Abstract
Background
According to consensus documents, sweat testing remains the standard for diagnosing cystic fibrosis. We studied a novel method of sweat analysis on two chemistry platforms: Dimension Vista (Siemens Healthcare Diagnostics) and Aeroset (Abbott).
Methods
Sweat collected from healthy individuals and aqueous samples in the critical range for sweat analysis were measured using the Orion 290 plus (Thermo Electron Corporation), Chiron 925 chloride analyser (Sherwood Scientific Ltd), flame photometry, sweat-chek analyser and indirect ion-selective electrodes (ISE) of Vista and Aeroset. For measurement on ISE, an addition protocol was designed.
Results
Within-run and between-run variation coefficients of Aeroset and Vista remained <5% as prescribed by UK guidelines. The correlation between the chloride analyser and chloride concentrations analysed on Vista or Aeroset was, respectively, y = 0.96x + 7.61 (r 2 0.990) and y = 0.97x + 1.10 (r 2 0.930) and between sweat-chek (conductivity) and sodium concentrations analysed on Vista or Aeroset was, respectively, y = 1.08x - 4.0 (r 2 0.967) and y = 1.13x - 1.00 (r 2 0.939). When classified according to medical decision rules (30 mmol/L and 60 mmol/L chloride, combined with sodium concentration), the same classification was found with the ISE-module and conventional methods for all samples.
Conclusion
We describe a simple procedure for sweat analysis using the ISE-module of two different chemistry platforms. This procedure meets the requirements as described in UK guidelines.
Introduction
According to both US and UK consensus documents, sweat testing remains the standard for diagnosing cystic fibrosis (CF). 1,2 In general, chloride is the most important electrolyte in the diagnosis of CF since this ion discriminates best between CF patients and healthy individuals. Concurrent sodium measurement, however, is recommended as an internal quality control. 3 Although in the past, methods for simultaneous determination of sodium and chloride using ion-selective electrode (ISE) technology have been evaluated, 4,5 the use of indirect ISE as found on routine chemistry analysers is still very uncommon. Problems such as insufficient precision and accuracy of ISE-modules in the critical range for sweat analysis combined with the small volume of the sweat samples seem to outweigh the benefits of the ISE. Indirect ISE methods described thus far require dedicated equipment and use relatively large amounts of sample. 4,5 In the present paper, we describe a novel method of sweat analysis on two chemistry platforms, which overcomes these problems and meets the requirements published in the UK guidelines. 1
Materials and methods
Two different methods were compared for measuring chloride in the sweat samples: the Chiron 925 chloride analyser (Sherwood Scientific Ltd, Cambridge, UK) and an indirect ISE in the urine module of Aeroset (Abbott, IL, USA) and Dimension Vista 1500 (Siemens Healthcare Diagnostics, Deerfield, IL, USA). The sodium content of all samples was determined using an ISE (Aeroset and Vista) and was compared with conductivity measurement on the Sweat-chek analyser (Model 3100, Wescor, Logan, UT, USA). Moreover, chloride and sodium measurement on the Aeroset was compared with chloride measurement on the Orion 290 plus (Thermo Electron Corporation, Beverly, CA, USA) and sodium measurement on the flame photometer (ISE IL 943, Instrumentation Laboratory BV, Breda, the Netherlands). All devices were routinely calibrated and controlled using lyphochek and liquichek 1 and 2 (Bio-Rad, Hercules, CA, USA) and homemade internal controls. The sample volume required for measurement of chloride and sodium on the ISE-module is 80 μL for Dimension Vista and 100 μL for Aeroset. The minimal volume of a correctly collected sweat sample with the Macroduct® coil system (Wescor) is 20 μL. Dilution of the sweat sample to provide enough sample for measurement on Aeroset and Vista would result in sodium and chloride concentrations below the lowest detection limit of the ISE-modules (5 mmol/L Na and 10 mmol/L Cl). An addition protocol was designed to ensure linearity and to meet detection limits of the ISE-modules. Both for Aeroset and Vista, a 20 μL sample was added to an aqueous standard of 40 mmol/L NaCl to achieve a total volume of 100 μL for the Aeroset and 80 μL for the Vista, sufficient for a duplicate measurement of sodium and chloride. In every run three aqueous standards (25, 50 and 90 mmol/L NaCl) and two control samples (lyphochek 1 and 2) were measured using the addition protocol. The values measured for the aqueous standards were plotted against the concentration of NaCl that was originally dissolved for the aqueous standard and a regression line was calculated. This regression line was used to calculate the chloride or sodium concentration in the original control or sweat sample.
