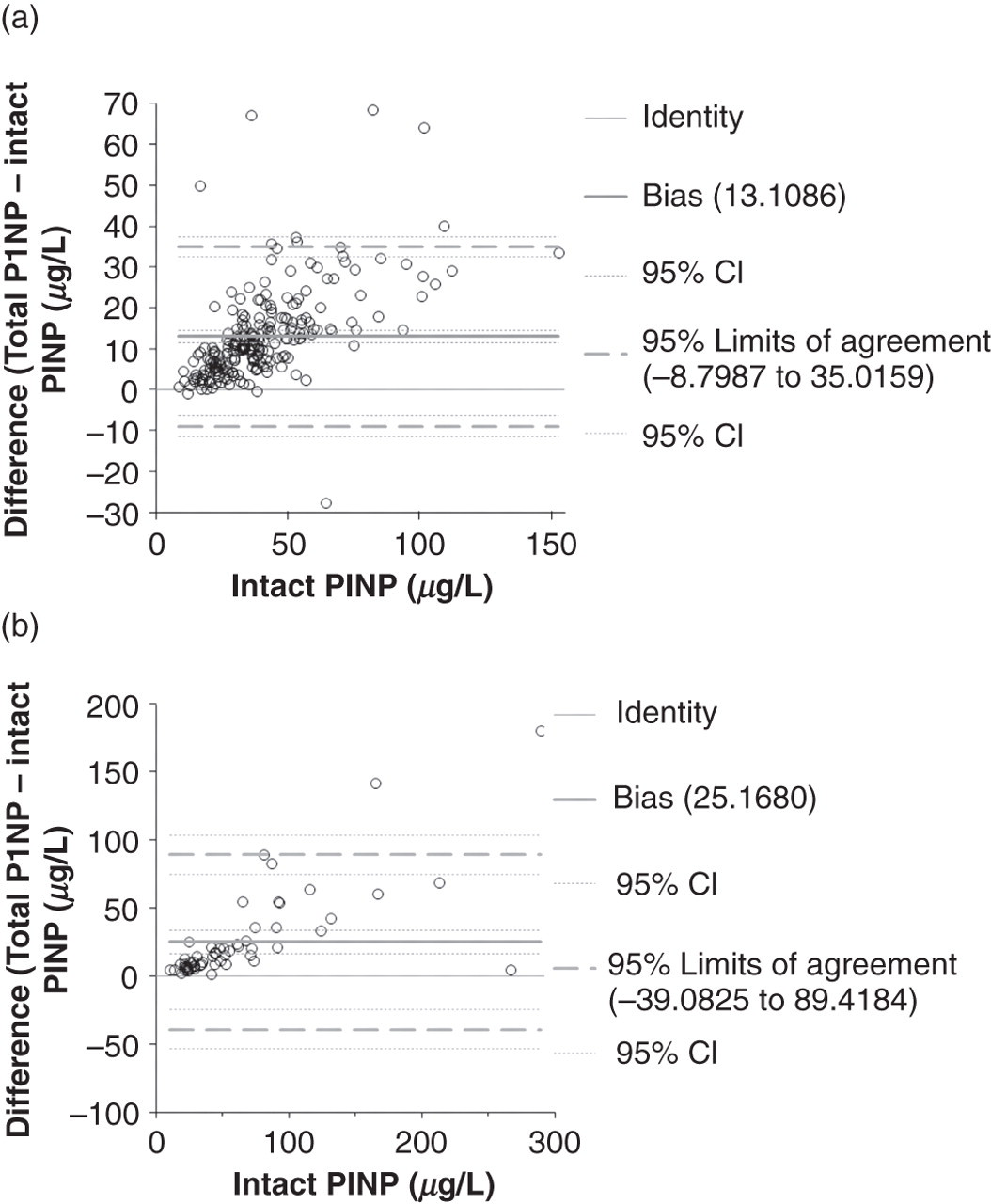
Comparison of intact PINP versus total P1NP in different patient groups of (a) patients with breast cancer and (b) patients with breast cancer and bone metastases by using difference plots. PINP, aminoterminal propeptide of type I procollagen
Pearson's linear correlations (r) and Passing–Bablok regressions of intact PINP and total P1NP grouped by different patient groups
PINP, aminoterminal propeptide of type I procollagen
In addition, ICTP was measured from all the patients and compared with the calculated concentrations of the monomer. The concentrations of the monomer were obtained by subtracting the concentration of the intact PINP from the concentration of the total P1NP. Intact PINP measures only the trimeric form and total P1NP measures the trimeric and monomeric forms, thus the concentration of the monomeric form can be calculated. ICTP correlated highly significantly with the monomer in patients with bone metastases (r = 0.894), but the correlation was lower with patients without bone metastases (r = 0.507) (Figures 2a and b). Two of the patients without bone metastases had high concentrations of ICTP (38.2 and 39.5 μg/L) without high amounts of monomeric PINP. The former patient had uraemia, which resulted in kidney transfer and the latter patient died within three weeks. We have previously observed that ICTP is highly increased when a person is about to die. 13
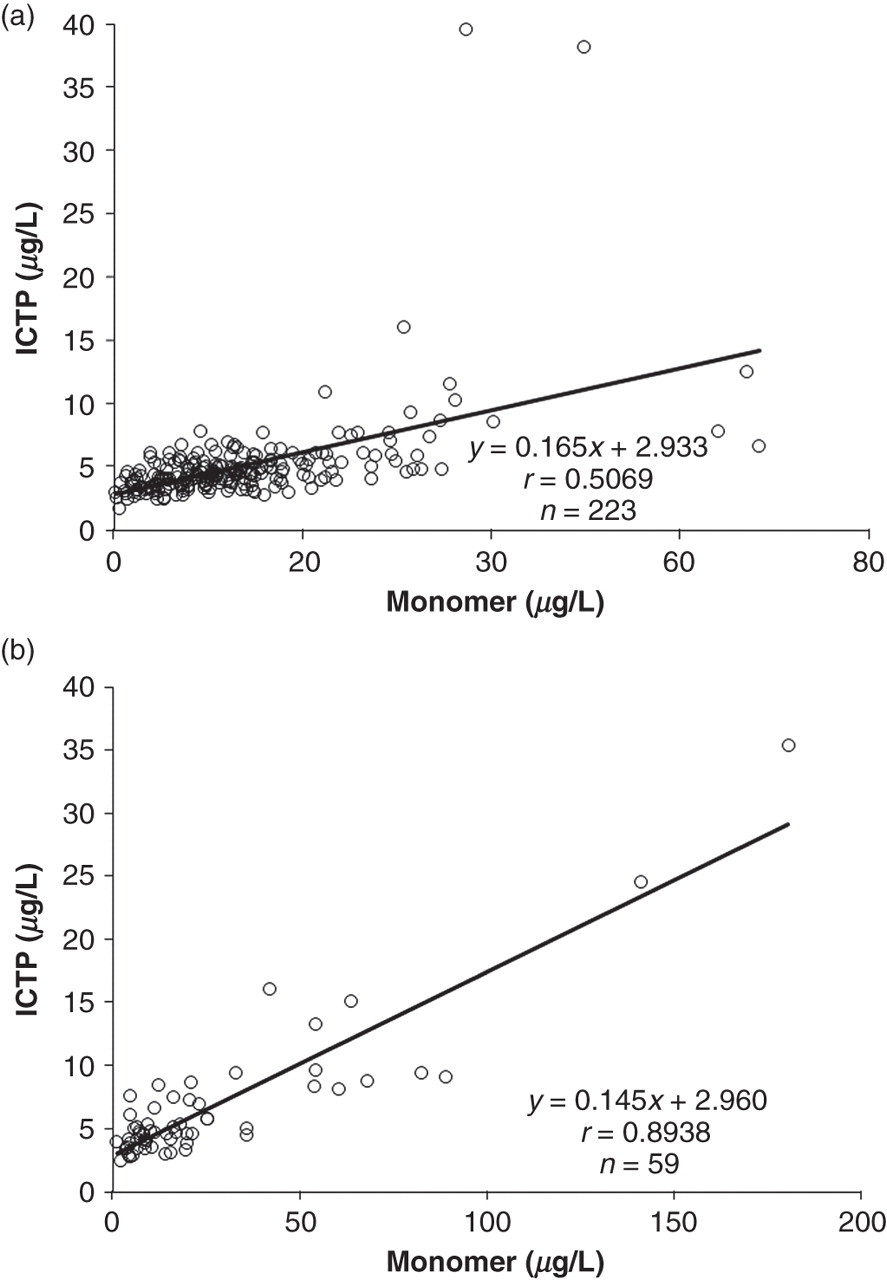
