Abstract
Background
Antithyroglobulin antibodies are a prevalent cause of interference in serum thyroglobulin immunoassays. Current guidelines recommend that antithyroglobulin antibodies should be measured concurrently with thyroglobulin when monitoring thyroid cancer patients post‐thyroidectomy. However, the concordance between different antithyroglobulin assays has been questioned despite the availability of an international thyroglobulin antibody Reference Preparation.
Methods
Four antithyroglobulin assays currently in use in UK laboratories (Siemens Immulite®, Brahms GmbH, PerkinElmer AutoDELFIA and Siemens ADVIA Centaur®) were compared in a cohort of 145 thyroid cancer patients.
Results
Using reference data provided by the kit manufacturer, concordance between the assays was 74%. Adjusting the cut-offs to maximize agreement increased concordance to 90%. Recovery of exogenous thyroglobulin using the Brahms Tg-plus immunoradiometric assay was neither a specific nor a sensitive test for the presence of a positive antibody result by any assay.
Conclusions
Despite the availability of an international reference preparation, current antithyroglobulin assays show unacceptable variance.
Introduction
Current guidelines
1
recommend that thyroglobulin antibodies (TgAb) should be measured concurrently with thyroglobulin (Tg) when monitoring thyroidectomized thyroid cancer patients to alert the laboratory to the possibility of assay interference. The concordance between different TgAb assays has been questioned despite the availability of an International TgAb Reference Preparation (MRC65/93 –
Methods
Four TgAb assays in current use in UK laboratories were used to test samples from 145 patients attending the Addenbrooke's Thyroid Cancer Clinic. All samples were assayed using the Brahms anti-TG radio-ligand immunoassay (Br) provided by Brahms GmbH (Hennigsdorf, Germany), AutoDELFIA time-resolved non-competitive sandwich fluoroimmunoassay (De; Perkin–Elmer, Turku, Finland), Siemens ADVIA Centaur® chemiluminescent competitive immunoassay (Ce; CA, USA) and Siemens Immulite® enzyme labelled chemiluminescent immunometric assay (Im). The De and Im assays were calibrated to MRC65/93, and the Ce assay calibrated to MRC65/93 with a quoted conversion factor of 2.8. The Br assay was calibrated to CRM 457 with a conversion factor of 2. The analytical sensitivity of each assay was 27 U/mL, 10 U/mL, 1 IU/mL and 2.2 IU/mL for the Br, Ce, De and Im assays, respectively.
Recovery of exogenous Tg was ascertained using the Brahms Tg-plus immunoradiometric assay using the protocol provided. Essentially either 2 or 100 ng/mL excess Tg is added to samples with an original concentration of less than or greater than 10 ng/mL original, respectively, and the Tg recovery calculated.
Adjusting the manufacturer's reference ranges using a manual iterative method increased concordance between TgAb assays.
Results
Results from the TgAb methods showed significant variance (P < 0.0001 Freidman rank test). Seventeen patients were positive and ninety negative for TgAbs by all four assays using criteria provided by the manufacturers (De 35 IU/mL, Im 40 IU/mL, Ce 60 U/mL and Br 100 IU/mL). This gave a concordance between assays of 74% (Figure 1a). Twenty-two patients were positive by one assay alone, eight by two assays and eight by three assays.
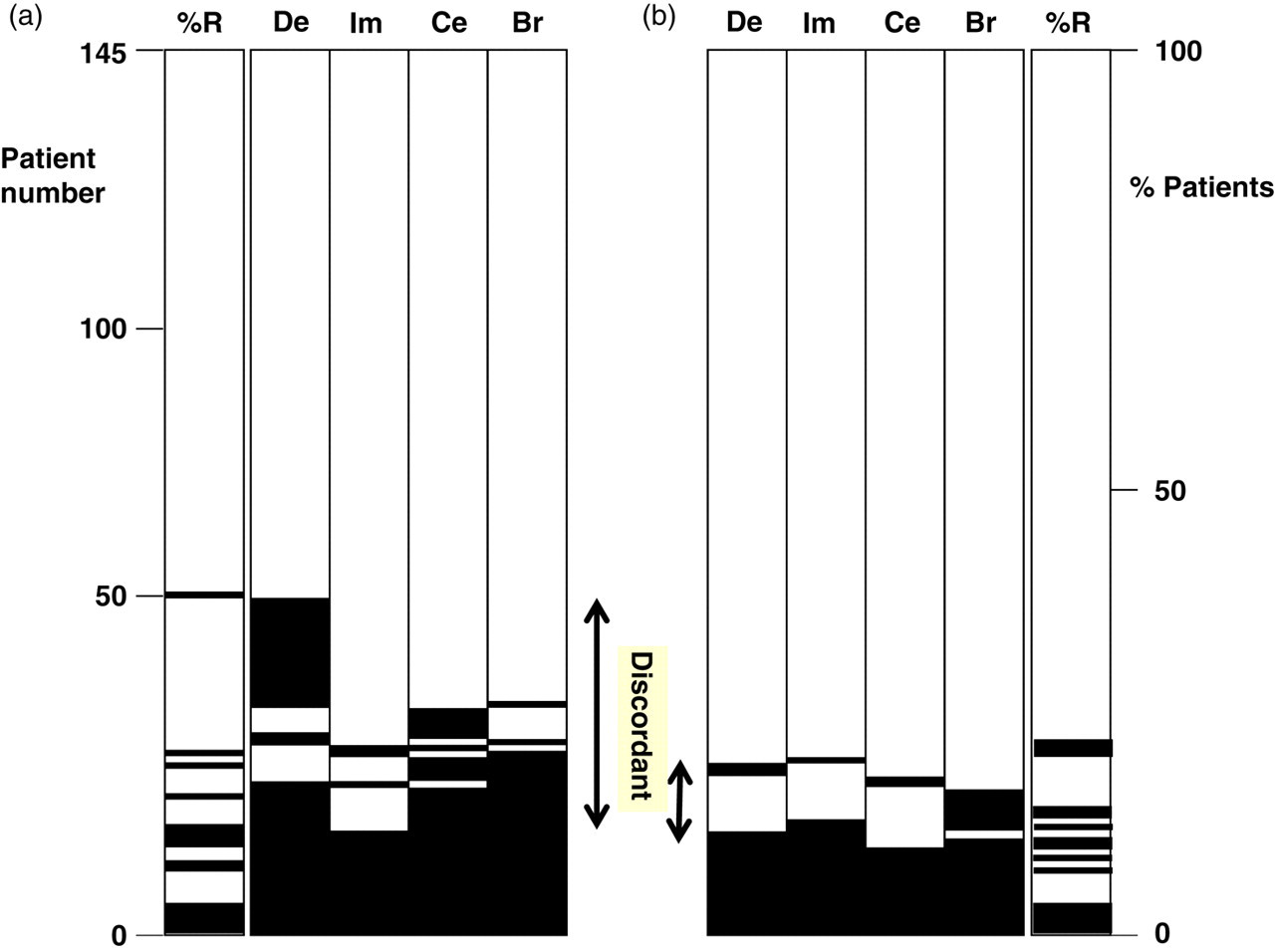