Leftover sweat samples were used, collected from nine healthy adults using the Macroduct® coil system for sweat collection after stimulation in the presence of pilocarpine. A minimum of 20 μL sweat was collected per stimulation. Since it is difficult to collect sweat samples covering the full critical range for sweat analysis, 23 aqueous samples were prepared by dissolving NaCl (Merck KgaA, Darmstadt, Germany) in distilled water.
For interpretation of the results, medical decision rules were used as published in the Dutch guidelines for diagnostics and treatment of CF. According to these rules, patients with a chloride concentration above 60 mmol/L and [chloride] > [sodium] are classified as strongly suspect of CF. Patients with a chloride concentration between 30 and 60 mmol/L and [chloride] > [sodium] possibly are CF patients and a new sweat sample should be collected to repeat analysis. When chloride concentration is below 30 mmol/L or between 30 and 100 mmol/L and [chloride] < [sodium + 10 mmol/L], the patient should be classified as CF negative.
EP Evaluator (Data innovations, South Burlington, VT, USA) was used to evaluate data obtained for linearity, precision and correlation.
Results
Within-run and between-run precision was assessed according to CLSI EP5 protocol. For this, aqueous standards of 30 and 60 mmol/L were measured two times in duplicate every day during five days. Variation coefficients (CVs) for sodium measurement at a sodium concentration of 30 mmol/L and 60 mmol/L were, respectively, 4.7% and 4.6% (Dimension Vista), 4.6% and 4.8% (Aeroset) and 5.5% and 3.4% (flame photometry). The Sweat-chek measures conductivity with a CV of 5.0% at 30 mmol/L sodium and a CV of 2.2% at the level of 60 mmol/L sodium. At 30 and 60 mmol/L chloride the CVs are, respectively, 4.8% and 4.5% (Vista), 2.9% and 2.3% (Aeroset), 2.5% and 1.8% (Chiron 925 chloride analyser), and 6.6% and 4.7% (Orion 290 plus). Aeroset and Vista passed the tests with a CV < 5% as prescribed by UK guidelines. Between-run precision over five days showed mean values on Vista and Aeroset of, respectively, 38.3 and 38.5 mmol/L for sodium and 39.2 and 36.9 mmol/L for chloride. The correlation between traditional methods and ISE-modules was determined using CLSI EP9. Twenty-three aqueous standards and nine sweat samples were measured in duplicate. The regression equation calculated for the Chiron 925 chloride analyser and Vista or Aeroset was, respectively, y = 0.96x + 7.61 (r 2 0.990) (Figure 1) and y = 0.97x + 1.10 (r 2 0.930), and between Sweat-chek and Vista or Aeroset, y = 1.08x − 4.0 (r 2 0.967) and y = 1.13x − 1.00 (r 2 0.939). The regression equation calculated for the Aeroset and Orion 290 plus was y = 0.72x + 1.89 (r 2 0.954), and the Aeroset and flame photometer y = 0.98x + 3.50 (r 2 0.976). When classified according to the medical decision rules published in the Dutch guidelines, the same classification was found with the novel ISE method and traditional methods.