Regression charts of measured ICTP versus calculated monomer in patient group of (a) patients with breast cancer and (b) patients with breast cancer and bone metastases. ICTP, cross-linked carboxyterminal telopeptide of type I collagen
The purification of the monomeric PINP was carried out by using several chromatographic techniques. The SP15 radioimmunoassay, which measures monomeric and trimeric forms of PINP, was used to monitor the location of the monomer. Sephacryl S-100 size exclusion chromatography resulted in two peaks, the first peak containing traces of trimeric PINP and the second peak monomeric PINP. Monomer fractions were pooled and further separated by C8 RP-HPLC. The single fraction of the highest monomeric concentration was characterized by SDS-PAGE and MALDI-TOF MS. Only a single band was visible in the SDS-PAGE gel. The alpha1-chain of PINP consists of 161 amino acids with molecular weight of 14224.02. The molecular weight of the purified monomer peptide in MALDI-TOF MS was 10576.41 (Figure 3). Thus, the alpha1-chain of PINP was cleaved after Arg (120), most likely by a trypsin-like protease.
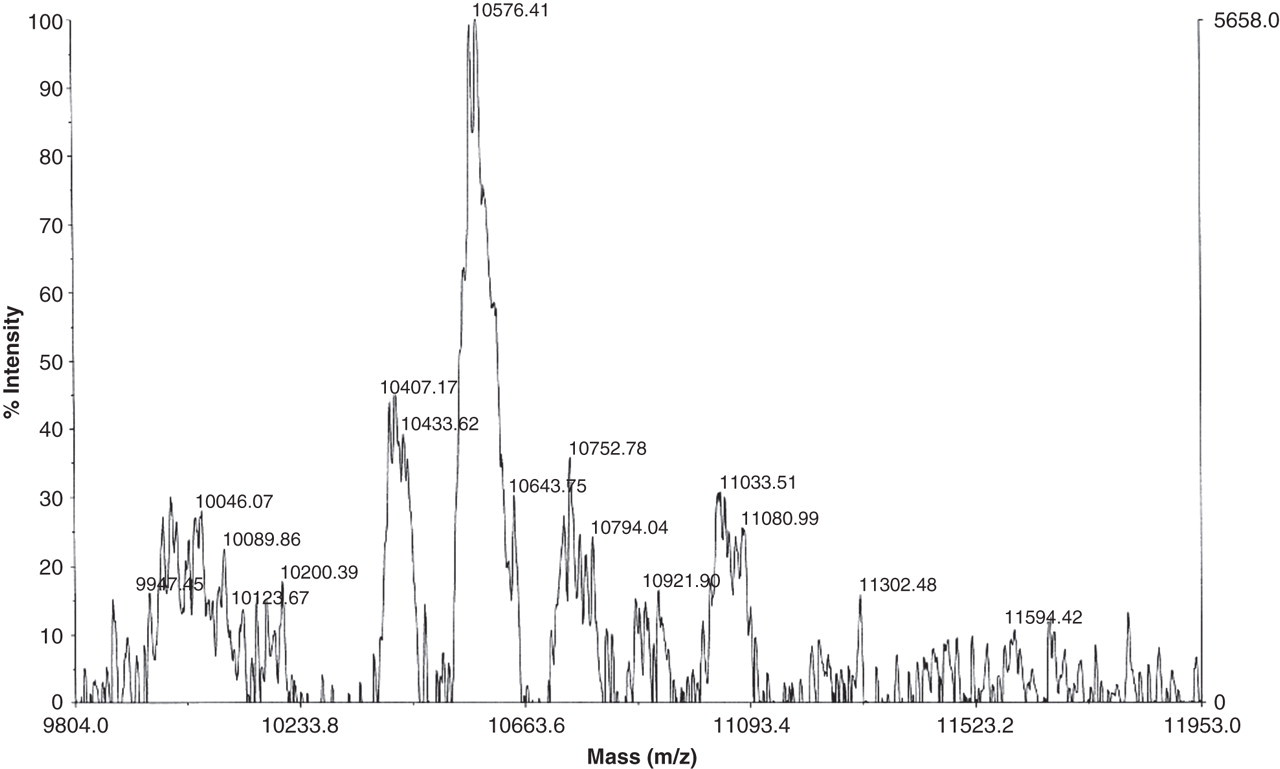
Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry analysis of the purified monomeric aminoterminal propeptide of type I procollagen
Discussion