Concordance between TgAb assays and Tg recovery. Black bars show positive results for each patient, with patient number on the y-axis. The x-axis shows the assay: De, DELFIA; Im, Immulite; Ce, Centaur; Br, Brahms; %R, recovery. Panel a shows manufacturers’ cut-offs, panel b the optimized cut-offs. TgAb, thyroglobulin antibodies; Tg, thyroglobulin
Fourteen patients showed <70% recovery of exogenous Tg (Figure 1a) using the Brahms Tg assay (manufacturer's cut-off). Of these 14, one patient did not have a positive TgAb result by any assay. Tg recovery was >70% in seven patients with positive TgAb results by all four assays.
The overall concordance could be increased to 90% by adjusting cut-offs (De 80 IU/mL, Ce 160 U/mL, Br 120 U/mL, Im unchanged 40 IU/mL – Figure 1b).
Discussion
The TgAb methods in this study do not agree well despite the availability of the international standard. This study complements the work of Krahn and Dembinski 2 who compare three TgAb assays and an earlier study by Spencer et al. 3 looking at 12 different TgAb methods. While a direct comparison of these studies is not possible, agreement between assays was 17/55 (four assays, this study), 3/54 (three assays) 2 and 4/42 (12 assays) 3 in samples that were positive by any of the assays used.
Adjusting assay cut-offs can increase concordance between the assays used in this study; however, in lieu of a gold standard Tg assay, the effects of these adjustments on the ability to detect significant Tg assay interference cannot be determined. For similar reasons, it is also difficult to ascertain whether different Tg assays are more or less susceptible to TgAb interference.
Using Tg recovery as an alternative to TgAb assay was neither a specific nor a sensitive test for the presence of TgAbs in this cohort.
A major obstacle to solving this diagnostic challenge is the heterogenous nature of TgAbs. While antibody interference is well described in serum immunoassay methods, common strategies to circumvent this problem, in particular by immunosubtraction, are unlikely to work in this context. Tg must be dissociated from the interfering TgAb prior to assay as any attempt to deplete immunoglobulin will also remove the analyte in question.
As assay interference is dependent on assay architecture, single-site immunoassays for Tg are an alternative approach to this problem. Clearly, a discrepancy between these two types of assay is a good indication for the presence of assay interference 5 and this is recommended in current guidelines. 1 However, in this situation, neither assay may provide the correct Tg concentration.
Trypsin digestion and immunocapture of Tg-specific peptides followed by mass spectrometric analysis have been proposed as a possible solution for antibody interference, 6 but currently these methods do not have the required sensitivity for clinical applications and are relatively cumbersome compared with sandwich immunoassays.
This study is in agreement with previous work suggesting that TgAbs are prevalent in patients with thyroid cancer and the likelihood of interference in Tg immunoassay is high. However, current methods for TgAb quantitation do not agree well, and the relation between antibody concentration and the presence of TgAb interference in Tg assays remains obscure. Given a negative TgAb result, we do not know how much reassurance can be provided to clinicians concerning the validity of serum Tg estimations.
Footnotes
DECLARATIONS