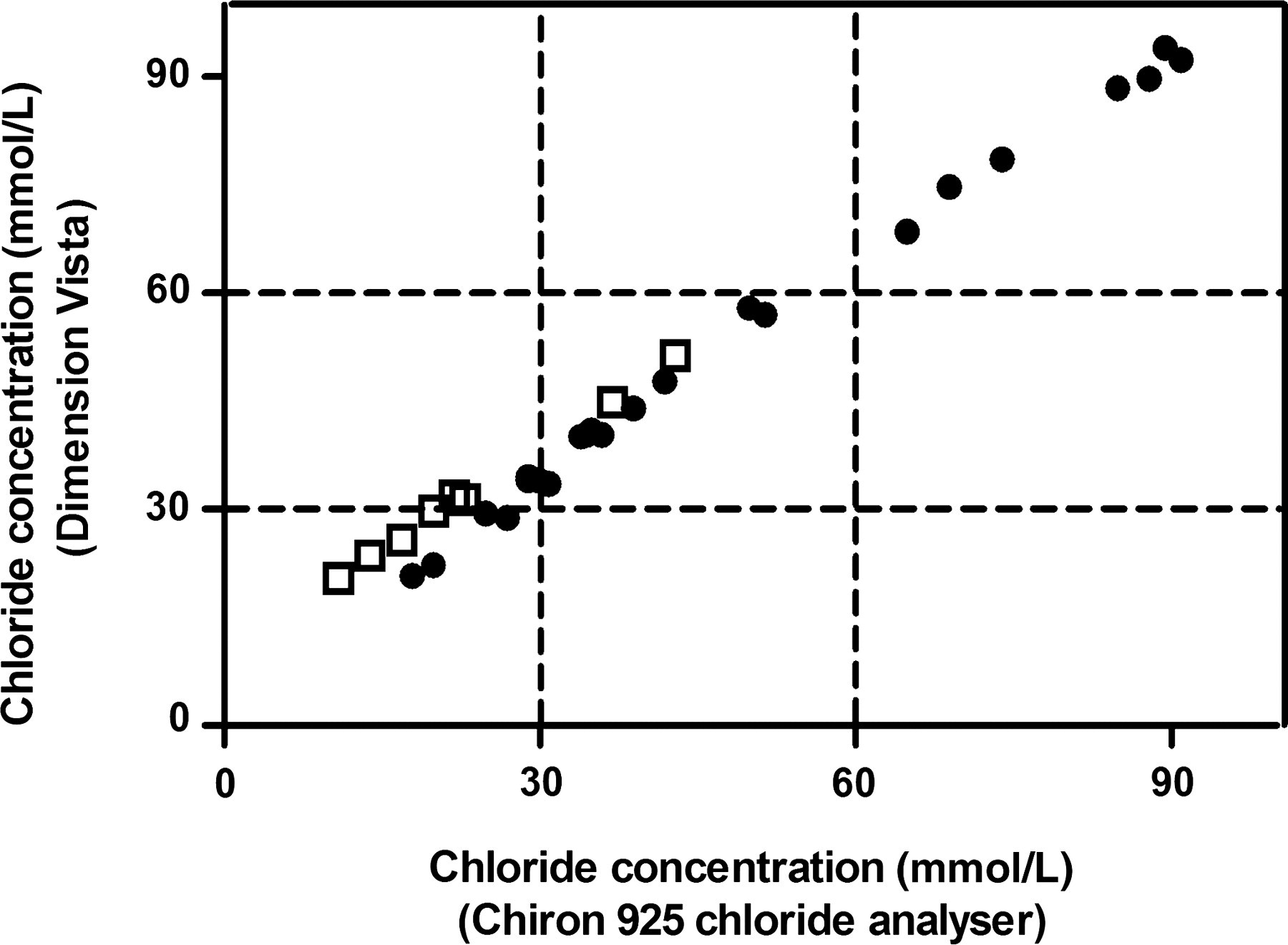
Correlation between the Chiron 925 chloride analyser and Dimension Vista using nine sweat samples (open squares) and 23 aqueous samples (closed circles). The dotted lines indicate the chloride concentration according to the medical decision rules published in Dutch guidelines for sweat analysis
Discussion
Currently, most laboratories use dedicated equipment for sweat analysis, such as the flame photometer and the Chiron 925 chloride analyser. This demands special expertise and is time consuming. In the present study, we show that the indirect ISE-module of regular chemistry platforms provides a good alternative to the dedicated equipment. Linearity and precision of electrolyte measurement on the ISE-module meets the standards described in the guidelines for the performance of the sweat test by Green et al. 1 and correlates well with the commonly used methods.
Qualitative correlation between the Chiron 925 chloride analyser and Vista shows a significant intercept (Figure 1). Due to this intercept, five samples would be classified differently according to the medical decision rules. However, when sodium measurements within the same samples are also taken into account as recommended 3 and described in the Dutch clinical decision rules, classification of these five samples is the same as found using the conventional methods. Interpretation of the data provided by the Aeroset resulted in the same classification for all samples as found with the conventional methods.
Regular chemistry platforms with an indirect ISE-module are present in many clinical laboratories. The use of these platforms for sweat analysis saves the costs of dedicated devices for sweat analysis, is simple and therefore claims less staff time. Moreover, the indirect ISE-module provides a means to measure sodium and chloride at the same time in the same sample in duplicate, even though sample size is limited. The method has been implemented successfully on two different chemistry platforms. Moreover, pilot studies with Beckman LX and Roche Integra systems show similar results as found with Vista and Aeroset. This indicates that the procedure can be used more universally.
DECLARATIONS