In healthy individuals, the intact and total PINP assays give similar results as reported previously. 7 However, especially in haemodialysis patients, the results were very different. 9 We have found out that the difference was due to a large increase of the monomeric forms, which accumulate in the serum when the kidneys are not functioning. The accumulation of the breakdown products caused by decreased kidney function is a normal phenomenon. The clearances of the trimeric and monomeric forms of PINP are different. Intact PINP is rapidly taken up by liver endothelial cells, but the clearance of the monomeric form is probably dependent on kidney function. 8,9 However, the accumulation of the monomeric form could induce problems when the assay results are interpreted. We previously tested bedridden old patients who most likely have an increased catabolism of connective tissue. We measured serum ICTP concentration in the same samples and observed that many of the tested patients had increased ICTP concentrations as reported previously in our laboratory. 13
In this study we tested another patient group which could have increased catabolism of connective tissue. In fact, breast cancer is considered a typical osteolytic tumour. 14 Although the correlations between intact and total PINP were highly significant in different groups of patients (Table 1), we observed differences in a detailed analysis. In the difference plot the proportion of monomer (total–intact PINP) was increased when the synthesis rate of type I procollagen was increasing (Figure 1). Thus, in the Passing–Bablok regressions the slope did not contain value 1, which means that these methods do not measure the same antigens. In addition, the correlation between monomer and ICTP was highly significant (Figure 2). Since ICTP concentration reflects increased catabolism of type I collagen, the results indicate that the monomeric form originates from tissue collagen breakdown.
In this study, we purified the monomeric form from the ascitic fluid and found out that this fragment was a truncated peptide of α 1(I) chains of PINP. The purification was followed by a variant of the PINP assay (SP15 RIA), which detects only the aminoterminal parts of the monomer. Molecular weight analysis reveals that this purified monomer was cleaved after arginine between a globular domain that resembles a von Willebrand factor C repeat and a triple helical domain. This result means that an active enzymatic function is needed to produce the monomeric fragments isolated from ascitic fluid. Since it was possible to purify intact PINP from the same ascitic fluid, it was not likely that the intact form of PINP could readily be denaturized by heat into the monomeric form. 15
This study shows that although intact and total PINP can give similar results, there are differences that should be recognized. In breast cancer especially with bone metastases, the total PINP assay gives higher concentrations than intact PINP. This difference is due to the fact that the total PINP assay measures both the intact PINP and monomeric form which is not derived from synthesis of type I procollagen but from the degradation of the whole procollagen instead. 6,8,9
DECLARATIONS
